Abstract
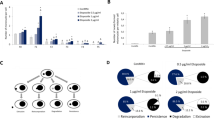
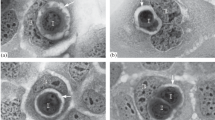
Many cytological studies have reported that the numbers of binucleated cells were elevated in various tumors. However, binucleated cells are observed in not only malignant tumors but also normal tissues. Thus, the clinical significance of binucleated cells is controversial. Here we attempted to elucidate the characteristics of binucleated HeLa cells using time-lapse microscopy. To examine the frequency, viability, proliferation, and formation mechanism of binucleated cells, we grew HeLa cells on chamber slides and tissue culture dishes in DMEM supplemented with (10, 3, 1 and 0.5 % media) and without fetal bovine serum (0 % medium). The proliferation was evaluated by the medium improvement examination (cultured for 2 more days in 10% medium after culturing in 0% medium; starvation). In the 0 % medium, 150 binucleated cells were formed by cytokinesis failure. There were significantly more binucleated cells in the 0 % medium than in the 10, 3, 1 and 0.5 % media. About twice the number of binucleated cells underwent mitosis in the improvement examinations than in the serum-free examination. We found here that starvation induced the binucleation of HeLa cells and that some binucleated cells can reproduce. These findings might be helpful for understanding binucleated cells in tumors.





Similar content being viewed by others
References
Bollmann M, Bánkfalvi A, Trosic A, Speich N, Schmittt C, Bollmann R (2005) Can we detect cervical human papillomavirus (HPV) infection by cytomorphology alone? Diagnostic value of non-classic cytological signs of HPV effect in minimally abnormal Pap tests. Cytopathology 16:13–21
Ducos GM, Morizur SC, Couton D, Bregerie O, Desdouets C (2007) Liver tetraploidization is controlled by a new process of incomplete cytokinesis. J Cell Sci 120:3633–3639
Engel FB, Schebesta M, Keating MT (2006) Anillin localization defect in cardiomyocyte binucleation. J Mol Cell Cardiol 41:601–612
Esumi H, Izuishi K, Kato K, Hashimoto K, Kurashima Y, Kishimoto A, Ogura T, Ozawa T (2002) Hypoxia and nitric oxide treatment confer tolerance to glucose starvation in a 5′-AMP-activated protein kinase-dependent manner. J Biol Chem 277:32791–32798
Fujiwara T, Bandi M, Nitta M, Ivanova EV, Bronson RT, Pellman D (2005) Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature 437:1043–1047
Ganem NJ, Storchova Z, Pellman D (2007) Tetraploidy, aneuploidy and cancer. Curr Opin Genet Dev 17:157–162
Guidotti JE, Bregerie O, Robert A, Debey P, Brechot C, Desdouets C (2003) Liver cell polyploidization: a pivotal role for binuclear hepatocytes. J Biol Chem 278:19095–19101
Hu L, Ceresa BP (2009) Characterization of the plasma membrane localization and orientation of HPV16 E5 for cell–cell fusion. Virology 383:135–143
Hu L, Plafkera K, Vorozhko V, Zuna RE, Hanigan MH, Gorbsky GJ, Plafker SM, Angeletti PC, Ceresa BP (2009) Human papillomavirus 16 E5 induces bi-nucleated cell formation by cell–cell fusion. Virology 384:125–134
Johmura Y, Shimada M, Misaki T, Naiki-Ito A, Miyoshi H, Motoyama N, Ohtani N, Hara E, Nakamura M, Morita A, Takahashi S, Nakanishi M (2014) Necessary and sufficient role for a mitosis skip in senescence induction. Mol Cell 55:73–84
Kimura N, Dota K, Araya Y, Ishidate T, Ishizaka M (2009) Scoring system for differential diagnosis of malignant mesothelioma and reactive mesothelial cells on cytology specimens. Diagn Cytopathol 37:885–890
Liu BP, Chong EY, Cheung FW, Duan JA, Che CT, Liu WK (2005) Tangutorine induces p21 expression and abnormal mitosis in human colon cancer HT-29 cells. Biochem Pharmacol 70:287–299
Minimo C, Zakowski M, Lin O (2002) Cytologic findings of malignant vascular neoplasms: a study of twenty-four cases. Diagn Cytopathol 26:349–355
Mizoguchi K, Ishikawa K (1994) Medical technology—color atlas and histopathology. Ishiyaku, Tokyo
Morin C, Bairati I, Bouchard C, Fortier M, Roy M, Moore L, Meisels A (2000) Cytologic predictors of cervical intraepithelial neoplasia in women with an ASCUS Pap smear. Acta Cytol 44:576–586
Morizur SC, Merlen G, Couton D, Desdouets C (2010) Polyploidy and liver proliferation—central role of insulin signaling. Cell Cycle 9:460–466
Nakanishi A, Han X, Saito H, Taguchi K, Ohta Y, Imajoh-Ohmi S, Miki Y (2007) Interference with BRCA2, which localizes to the centrosome during S and early M phase, leads to abnormal nuclear division. Biochem Biophys Res Commun 355:34–40
Normand G, King RW (2010) Understanding cytokinesis failure. Adv Exp Med Biol 676:27–55
Papanicolaou GN (1942) A new procedure for staining vaginal smears. Science 95:432
Pohl C, Jentsch S (2008) Final stages of cytokinesis and midbody ring formation are controlled by BRUCE. Cell 132:832–845
Polunovsky VA, Ingbar DH, Bitterman PB (1996) Cell fusion to study nuclear-cytoplasmic interactions in endothelial cell apoptosis. Am J Pathol 149:115–128
Rinaldo C, Moncada A, Gradi A, Ciuffini L, D’Eliseo D, Siepi F, Prodosmo A, Giorgi A, Pierantoni GM, Trapasso F, Guarguaglini G, Bartolazzi A, Cundari E, Schininà ME, Fusco A, Soddu S (2012) HIPK2 controls cytokinesis and prevents tetraploidization by phosphorylating histone H2B at the midbody. Mol Cell 47:87–98
Stelow EB, Bardales RH, Shami VM, Woon C, Presley A, Mallery S, Lai R, Stanley MW (2006) Cytology of pancreatic acinar cell carcinoma. Diagn Cytopathol 34:367–372
Stoll LM, Duffield AS, Johnson MW, Ali SZ (2011) Acute myeloid leukemia with myelodysplasia-related changes with erythroid differentiation involving pleural fluid: a case report and brief cytopathologic review. Diagn Cytopathol 39:451–454
Zeng ZY, Chen JM (2009) Cell–cell fusion: human multinucleated osteoclasts. Cent Eur J Biol 4:543–548
Acknowledgments
The authors would like to thank Kaori Furuya and Ayako Ogino for their technical assistance.
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nishimura, K., Watanabe, S., Hayashida, R. et al. Binucleated HeLa cells are formed by cytokinesis failure in starvation and keep the potential of proliferation. Cytotechnology 68, 1123–1130 (2016). https://doi.org/10.1007/s10616-015-9869-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-015-9869-6