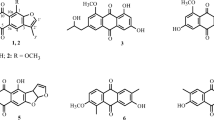
Two new indole alkaloids, 6-(4-methoxyphenoxy)-4-methoxy-2-methyl-1H-indole (1) and 6-(3,5-dimethoxyphenoxy)-4-methoxy-2-methyl-1H-indole (2), along with four known ones (3–6), were isolated from the fermentation products of the cigar tobacco-derived endophytic fungus Aspergillus oryzae. Their structures were elucidated by spectroscopic methods, including extensive 1D and 2D NMR techniques. Compounds 1 and 2 were evaluated for their antibacterial and antioxidant activities. The results showed that compounds 1 and 2 exhibited good anti-methicillin-resistant Staphylococcus aureus (anti-MRSA) activity with IZD of 22.4 ± 2.4 and 24.6 ± 2.2 mm, respectively.

Similar content being viewed by others
References
S. Gupta, P. Chaturvedi, M. G. Kulkarni, and J. Van Staden, Biotechnol. Adv., 39, 107462 (2019).
I. T. Malik and H. Brotz-Oesterhelt, Nat. Prod. Rep., 34, 815 (2017).
C. Schinke, T. Martins, S. C. N. Queiroz, I. S. Melo, and F. G. R. Reyes, J. Nat. Prod., 80, 121 (2017).
T. T. Liu, L. P. Wang, L. Zhang, H. Y. Jiang, Y. N. Zhang, and L. G. Mao, Nat. Prod. Res., 34, 1430 (2020).
F. Bongomin, C. R. Batac, M. D. Richardson, and D. W. Denning, Mycopathologia, 183, 485 (2018).
J. M. Restrepo-Florez, A. Bassi, and M. R. Thompson, Int. Biodeterior. Biodegrad., 88, 83 (2014).
S. Skanda and B. S. Vijayakumar, Curr. Microbiol., 78, 1317 (2021).
M. Y. Deng, X. Chen, Z. Y. Shi, and S. S. Xie, Fitoterapia, 151, 104882 (2021).
G. M. Daba, W. A. Elkhateeb, and F. A. Mostafa, Bioresour. Bioprocess., 8, 52 (2021).
M. Zhou, K. Zhou, P. He, K. M. Wang, R. Z. Zhu, Y. D. Wang, W. Dong, G. P. Li, H. Y. Yang, Y. Q. Ye, G. Du, X. M. Li, and Q. F. Hu, Planta. Med., 82, 414 (2016).
R. Orfali, S. Perveen, M. F. Khan, A. F. Ahmed, M. A. Wadaan, A. M. Al-Taweel, A. S. Alqahtani, F. A. Nasr, S. Tabassum, P. Luciano, G. Chianese, J. H. Sheu, and O. Taglialatela-Scafati, Mar. Drugs., 19, 333 (2021).
R. Ren, C. J. Chen, S. S. Hu, H. M. Ge, W. Y. Zhu, R. X. Tan, and R. H. Jiao, Chem. Biodiv., 12, 371 (2015).
W. G. Wang, L. Q. Du, S. L. Sheng, A. Li, Y. P. Li, G. G. Cheng, G. P. Li, G. L. Sun, Q. F. Hu, and Y. Matsuda, Org. Chem. Front., 6, 571 (2019).
C. W. Liu, A. Minami, T. Ozaki, T, J. Wu, H. Kawagishi, J. I. Maruyama, and H. Oikawa, J. Am. Chem. Soc., 141, 15519 (2019).
L. Liu, L. Bao, L. Wang, K. Ma, J. J. Han, Y. L. Yang, R. X. Liu, J. W. Ren, W. B. Yin, W. Z. Wang, and H. W. Liu, J. Org. Chem., 83, 812 (2018).
X. Hu, Q. W. Xia, Y. Y. Zhao, Q. H. Zheng, Q. Y. Liu, L. Chen, and Q. Q. Zhang, Chem. Pharm. Bull., 46, 942 (2015).
M. Shaaban, M. M. El-Metwally, and H. Nasr, Nat. Prod. Res., 28, 86 (2014).
D. Nikolic, T. Godecke, S. N. Chen, J. White, D. C. Lankin, G. F. Pauli, and R. B. van Breemen, Fitoterapia, 83, 441 (2012).
P. L. Wu, Y. L. Hsu, and C. W. Jao, J. Nat. Prod., 69, 1467 (2006).
H. Chen, J. Bai, Z. F. Fang, S. S. Yu, S. G. Ma, S. Xu, Y. Li, J. Qu, J. H. Ren, L. Li, Y. K. Si, and X. G. Chen, J. Nat. Prod., 74, 2438 (2011).
F. Tabassum, C. M. Hasan, M. M. Masud, S. Jamshidi, K. M. Rahman, and M. Ahsan, Phytochemistry, 186, 112744 (2021).
M. P. Epplin, A. Mohan, L. D. Harris, Z. J. Zhu, K. L. Strong, J. Bacsa, P. Le, D. S. Menaldino, S. F. Traynelis, and D. C. Liotta, J. Med. Chem., 63, 7569 (2020).
D. R. Beukes, M. T. Davies-Coleman, M. Kelly-Borges, M. K. Harper, and D. J. Faulkner, J. Nat. Prod., 61, 699 (1998).
H. Tanak, A. Agar, and M. Yavuz, J. Mol. Model., 16, 577 (2010).
F. D. Kong, S. L. Zhang, S. Q. Zhou, Q. Y. Ma, Q. Y. Xie, J. P. Chen, J. H. Li, L. M. Zhou, J. Z. Yuan, Z. Hu, H. F. Dai, X. L. Huang, and Y. X. Zhao, J. Nat. Prod., 82, 3456 (2019).
K. Xu, X. L. Yuan, C. Li, and X. D. Li, Mar. Drugs., 18, 54 (2020).
M. L. Wang, R. Chen, F. J. Sun, P. R. Cao, X. R. Chen, and M. H. Yang, Tetrahedron Lett., 68, 152914 (2021).
Clinical and Laboratory Standards Institute, Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard, Vol. 32, Clinical and Laboratory Standards Institute, Wayne, Pa, USA, 9th Edition, 2012.
Acknowledgment
This project was supported financially by the Foundation of Chemical and Biological Innovation Studio of Yunnan Industrial Co., Ltd., the Foundation of Yunnan Tobacco Industry Co., Ltd. (No. 2020JC02), the National Natural Science Foundation of China (No. 21967021), and the Foundation of Yunnan Innovative Research Team (2019HC020).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2022, pp. 920–923.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Mf., Xiao, D., Zhu, LC. et al. Indole Alkaloids from the Cigar Tobacco-Derived Endophytic Fungus Aspergillus oryzae and Their Antibacterial Activity. Chem Nat Compd 58, 1093–1097 (2022). https://doi.org/10.1007/s10600-022-03872-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-022-03872-x