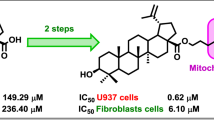
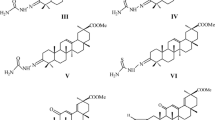
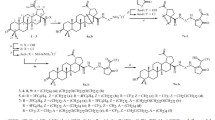
A new lupane triterpenoid containing a 1α,2β,3β-triacetoxy-functionalized ring A was synthesized from betulinic acid. Its conjugate with the membrane-penetrating cationic compound F16 was prepared. The conjugate exhibited significantly more pronounced antitumor activity than betulinic acid (betulinic acid IC50 34.5 μM, conjugate IC50 6.3 μM) in studies of the cytotoxic activity against H1299 and A549 lung adenocarcinoma cells but did not show selectivity between tumor cells and noncancer murine embryonic fibroblasts (MEFs).
Similar content being viewed by others
References
A. Hordyjewska, A. Ostapiuk, A. Horecka, and J. Kurzepa, Phytochem. Rev., 18, 929 (2019).
X. Zhang, J. Hu, and Y. Chen, Mol. Med. Rep., 14, 4489 (2016).
S. Amiria, S. Dastghaib, M. Ahmadic, P. Mehrbod, F. Khadem, H. Behrouj, M.-R. Aghanoori, F. Machaj, M. Ghamsari, J. Rosik, A. Hudecki, A. Afkhami, M. Hashemi, M. J. Los, P. Mokarram, T. Madrakian, and S. Ghavami, Biotechnol. Adv., 38, 107409 (2020).
S. C. Jonnalagadda, M. A. Corsello, and C. E. Sleet, Anti-Cancer Agents Med. Chem., 13, 1477 (2013).
D.-M. Zhang, H.-G. Xu, L. Wang, Y.-J. Li, P.-H. Sun, X.-M. Wu, G.-J. Wang, W.-M. Chen, and W.-C. Ye, Med. Res. Rev., 35, 1127 (2015).
W. Jiang, X. Li, S. Dong, and W. Zhou, Biomed. Pharmacother., 142, 111990 (2021).
V. Zuco, R. Supino, S. C. Righetti, L. Cleris, E. Marchesi, C. Gambacorti-Passerini, and F. Formelli, Cancer Lett., 175, 17 (2002).
A. Yu. Spivak, D. A. Nedopekina, R. R. Gubaidullin, E. V. Davletshin, A. A. Tukhbatullin, V. A. D′yakonov, M. M. Yunusbaeva, L. U. Dzhemileva, and U. M. Dzhemilev, Med. Chem. Res., 30, 940 (2021).
K. N. Belosludtsev, A. I. Ilzorkina, N. V. Belosludtseva, V. A. Sharapov, N. V. Penkov, D. A. Serov, M. N. Karagyaur, D. A. Nedopekina, E. V. Davletshin, M. E. Solovieva, A. Yu. Spivak, U. Sh. Kuzmina, Yu. V. Vakhitova, V. S. Akatov, and M. V. Dubinin, Biomedicines, 10, 2903 (2022).
L. Huang, H. Luo, Q. Li, D. Wang, J. Zhang, X. Hao, and X. Yang, Eur. J. Med. Chem., 95, 64 (2015).
Y. Lei, X. Xie, X. Cao, J. Chen, G. Chen, Y. Chen, G. Li, J. Qin, F. Peng, and C. Peng, Drug Des., Dev. Ther., 15, 3863 (2021).
B. Siewert, E. Pianowski, A. Obernauer, and R. Csuk, Bioorg. Med. Chem., 22, 594 (2014).
A. T. Nelson, A. M. Camelio, K. R. Claussen, J. Cho, L. Tremmel, J. DiGiovanni, and D. Siegel, Bioorg. Med. Chem. Lett., 25, 4342 (2015).
A. Yu. Spivak, D. A. Nedopekina, E. R. Shakurova, R. R. Khalitova, R. R. Gubaidullin, V. N. Odinokov, U. M. Dzhemilev, Yu. P. Bel′skii, N. V. Bel′skaya, S. A. Stankevich, E. V. Korotkaya, and V. A. Khazanov, Russ. Chem. Bull., 62, 188 (2013).
X. Liu, X. Zang, X. Yin, W. Yang, J. Huang, J. Huang, C. Yu, C. Ke, and Y. Hong, Bioorg. Chem., 97, 103694 (2020).
L. R. Huang, X. J. Hao, Q. J. Li, D. P. Wang, J. X. Zhang, H. Luo, and X. S. Yang, J. Nat. Prod., 79, 721 (2016).
D. S. H. L. Kim, Z. Chen, V. T. Nguyen, J. M. Pezzuto, S. Qui, and Z.-Z. Lu, Synth. Commun., 27, 1607 (1997).
Acknowledgment
The research was supported by Russian Science Foundation Grant No. 23-23-00098, https://rscf.ru/project/23-23-00098/. Structural studies of synthesized compounds were performed at the Agidel Center for Common Use, UFRC, RAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2023, pp. 730–735.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Spivak, A.Y., Davletshin, E.V., Nedopekina, D.A. et al. Synthesis and Cytotoxic Activity of the Conjugate of an Oxygenated Betulinic Acid Analog with the Delocalized Lipophilic Cationic Compound F16. Chem Nat Compd 59, 865–871 (2023). https://doi.org/10.1007/s10600-023-04137-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-04137-x