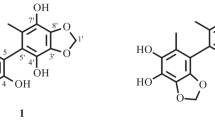
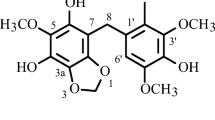
Cinnamosmophin (1), along with 12 known compounds, was isolated from the barks of Cinnamomum osmophloeum (Lauraceae). Their structures were unambiguously elucidated by NMR spectroscopic analyses.
Similar content being viewed by others
References
T. W. Hu, Y. T. Lin, and C. K. Ho, Bull. Taiwan For. Res. Inst. Eng., 78, 18 (1985).
C. Y. Chen, C. H. Chen, Y. C. Lo, B. N. Wu, H. M. Wang, W. L. Lo, C. M. Yen, and R. J. Lin, J. Nat. Prod., 71, 933 (2008).
P. L. Kuo, C. Y. Chen, T. F. Tzeng, C. C. Lin, and Y. L. Hsu, Toxicol. Appl. Pharm., 229, 215 (2008).
C. Y. Chen, Y. L. Hsu, Y. C. Tsai, and P. L. Kuo, Food Chem. Toxicol., 46, 2476 (2008).
T. Z. Liu, J. T. Cheng, S. J. Yiin, C. Y. Chen, C. H. Chen, C. H. Chen, M. J. Wu, and C. L. Chern, Food Chem. Toxicol., 46, 321 (2008).
C. Y. Chen, Y. L. Hsu, Y. Y. Chen, J. Y. Hung, M. S. Huang, and P. L. Kuo, Eur. J. Pharmacol., 574, 94 (2007).
P. L. Kuo, C. Y. Chen, and Y. L. Hsu, Cancer Res., 67, 7406 (2007).
C. L. Lin, C. L. Kao, W. J. Li, H. T. Li, and C. Y. Chen, Chem. Nat. Compd., 54, 762 (2018).
G. C. Huang, C. L. Kao, W. J. Li, C. H. Yeh, H. T. Li, and C. Y. Chen, Chem. Nat. Compd., 54, 869 (2018).
C. Y. Chen, C. L. Lin, C. L. Kao, C. T. Chen, and H. T. Li, Chem. Nat. Compd., 55, 922 (2019).
C. T. Chen, C. L. Kao, W. J. Li, H. C. Yeh, H. T. Li, and C. Y. Chen, Chem. Nat. Compd., 55, 1185 (2019).
C. Y. Chen, C. T. Chen, S. L. Liu, C. L. Kao, W. J. Li, H. C. Yeh, H. T. Li, and H. W. Chang, Chem. Nat. Compd., 56, 611 (2020).
C. Y. Chen, C. M. Liu, H. M. Wu, H. C. Yeh, W. J. Li, H. T. Li, and H. W. Chang, Chem. Nat. Compd., 56, 621 (2020).
C. Y. Chen, F. R. Chang, and Y. C. Wu, J. Chin. Chem. Soc., 46, 77 (1999).
A. K. Sinha, S. C. Verma, and U. K. Sharma, J. Sep. Sci., 30, 15 (2007).
X. Tang, S. Chen, and L. Wang, Nat. Prod. Res., 25, 320 (2011).
E. Games, M. Guerreiro, F. R. Santana, N. M. Pinheiro, E. A. de Oliveira, F. D. Lopes, C. R. Olivo, I. F. Tiberio, M. A. Martins, J. H. G. Lago, and C. M. Prado, Molecules, 21, 1390 (2016).
H. R. Gazi, M. S. Rahman, M. H. Sohrab, B. Begum, and M. A. Rashid, Turk. J. Pharm. Sci., 9, 335 (2012).
J. R. Steter, J. O. S. Ponto′lio, M. I. C. F. Costa, and J. R. Romero, Polyhedron, 26, 996 (2007).
S. Mohottalage, R. Tabacchi, and P. M. Guerin, Flavour Fragr. J., 22, 130 (2007).
R. O. S. Fontenelle, S. M. Morais, E. H. S. Brito, R. S. N. Brilhante, R. A. Cordeiro, Y. C. Lima, N. V. G. P. S. Brasil, A. J. Monteiro, J. J. C. Sidrim, and M. F. G. Rocha, Molecules, 16, 6422 (2011).
S. Sitrallah and J. Merza, Chem. Mat. Res., 8, 13 (2016).
Acknowledgment
This investigation was supported by a grant from Yuan′s General Hospital and Fooyin University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2022, pp. 671–673.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, C.Y., Kao, C.L., Yeh, H.C. et al. A Novel Biaryl Ether from Cinnamomum osmophloeum. Chem Nat Compd 58, 793–795 (2022). https://doi.org/10.1007/s10600-022-03799-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-022-03799-3