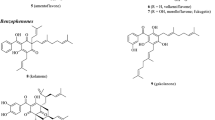
New O-glycosides of herbacetin and gossypetin (1–6) and eight known compounds were isolated from leaves of Rhodiola rosea L. [Sedum roseum (L.) Scop., Crassulaceae]. According to UV, IR, and NMR spectroscopic and mass spectrometric data, the isolated compounds were herbacetin-3-O-(3′′ -O-malonyl)-β-Dglucopyranoside-7-O-β-D-glucopyranoside-8-O-β-D-glucuronopyranoside (rhodirosin A, 1), herbacetin- 3-O-(3′′,4′′-di-O-acetyl)-β-D-glucopyranoside-8-O-β-D-glucuronopyranoside (rhodirosin B, 2), gossypetin- 8-O-(2′′-O-malonyl)-β-D-glucuronopyranoside (rhodirosin C, 3), gossypetin-8-O-(2′′-O-acetyl)-β-Dglucuronopyranoside (rhodirosin D, 4), herbacetin-8-O-(2′′-O-malonyl)-β-D-glucuronopyranoside (rhodirosin E, 5), and herbacetin-8-O-(2′′ -O-acetyl)-β-D-glucuronopyranoside (rhodirosin F, 6). Flavonoids 3 and 4 possessed the highest antiradical activity and inhibited the activity of α-amylase.
Similar content being viewed by others
References
H. Tao, X. Wu, J. Cao, Y. Peng, A. Wang, J. Pei, J. Xiao, S. Wang, and Y. Wang, Med. Res. Rev., 2019, 1 (2019).
V. A. Kurkin, G. G. Zapesochnaya, and A. N. Shchavlinskii, Chem. Nat. Compd., 20, 623 (1984).
G. G. Zapesochnaya, V. A. Kurkin, and A. N. Shchavlinskii, Chem. Nat. Compd., 21, 464 (1985).
A. Petsalo, J. Jalonen, and A. Tolonen, J. Chromatogr., 1112, 224 (2006).
D. N. Olennikov, N. K. Chirikova, A. G. Vasilieva, and I. A. Fedorov, Antioxidants, 9, 526 (2020).
M. Wolbis, Phytochemistry, 28, 2187 (1989).
D. van Diermen, M. Pierreclos, and K. Hostettmann, Planta Med., 75, PJ49 (2009); D. van Diermen, Phytochemical Investigation of Two Crassulaceae Species: Rhodiola rosea L., the New “Herbal Stress Buster”, and Sedum dasyphyllum L., These de Doctorat, University of Geneve, Geneve, 2009, 220 pp.
M. Sikorska, I. Matlawska, and R. Franski, Acta Physiol. Plant., 26, 291 (2004).
D. N. Olennikov and N. K. Chirikova, Chem. Nat. Compd., 56, 1048 (2020).
N. I. Kashchenko, D. N. Olennikov, and N. K. Chirikova, Plants, 10, 2317 (2021).
D. N. Olennikov and N. K. Chirikova, Chem. Nat. Compd., 55, 1032 (2019).
D. N. Olennikov, N. K. Chirikova, N. I. Kashchenko, T. G. Gornostai, I. Y. Selyutina, and I. N. Zilfikarov, Int. J. Mol. Sci., 18, 2579 (2017).
M. Akabane, A. Yamamoto, S. Aizawa, A. Taga, and S. Kodama, Anal. Sci., 30, 739 (2014).
D. N. Olennikov, C. S. Kirillina, and N. K. Chirikova, Antioxidants, 10, 1300 (2021).
D. N. Olennikov, V. V. Chemposov, and N. K. Chirikova, Plants, 10, 2525 (2021).
Acknowledgment
The studies were supported by the Ministry of Education and Science of the Russian Federation (Projects Nos. 121030100227-7 and FSRG-2020-0019).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2022, pp. 854–858.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Olennikov, D.N., Chirikova, N.K. New Metabolites of Rhodiola rosea I. Glycosides of Herbacetin and Gossypetin. Chem Nat Compd 58, 1014–1020 (2022). https://doi.org/10.1007/s10600-022-03857-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-022-03857-w