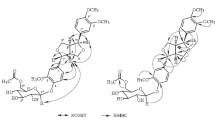
A study of the chemical composition of polar compounds from seeds of Trigonella caerulea (L.) Ser. (Fabaceae) revealed the presence of monosaccharides (2 and 3); oligosaccharides (4–7); pinitol (8) and galactopinitols (1 and 9–11), including the new compound 1; glycosides of diosgenin (12–15); and alkaloids (16–22). Mass spectrometry, chemical transformations, and NMR spectroscopy established that 1 was O-(α-Dgalactopyranosyl)-(1→6)-O-(α-D-galactopyranosyl)-(1→6)-O-(α-D-galactopyranosyl)-(1→6)-O-(α-Dgalactopyranosyl)-(1→2)-4-O-methyl-chiro-inositol (tetragalactopinitol A).

Similar content being viewed by others
References
M. Ranjbar, R. Karamian, and Z. Hajmoradi, Chem. Nat. Compd., 45, 116 (2009).
P. Hanelt, Mansfeld′s Encyclopedia of Agricultural and Horticultural Crops (Except Ornamentals), Springer, Berlin, Heidelberg, Germany, 2001, p. 864.
T. Visuvanathan, L. T. L. Than, J. Stanslas, S. Y. Chew, and S. Vellasamy, Plants, 11, 1450 (2022).
C. K. Atal and S. P. Sood, J. Pharm. Pharmacol., 16, 627 (1964).
N. G. Bogacheva, M. M. Gorokhova, V. N. Kudryavtseva, V. P. Kiselev, and L. M. Kogan, Chem. Nat. Compd., 10, 1063 (1976).
P. Brenac and Y. Sauvaire, Biochem. Syst. Ecol., 24, 157 (1996).
M. Dinu, R. Ancuceanu, D.-C. Ilie, C. Munteanu, and C.A. Aram, Farmacia, 61, 13 (2013).
M. A. Farag, D. M. Rasheed, M. Kropf, and A. G. Heiss, Anal. Bioanal. Chem., 408, 8065 (2016).
T. F. Schweizer, I. Horman, and P. Wursch, J. Sci. Food Agric., 29, 148 (1978).
P. Szczecinski, A. Gryff-Keller, M. Horbowicz, and L.B. Lahuta, J. Agric. Food Chem., 48, 2717 (2000).
Y. Tan, X. Xiao, J. Yao, F. Han, H. Lou, H. Luo, and W. Pan, Chem. Res. Chin. Univ., 33, 80 (2016).
S. W. Kim, K. C. Chung, K. H. Son, and S. S. Kang, Korean J. Pharmacogn., 20, 76 (1989).
C.-Y. Li, C.-H. Lin, and T.-S. Wu, Chem. Pharm. Bull., 53, 347 (2005).
L. Mester, L. Szabados, M. Mester, and N. Yadav, Planta Med., 35, 339 (1979).
L. Delso, C. Lafuente, J. Munoz-Embid, and M. Artal, J. Mol. Liq., 290, 111236 (2019).
S. Bas, R. Kusy, M. Pasternak-Suder, C. Nicolas, J. Mlynarski, and O. R. Martin, Org. Biomol. Chem., 16, 1118 (2018).
F. Chastellain and P. Hirsbrunner, Z. Anal. Chem., 278, 207 (1976).
J. S. Kim, Y. H. Kim, Y. W. Seo, and S. Park, Biotechnol. Bioprocess Eng., 12, 308 (2007).
I. Lee, J. Vo, Q. Gao, P. Purohit, V. Zarraga, S. Babajanian, P. Chang, and G. Swanson, J. AOAC Int., 103, 1140 (2020).
D. N. Olennikov, Metabolites, 13, 3 (2023).
D. N. Olennikov and T. G. Gornostai, Polymers, 15, 1257 (2023).
D. N. Olennikov, Separations, 10, 255 (2023).
D. N. Olennikov, V. V. Chemposov, and N. K. Chirikova, Foods, 11, 2801 (2022).
I. Ciukanu and F. Kerek, Carbohydr. Res., 131, 209 (1984).
B. Lindberg, Methods Enzymol., 28, 178 (1972).
I. Black, C. Heiss, and P. Azadi, Anal. Chem., 91, 13787 (2019).
Acknowledgment
The research was supported by the Ministry of Science and Higher Education of the Russian Federation (Project No. 121030100227-7).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2023, pp. 878–881.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Olennikov, D.N. New Galactopinitol from Seeds of Trigonella caerulea. Chem Nat Compd 59, 1038–1041 (2023). https://doi.org/10.1007/s10600-023-04191-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-04191-5