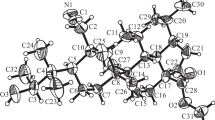
A 1-cyano-2,3-secolupane derivative with an ethylketone fragment was synthesized from 2-hydroxyoximodihydrobetulonic acid methyl ester via a Grignard reaction followed by a Beckmann rearrangement and was used further to prepare α-bromo-substituted diastereomers. Nitrile-anionic cyclization of the ethylketone and α-bromo isomers produced cyclic derivatives with an alkenenitrile and an α,β-unsaturated ketone in ring A, respectively. The obtained compounds did not exhibit cytotoxic activity according to biological screening data.

Similar content being viewed by others
References
C. Genet, A. Strehle, C. Schmidt, G. Boudjelal, A. Lobstein, K. Schoonjans, M. Souchet, J. Auwerx, R. Saladin, and A. Wagner, J. Med. Chem., 53, 178 (2010).
C. Genet, C. Schmidt, A. Strehle, K. Schoonjans, J. Auwerx, R. Saladin, and A. Wagner, ChemMedChem, 5, 1983 (2010).
R. Csuk, C. Nitsche, R. Sczepek, S. Schwarz, and B. Siewert, Arch. Pharm. (Weinheim, Ger.), 346, 232 (2013).
A. I. Govdi, N. V. Sokolova, I. V. Sorokina, D. S. Baev, T. G. Tolstikova, V. I. Mamatyuk, D. S. Fadeev, S. F. Vasilevsky, and V. G. Nenajdenko, MedChemComm, 6, 230 (2015).
G. Fontana, M. Bruno, M. Notarbartolo, M. Labbozzetta, P. Poma, A. Spinella, and S. Rosselli, Bioorg. Chem., 90, 103054 (2019).
A. V. Pereslavtseva, I. A. Tolmacheva, P. A. Slepukhin, O. S. El′ tsov, I. I. Kucherov, V. F. Eremin, and V. V. Grishko, Chem. Nat. Compd., 49, 1059 (2014).
A. V. Konysheva, V. O. Nebogatikov, I. A. Tolmacheva, M. V. Dmitriev, and V. V. Grishko, Eur. J. Med. Chem., 140, 74 (2017).
A. V. Konysheva, I. A. Tolmacheva, D. V. Eroshenko, and V. V. Grishko, Chem. Nat. Compd., 53, 497 (2017).
A. V. Konysheva, A. E. Zhukova, M. V. Dmitriev, and V. V. Grishko, Chem. Nat. Compd., 54, 1094 (2018).
A. V. Konysheva, D. V. Eroshenko, and V. V. Grishko, Nat. Prod. Commun., 14, 1 (2019).
G. F. Krainova, O. N. Gagarskikh, and V. V. Grishko, Chem. Nat. Compd., 58, 693 (2022).
F. F. Fleming, L. A. Funk, R. Altundas, and V. Sharief, J. Org. Chem., 67, 9414 (2002).
V. V. Grishko, N. V. Galaiko, I. A. Tolmacheva, I. I. Kucherov, V. F. Eremin, E. I. Boreko, O. V. Savinova, and P. A. Slepukhin, Eur. J. Med. Chem., 83, 601 (2014).
CrysAlisPro, Agilent Technologies, Version 1.171.37.33 (release 27-03-2014 CrysAlis171.NET).
G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 71, 3 (2015).
G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 71, 3 (2015).
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. Puschmann, J. Appl. Crystallogr., 42, 339 (2009).
B. Keil, Laboratoriumstechnik der organische Chemie, Akademie-Verlag, Berlin (1961) [Russian translation, Mir, Moscow, 1966, 591 pp].
T. Mosmann, J. Immunol. Methods, 65, 55 (1983).
Acknowledgment
The work was financially supported by a grant from the Russian Science Foundation No. 21-13-00161. We thank the Center for Collective Use, PFRC, UrB, RAS, Research on Materials and Compounds, for facilitating the spectral and analytical studies and Candidate M. V. Dmitriev, Department of Organic Chemistry, Perm State Natl. Res. Univ., for performing the XSA of 4.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ttranslated from Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2023, pp. 83–86.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Galaiko, N.V., Beloglazova, Y.A. & Grishko, V.V. Synthesis and Intramolecular Cyclization of 2,3-Seco-Lupane Triterpenoids with an Ethylketone Fragment. Chem Nat Compd 59, 94–98 (2023). https://doi.org/10.1007/s10600-023-03925-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-03925-9