Abstract
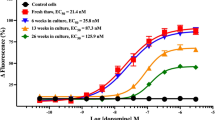
Standard tissue culture methods advise freezing cells in small aliquots (≤1 × 107 cells in 1 mL), and storing in liquid nitrogen. This is inconvenient for laboratories culturing large quantities of insect cells for recombinant baculovirus expression, owing to the length of time taken to produce large scale cultures from small aliquots of cells. Liquid nitrogen storage requires use of specialized cryovials, personal protective equipment and oxygen monitoring systems. This paper describes the long-term, large scale cryopreservation of 8 × 108 insect cells at −80 °C, using standard 50 mL conical tubes to contain a 40 mL cell suspension. Sf9, Sf21 and High 5 cells were recovered with a viability > 90 % after storage for one year under these conditions, which compared favorably with the viability of cells stored in liquid nitrogen for the same length of time. Addition of green fluorescent protein encoding baculovirus demonstrated that cells were “expression ready” immediately post thaw. Our method enables large scale cultures to be recovered rapidly from stocks cryopreserved at −80 °C, thus avoiding the inconvenience, hazards and expense associated with liquid nitrogen.




Similar content being viewed by others
References
Barford D, Takagi Y, Schultz P, Berger I (2013) Baculovirus expression: tackling the complexity challenge. Curr Opin Struct Biol 23:357–364. doi:10.1016/j.sbi.2013.03.009
Bieniossek C, Imasaki T, Takagi Y, Berger I (2012) MultiBac: expanding the research toolbox for multiprotein complexes. Trends Biochem Sci 37:49–57. doi:10.1016/j.tibs.2011.10.005
Brennan DF, Dar AC, Hertz NT, Chao WC, Burlingame AL, Shokat KM, Barford D (2011) A Raf-induced allosteric transition of KSR stimulates phosphorylation of MEK. Nature. doi:10.1038/nature09860
Chambers SP, Austen DA, Fulghum JR, Kim WM (2004) High-throughput screening for soluble recombinant expressed kinases in Escherichia coli and insect cells. Protein Expr Purif 36:40–47. doi:10.1016/j.pep.2004.03.003
Chao WC, Kulkarni K, Zhang Z, Kong EH, Barford D (2012) Structure of the mitotic checkpoint complex. Nature 484:208–213. doi:10.1038/nature10896
Davis TR, Trotter KM, Granados RR, Wood HA (1992) Baculovirus expression of alkaline phosphatase as a reporter gene for evaluation of production, glycosylation and secretion. Biotechnology 10:1148–1150
Dore AS, Kilkenny ML, Rzechorzek NJ, Pearl LH (2009) Crystal structure of the rad9-rad1-hus1 DNA damage checkpoint complex–implications for clamp loading and regulation. Mol Cell 34:735–745. doi:10.1016/j.molcel.2009.04.027
Drugmand JC, Schneider YJ, Agathos SN (2011) Insect cells as factories for biomanufacturing. Biotechnol Adv. doi:10.1016/j.biotechadv.2011.09.014
Enchev RI, Schreiber A, Beuron F, Morris EP (2010) Structural insights into the COP9 signalosome and its common architecture with the 26S proteasome lid and eIF3. Structure 18:518–527. doi:10.1016/j.str.2010.02.008
Fernandes F, Teixeira AP, Carinhas N, Carrondo MJ, Alves PM (2013) Insect cells as a production platform of complex virus-like particles. Expert Rev Vaccines 12:225–236. doi:10.1586/erv.12.153
Hashimoto Y, Zhang S, Blissard GW (2010) Ao38, a new cell line from eggs of the black witch moth, Ascalapha odorata (Lepidoptera: Noctuidae), is permissive for AcMNPV infection and produces high levels of recombinant proteins. BMC Biotechnol 10:50. doi:10.1186/1472-6750-10-50
He J, Chao WC, Zhang Z, Yang J, Cronin N, Barford D (2013) Insights into degron recognition by APC/C coactivators from the structure of an Acm1-Cdh1 complex. Mol Cell 50:649–660. doi:10.1016/j.molcel.2013.04.024
Hink WF, Thomsen DR, Davidson DJ, Meyer AL, Castellino FJ (1991) Expression of three recombinant proteins using baculovirus vectors in 23 insect cell lines. Biotechnol Prog 7:9–14. doi:10.1021/bp00007a002
Hitchman RB, Possee RD, King LA (2009) Baculovirus expression systems for recombinant protein production in insect cells. Recent Pat. Biotechnol 3:46–54
King LA, Possee RD (1992a) 5.8.1 Freezing and storage of cells in liquid nitrogen. In: The baculovirus expression system: a laboratory guide, 1st edn. Chapman & Hall, pp 102–103
King LA, Possee RD (1992b) 6.3.1 Standard plaque-assay. In: The baculovirus expression system: a laboratory guide, 1st edn. Chapman & Hall, London, pp 111–114
Kulakosky PC, Klinge CM (2003) Maximizing production of estrogen receptor beta with the baculovirus expression system. Biotechniques 34(334–338):334–345
McCullough J, Haley R, Clay M, Hubel A, Lindgren B, Moroff G (2010) Long-term storage of peripheral blood stem cells frozen and stored with a conventional liquid nitrogen technique compared with cells frozen and stored in a mechanical freezer. Transfusion 50:808–819. doi:10.1111/j.1537-2995.2009.02482.x
Murphy CI, Piwnica-Worms H, Grunwald S, Romanow WG, Francis N, Fan HY (2004) Maintenance of insect cell cultures and generation of recombinant baculoviruses current protocols in molecular biology. Frederick M, Ausubel [et al] (eds) Chapter 16:Unit 16 10 doi:10.1002/0471142727.mb1610s65
Oliver AW, Swift S, Lord CJ, Ashworth A, Pearl LH (2009) Structural basis for recruitment of BRCA2 by PALB2. EMBO Rep 10:990–996. doi:10.1038/embor.2009.126
Pasumarthy MK, Murhammer DW (1994) Clonal variation in the Spodoptera frugiperda IPLB-SF21-AE insect cell population. Biotechnol Prog 10:314–319. doi:10.1021/bp00027a012
Ratajczak MZ, Kregenow DA, Kuczynski WI, Ratajczak J, Gewirtz AM (1994) The storage of cells from different tumor lines in a mechanical freezer at −80 °C. Comparison to cryopreservation in liquid nitrogen. Mater Med Pol 26:69–72
Ratajczak MZ, Ratajczak J, Kregenow DA, Marlicz W, Machalinski B, Simon M, Luger S, Gewirtz AM (1997) An optimization study on unprogrammed cryopreservation of human CD34+ bone marrow cells and their subsequent storage in an −80 °C mechanical freezer. Ann Transplant 2:5–11
Rhiel M, Mitchell-Logean CM, Murhammer DW (1997) Comparison of Trichoplusia ni BTI-Tn-5B1-4 (high five) and Spodoptera frugiperda Sf-9 insect cell line metabolism in suspension cultures. Biotechnol Bioeng 55:909–920. doi:10.1002/(SICI)1097-0290(19970920)55:6<909:AID-BIT8>3.0.CO;2-K
Schreiber A, Stengel F, Zhang Z, Enchev RI, Kong EH, Morris EP, Robinson CV, da Fonseca PC, Barford D (2011) Structural basis for the subunit assembly of the anaphase-promoting complex. Nature 470:227–232. doi:10.1038/nature09756
Simione FP Jr (1992) Key issues relating to the genetic stability and preservation of cells and cell banks. J Parenter Sci Technol 46:226–232
Summers MD, Smith GE (1987) A manual of methods for baculovirus vectors and insect cell-culture procedures. Tex Aes Bull pp 1–56
Vaughn JL, Goodwin RH, Tompkins GJ, McCawley P (1977) The establishment of two cell lines from the insect Spodoptera frugiperda (Lepidoptera; Noctuidae). In Vitro 13:213–217
Weiss SA, Smith GC, Kalter SS, Vaughn JL (1981) Improved method for the production of insect cell-cultures in large volume. In Vitro Cell Dev B 17:495–502
Wickham TJ, Davis T, Granados RR, Shuler ML, Wood HA (1992) Screening of insect cell lines for the production of recombinant proteins and infectious virus in the baculovirus expression system. Biotechnol Prog 8:391–396. doi:10.1021/bp00017a003
Zhang Z, Chang L, Yang J, Conin N, Kulkarni K, Barford D (2013a) The four canonical TPR subunits of human APC/C form related homo-dimeric structures and stack in parallel to form a TPR suprahelix. J Mol Biol 425:4236–4248. doi:10.1016/j.jmb.2013.04.004
Zhang Z, Yang J, Kong EH, Chao WC, Morris EP, da Fonseca PC, Barford D (2013b) Recombinant expression, reconstitution and structure of human anaphase-promoting complex (APC/C). Biochem J 449:365–371. doi:10.1042/BJ20121374
Acknowledgments
The baculovirus expressing green fluorescent protein was a gift from Vangelis Christodoulou. The work was funded by Cancer Research UK programme grants to David Barford, Laurence H. Pearl and Dale Wigley. We would also like to thank David Barford and Jane Sandall for assistance with the critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
La’Verne P. Rennalls and Kirstin J. L. Wood contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vyletova, L., Rennalls, L.P., Wood, K.J.L. et al. Long-term, large scale cryopreservation of insect cells at −80 °C. Cytotechnology 68, 303–311 (2016). https://doi.org/10.1007/s10616-014-9781-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-014-9781-5