Abstract
Background
Studies regarding the discrimination of non-neoplastic lesion, adenoma, and cancer of the stomach using magnifying endoscopy with flexible spectral imaging color enhancement system (FIME) in each different channel of that system have not yet been reported.
Aims
To ascertain the usefulness of FIME discriminating among the three kinds of gastric lesions.
Methods
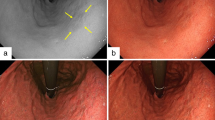
When finding a lesion to be a possible neoplasm using conventional endoscopy, the examiner discriminated among the three kinds of gastric lesions by observing the pit pattern on the lesion using magnifying endoscopy with white light (WLME) and then recorded the image. The procedure was repeated three more times with FIME with channel 0, 2, and 4. The four recorded images per lesion were then given to four raters for discriminating among the three kinds of gastric lesions.
Results
The proportion of agreement and the degree of agreement between endoscopic and pathological diagnosis (AEP) by WLME were 0.85 and 0.76, respectively, and those by FIME were 0.91 and 0.86, respectively. All AEPs from discriminations with FIME was higher than that with WLME. AEPs from the discriminations with FIME with channel 4 were higher than those with FIME with channel 0 or 2. The degree of interobserver agreement among the results of the four raters was 0.42 for WLME and 0.50–0.59 for FIME with each of the three different channels.
Conclusions
FICE is useful in discriminating among non-neoplastic lesions, adenoma, and cancer of the stomach, and channel 4 of the FICE is better than channel 0 and 2 for the discriminations.



Similar content being viewed by others
References
Goetz M, Kiesslich R. Advanced imaging of the gastrointestinal tract: research vs. clinical tools? Curr Opin Gastroenterol. 2009;25:412–421.
Pohl J, May A, Rabenstein T, Pech O, Ell C. Computed virtual chromoendoscopy: a new tool for enhancing tissue surface structures. Endoscopy. 2007;39:80–83.
Goda K, Tajiri H, Ikegami M, et al. Magnifying endoscopy with narrow band imaging for predicting the invasion depth of superficial esophageal squamous cell carcinoma. Dis Esophagus. 2009;22:453–460.
Nonaka K, Arai S, Ban S, et al. Prospective study of the evaluation of the usefulness of tumor typing by narrow band imaging for the differential diagnosis of gastric adenoma and well-differentiated adenocarcinoma. Dig Endosc. 2011;23:146–152.
Tischendorf JJ, Wasmuth HE, Koch A, Hecker H, Trautwein C, Winograd R. Value of magnifying chromoendoscopy and narrow band imaging (NBI) in classifying colorectal polyps: a prospective controlled study. Endoscopy. 2007;39:1092–1096.
Kato M, Kaise M, Yonezawa J, et al. Magnifying endoscopy with narrow-band imaging achieves superior accuracy in the differential diagnosis of superficial gastric lesions identified with white-light endoscopy: a prospective study. Gastrointest Endosc. 2010;72:523–529.
Ezoe Y, Muto M, Horimatsu T, et al. Magnifying narrow-band imaging versus magnifying white-light imaging for the differential diagnosis of gastric small depressive lesions: a prospective study. Gastrointest Endosc. 2010;71:477–484.
Yoon WJ, Lee DH, Jung YJ, et al. Histologic characteristics of gastric polyps in Korea: emphasis on discrepancy between endoscopic forceps biopsy and endoscopic mucosal resection specimen. World J Gastroenterol. 2006;12:4029–4032.
Dinis-Ribeiro M, da Costa-Pereira A, Lopes C, et al. Magnification chromoendoscopy for the diagnosis of gastric intestinal metaplasia and dysplasia. Gastrointest Endosc. 2003;57:498–504.
Tanaka K, Toyoda H, Kadowaki S, et al. Surface pattern classification by enhanced-magnification endoscopy for identifying early gastric cancers. Gastrointest Endosc. 2008;67:430–437.
Ohnita K, Isomoto H, Shikuwa S, et al. Magnifying chromoendoscopic findings of early gastric cancer and gastric adenoma. Dig Dis Sci. 2011. (Epub ahead of print). doi:10.1007/s10620-011-1638-6.
Japanese Gastric Cancer Association. Japanese classifications of gastric carcinoma-2nd English edition. Gastric Cancer. 1998;1:10–24.
Schlemper RJ, Riddel RH, Kato Y, et al. The Vienna classification of gastrointestinal epithelial neoplasia. Gut. 2000;47:251–255.
Cohen J. A coefficient of agreement for nominal scales. Educat Psychol Meas. 1960;20:37–46.
Fleiss JL. Statistical methods for rates and proportions. 2nd ed. New York: Wiley; 1981.
Participants in the Paris workshop. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003;58:S3–43.
Tajiri H, Doi T, Endo H, et al. Routine endoscopy using a magnifying endoscopy for gastric cancer diagnosis. Endoscopy. 2002;34:772–777.
Coriat R, Chryssostalis A, Zeitoun JD, et al. Computed virtual chromoendoscopy system (FICE): a new tool for upper endoscopy? Gastroenterol Clin Biol. 2008;32:363–369.
Mouri R, Yoshida S, Tanaka S, Oka S, Yoshihara M, Chayama K. Evaluation and validation of computed virtual chromoendoscopy in early gastric cancer. Gastrointest Endosc. 2009;69:1052–1058.
Tahara T, Shibata T, Nakamura M, et al. Gastric mucosal pattern by using magnifying narrow-band imaging endoscopy clearly distinguishes histological and serological severity of chronic gastritis. Gastrointest Endosc. 2009;70:246–253.
Okubo M, Tahara T, Shibata T, et al. Changes in gastric mucosal patterns seen by magnifying NBI during H. pylori eradication. J Gastroenterol. 2011;46:175–182.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, S.W., Lim, K.S., Lim, J.U. et al. Flexible Spectral Imaging Color Enhancement (FICE) Is Useful to Discriminate Among Non-neoplastic Lesion, Adenoma, and Cancer of Stomach. Dig Dis Sci 56, 2879–2886 (2011). https://doi.org/10.1007/s10620-011-1831-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1831-7