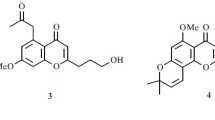
Two new chromones, 2-(2-(furan-2-yl)-2-oxoethyl)-6-methoxy-8-(2-oxopropyl)-chromone (1) and 2-(2-(furan-2-yl)-2-oxoethyl)-5-methoxy-8-(2-oxopropyl)-chromone (2), along with four known ones (3–6) were isolated from the stem bark of Cassia auriculata. The new compounds were elucidated by means of spectroscopic methods, whereas the known compounds were identified by comparison with the data in the literature. Compounds 1 and 2 were tested for their anti-tobacco mosaic virus (anti-TMV) activities. The results revealed that 1 and 2 exhibited potential anti-TMV activities with inhibition rates of 28.2 and 30.5% respectively, and these rates are close to that of the positive control. Compounds 1 and 2 were also tested for their anti-rotavirus activities, and compounds 1 and 2 showed potential activity with therapeutic index (TI) values of 14.8 and 15.9 respectively. These values are lower than that of the positive control.

Similar content being viewed by others
References
A. A. H. Abdel-Rahman, A. K. Shaban, I. F. Nassar, D. S. El-Kady, N. S. Ismail, S. F. Mahmoud, H. M. Awad, and W. A. El-Sayed, Molecules, 26 (13), 3923 (2021).
E. W. Matthew, A. S. Scott, and R. S. Brent, Curr. Opin. Chem. Biol., 14, 347 (2010).
Y. Amen, M. Elsbaey, A. Othman, M. Sallam, and K. Shimizu, Molecules, 26 (24), 7646 (2021).
N. A. ul Mohsin, M. Irfan, S. ul Hassan, and U. Saleem, Pharm. Chem. J., 54, 241 (2020).
A. T. Benny, S. D. Arikkatt, C. G. Vazhappilly, S. Kannadasan, R. Thomas, M. S. Leelabaiamma, E. K. Radhakrishnan, and P. Shanmugam, Mini-Rev. Med. Chem., 22 (7), 1030 (2022).
D. L. Wu, Flora of China, Vol. 39, Chinese Science Press, Beijing, 1988, p. 123.
J. Cai, J. Q. He, X. M. Wang, X. Shu, and L. Wu, Chin. J. Agric. Sci. Bull., 27, 142 (2011).
B. Kumar, V. Bajpai, V. Gond, S. Pal, and N. Chattopadhyay, Phytochemistry of Plants of Genus Cassia, CRC Press, 2021.
Y. D. Duan, Y. Y. Jiang, F. X. Guo, L. X. Chen, L. L. Xu, W. Zhang, and B. Liu, Fitoterapia, 135, 114 (2019).
M. Zhou, K. Zhou, X. M. Gao, Z. Y. Jiang, J. J. Lv, Z. H. Liu, G. Y. Yang, M. M. Miao, C. T. Che, and Q. F. Hu, Org. Lett., 17 (11), 2638 (2015).
P. S. Yang, J. M. Dai, X. J. Gu, W. Xiong, D. Q. Huang, S. Y. Qiu, J. N. Zheng, Y. Li, F. X. Yang, and M. Zhou, Molecules, 27, 3129 (2022).
L. M. Liao, Y. Q. Sun, J. Li, W. S. Kong, X. Liu, Y. Xu, H. T. Huang, W. L. Zeng, Q. L. Mi, G. Y. Yang, Q. F. Hu, and Y. K. Li, Chem. Nat. Compd., 56, 58 (2020).
J. R. Jiang, J. D. Zhang, G. Y. Yin, J. Q. Shi, B. B. Cai, W. W. Yang, L. L. Deng, L. Xu, T. Zhou, Q. F. Hu, M. Zhou, and W. S. Kong, Chem. Nat. Compd., 58, 414 (2022).
Q. L. Mi, W. S. Kong, Y. K. Li, X. Liu, W. L. Zeng, H. Y. Xiang, M. Dongiao, G. Y. Yang, Q. F. Hu, W. G. Wang, Q. Gao, and M. Zhou, Chem. Nat. Compd., 57, 432 (2021).
Z. Zhang, S. W. Ni, X. Xu, W. Z. Huang, S. S. Wang, H. Zhu, and X. M. Gao, China J. Chin. Mater. Med., 46 (15), 3873 (2021).
P. S. Vijayaraj, K. Muthukumar, J. Sabarirajan, and V. Nachiappan, Ind. J. Biochem. Biophys., 48, 54 (2011).
G. S. Seethapathy, D. Ganesh, J. U. S. Kumar, U. Senthilkumar, S. G. Newmaster, S. Ragupathy, R. U. Shaanker, and G. Ravikanth, Int. J. Legal. Med., 129, 693 (2015).
U. Namdaung, N. Aroonrerk, S. Suksamrarn, K. Danwisetkanjana, J. Saenboonrueng, W. Arjchomphu, and A. Suksamrarn, Chem. Pharm. Bull., 54 (10), 1433 (2006).
Q. F. Hu, B. Zhou, X. M. Gao, L. Y. Yang, L. D. Shu, Y. Q. Shen, G. P. Li, C. T. Che, and G. Y. Yang, J. Nat. Prod., 75, 1909 (2012).
G. Speranza, G. Fontana, S. Zanzola, and A. di Meo, J. Nat. Prod., 60, 692 (1997).
T. Zhou, F. X. Yang, B. B. Cai, F. Wu, Y. N. Zhu, L. Liu, C. B. Liu, J. Ling, W. S. Kong, G. Y. Yang, Q. F. Hu, and X. Liu, Chem. Nat. Compd., 57, 1019 (2021).
C. M. Johna, P. Sandrasaigaran, C. K. Tong, A. Adam, and R. Ramasamy, Cell. Immunol., 271, 474 (2011).
K. B. Thorat and V. V. Nimbalkar, World. J. Pharm. Res., 10 (4), 448 (2021).
W. G. Wang, J. Wang, Y. Xu, H. T. Huang, W. L. Zeng, J. Li, W. S. Kong, X. Liu, G. Qian, G. Y. Yang, Q. F. Hu, and Y. K. Li, Chem. Nat. Compd., 55, 1018 (2019).
G. H. Kong, Z. Y. Xia, F. Wu, Y. N. Zhu, J. Li, W. S. Kong, M. Zhou, G. Y. Yang, Q. F. Hu, and Y. P. Wu, Heterocycles, 100, 2083 (2020).
G. Y. Yang, J. M. Dai, Q. L. Mi, Z. J. Li, Xue-Mei Li, J. D. Zhang, J. Wang, Y. K. Li, W. G. Wang, M. Zhou, and Q. F. Hu, Phytochemistry, 198, 113137 (2022).
M. Zhou, M. M. Miao, G. Du, S. Z. Shang, W. Zhao, Z. H. Liu, G. Y. Yang, C. T. Che, Q. F. Hu, and X. M. Gao, Org. Lett., 16, 5016 (2014).
G. Y. Yang, J. M. Dai, Z. J. Li, J. Wang, F. X. Yang, X. Liu, J, Li, Q. Gao, X. M. Li, Y. K. Li, W. G. Wang, M. Zhou, and Q. F. Hu, Arch. Pharm. Res., 45, 572 (2022).
Acknowledgment
This project was supported financially by the Foundation of Yunnan Basic Research Program (No. 2019FD119), the National Natural Science Foundation of China (No. 32260111), the Yunnan Applied Basic Research Projects for Excellent Young Scholars (No. 202001AW079992), and the Yunnan Innovative Research Team (2019HC020).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 3, May–June, 2023, pp. 396–399.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, XS., Zheng, JN., Shen, SY. et al. Two New Chromones from Cassia auriculata and Their Antiviral Activity. Chem Nat Compd 59, 467–471 (2023). https://doi.org/10.1007/s10600-023-04025-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-04025-4