Abstract
Aim
Several models of experimental ulcerative colitis have been previously reported. To date, only one model of Crohn’s colitis that was induced by trinitrobenzene sulfonic acid has been described.
Materials and Methods
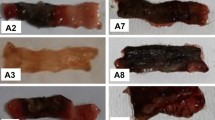
Twenty-one male albino rats were divided into three groups: group I: sham control group (n = 7), group II: acetic acid group (n = 7), group III: NaOH group (n = 7). On the day of induction, all rats were lightly anesthetized with intramuscular ketamine (8 mg/kg). A 6F plastic catheter was inserted rectally until the tip was 5 cm proximal to the anus. Then, 2 ml of 0.9% saline, 2 ml of 4% acetic acid, and 2 ml of 6.25% NaOH was administered to groups I, II, and III, respectively. All rats were sacrificed 5 days after colitis induction. The distal colon segment was assessed macroscopically and microscopically. In addition, malondialdehyde (MDA) and nitric oxide (NO) levels of the colonic tissue and changes in body weight were measured.
Results
Macroscopic and microscopic examinations of colonic tissue samples showed morphological similarities to human Crohn’s disease (CD). The MDA and NO levels of the colonic tissues were significantly higher in the NaOH group compared to the acetic acid and sham control groups (P = 0.001).
Conclusion
NaOH may be used to induce Crohn’s colitis as an experimental model.


Similar content being viewed by others
References
Bamias G, Nyce MR, De La Rue SA, Cominelli F. New concepts in the pathophysiology of inflammatory bowel disease. Ann Intern Med. 2005;143:895–904.
Fabia R, Willén R, Ar’Rajab A, Andersson R, Ahrén B, Bengmark S. Acetic acid-induced colitis in the rat: A reproducible experimental model for acute ulcerative colitis. Eur Surg Res. 1992;24:211–225.
Gaudio E, Taddei G, Vetuschi A, Sferra R, Frieri G, Ricciardi G, Caprilli R. Dextran sulfate sodium (DSS) colitis in rats: Clinical, structural, and ultrastructural aspects. Dig Dis Sci. 1999;44:1458–1475.
Morris GP, Beck PL, Herridge MS, Depew WT, Szewczuk MR, Wallace JL. Hapten-induced model of chronic inflammation and ulceration in the rat colon. Gastroenterology. 1989;96:795–803.
Pizarro TT, Arseneau KO, Cominelli F. Lessons from genetically engineered animal models XI. Novel mouse models to study pathogenic mechanisms of Crohn’s disease. Am J Physiol Gastrointest Liver Physiol. 2000;278:G665–G669.
Gehanno P, Guedon C. Inhibition of experimental esophageal lye strictures by penicillamine. Arch Otolaryngol. 1981;107:145–147.
Liu AJ, Richardson MA. Effects of N-acetylcysteine on experimentally induced esophageal lye injury. Ann Otol Rhinol Laryngol. 1985;94:477–482.
Wang H, Ouyang Q, Hu RW. Establishment of a trinitrobenzene sulfonic acid-induced rat colitis model. Chin J Dig Dis. 2002;3:13–17.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–275.
Cortas NK, Wakid NW. Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clin Chem. 1990;36:1440–1443.
Sato K, Kanazawa A, Ota N, Nakamura T, Fujimoto K. Dietary supplementation of catechins and alpha-tocopherol accelerates the healing of trinitrobenzene sulfonic acid-induced ulcerative colitis in rats. J Nutr Sci Vitaminol (Tokyo). 1998;44:769–778.
Li XL, Cai YQ, Qin H, Wu YJ. Therapeutic effect and mechanism of proanthocyanidins from grape seeds in rats with TNBS-induced ulcerative colitis. Can J Physiol Pharmacol. 2008;86:841–849.
Zwolinska-Wcislo M, Brzozowski T, Budak A, Kwiecien S, Sliwowski Z, Drozdowicz D, Trojanowska D, Rudnicka-Sosin L, Mach T, Konturek SJ, Pawlik WW. Effect of Candida colonization on human ulcerative colitis and the healing of inflammatory changes of the colon in the experimental model of colitis ulcerosa. J Physiol Pharmacol. 2009;60:107–118.
Thomson A, Hemphill D, Jeejeebhoy KN. Oxidative stress and antioxidants in intestinal disease. Dig Dis. 1998;16:152–158.
Lih-Brody L, Powell SR, Collier KP, Reddy GM, Cerchia R, Kahn E, Weissman GS, Katz S, Floyd RA, McKinley MJ, Fisher SE, Mullin GE. Increased oxidative stress and decreased antioxidant defenses in mucosa of inflammatory bowel disease. Dig Dis Sci. 1996;41:2078–2086.
Alzoghaibi MA, Al Mofleh IA, Al-Jebreen AM. Lipid peroxides in patients with inflammatory bowel disease. Saudi J Gastroenterol. 2007;13:187–190.
Gross SS, Wolin MS. Nitric oxide: Pathophysiological mechanisms. Annu Rev Physiol. 1995;57:737–769.
Rachmilewitz D, Eliakim R, Ackerman Z, Karmeli F. Direct determination of colonic nitric oxide level—A sensitive marker of disease activity in ulcerative colitis. Am J Gastroenterol. 1998;93:409–412.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koçak, E., Köklü, S., Akbal, E. et al. NaOH-Induced Crohn’s Colitis in Rats: A Novel Experimental Model. Dig Dis Sci 56, 2833–2837 (2011). https://doi.org/10.1007/s10620-011-1697-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1697-8