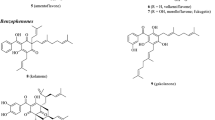
In the current work, a novel triterpene saponin, named ursolic acid 3-O-[α-L-rhamnopyranosyl(1"→6')]-β-D-glucopyranosyl-28-O-β-D-glucuronopyranoside (1), together with two known flavonol glycosides, kaemferol 3-O-α-L-rhamnopyranoside (2) and quercetin 3-O-β-D-glucopyranoside (3), was isolated from the stem outer barks of Pinus pumila by repeated column chromatography and vacuum liquid chromatography. The chemical structures of the secondary metabolites 1–3 were characterized by their spectroscopic data and other physico-chemical evidence, as well as by comparison with the literature. The secondary metabolites 2 and 3 were found in this species for the first time.

Similar content being viewed by others
References
Y. Amagai and G. Kudo, Alpine Bot., 129, 43 (2019).
Z. J. Li, L. Wu, C. L. Si, J. H. Wang, X. N. Yan, H. Y. Yu, R. Z. Cong, S. Y. Wang, and R. Wei, Chem. Nat. Compd., 55, 1187 (2019).
U. Zeb, R. N. Wang, P. B. Dong, N. Wang, T. T. Zhang, C. Z. Lin, and X. J. Wang, Mitochondrial DNA B, 4, 290 (2019).
N. H. Sa, N. T. Tam, N. T. H. Anh, T. D. Quan, D. D. Thien, D. T. Phong, and T. T. Thuy, Nat. Prod. Res., 32, 341 (2018).
Z. J. Li, F. Fan, C. L. Si, X. N. Yan, H. Y. Yu, J. H. Wang, and L. Wu, Chem. Nat. Compd., 56, 1128 (2020).
G. H. Chen, Y. C. Li, N. H. Lin, P. C. Kuo, and J. T. Tzen, Molecules, 23, 86 (2017).
M. Karapandzova, G. Stefkov, I. Cvetkovikj, J. P. Stanoeva, M. Stefova, and S. Kulevanova, Nat. Prod. Commun., 10, 987 (2015).
M. K. Langat, A. Helfenstein, C. Horner, P. Tammela, H. Hokkanen, D. Izotov, and D. A. Mulholland, Chem. Biodiv., 15, e1800056 (2018).
V. A. Hang, Y. V. Gatilov, Z. V. Dubovenko, and V. A. Pentegova, Chem. Nat. Compd., 16, 361 (1980).
A. V. Shpatov, S. A. Popov, O. I. Salnikova, E. N. Shmidt, S. W. Kang, S. M. Kim, and B. H. Um, Chem. Biodiv., 10, 198 (2013).
P. J. Houghton and A. Raman, Layer chromatography, Laboratory Handbook for the Fractionation of Natural Extracts, Springer, Berlin, 1998.
R. Wang, K. Wang, C. L. Si, Y. Y. Luo, W. Liu, X. X. Zhang, Y. Y. Tian, and J. H. Wang, Chem. Nat. Compd., 54, 717 (2018).
C. L. Si, Y. Gao, L. Wu, R. Liu, G. H. Wang, L. Dai, X. H. Li, and Y. M. Hong, Holzforschung, 71, 697 (2017).
W. Seebacher, N. Simic, R. Weis, R. Saf, and O. Kunert, Magn. Reson. Chem., 41, 636 (2003).
L. Wu, G. C. Wang, T. Shen, Q. Qiang, Q. Xue, M. Chen, M. Chen, J. M. Zhang, Y. Y. Luo, Y. M. Hong, C. L. Si, and W. C. Hu, Chem. Nat. Compd., 54, 210 (2018).
T. Miyase, K. I. Shiokawa, D. M. Zhang, and A. Ueno, Phytochemistry, 41, 1411 (1996).
C. L. Si, L. Wu, and Z. Y. Zhu, Biochem. Syst. Ecol., 37, 221 (2009).
Z. J. Li, J. X. Yang, H. Wang, R. Xu, Z. T. Rao, C. L. Si, J. Zhang, L. Sun, X. Y. Zhang, S. J. Han, Z. Sun, L. Wu, D. Liu, Y. Liu, and J. H. Wang, Chem. Nat. Compd., 55, 144 (2019).
N. Riaz, H. M. Rafiq, M. Saleem, B. Jabeen, M. Ashraf, I. Ahmed, T. Tahir, and R. B. Tareen, Chem. Nat. Compd., 55, 493 (2019).
C. L. Si, S. L. Chen, Z. J. Li, D. Liu, S. X. Nie, Y. Liu, L. Sun, X. Y. Zhang, and S. J. Han, Chem. Nat. Compd., 55, 252 (2019).
N. Kavtaradze, M. Alaniya, M. Masullo, A. Cerulli, and S. Piacente, Chem. Nat. Compd., 56, 70 (2020).
C. L. Si, X. H. Yang, Z. J. Li, J. S. Lu, X. Tao, J. Y. Zhang, W. Liu, and Y. S. Bae, Holzforschung, 72, 719 (2018).
W. C. Hu, X. F. Wang, L. Wu, T. Shen, L. L. Ji, X. H. Zhao, C. L. Si, Y. Y. Jiang, and G. C. Wang, Food Funct., 7, 1002 (2016).
C. L. Si, L. Wu, Z. Y. Zhu, J. K. Kim, D. Z. Kwon, and Y. S. Bae, Holzforschung, 63, 440 (2009).
M. Okawa, R. Akahoshi, K. Kawasaki, D. Nakano, R. Tsuchihashi, J. Kinjo, and T. Nohara, Chem. Pharm. Bull., 67, 159 (2019).
C. L. Si, G. H. Xu, X. F. Huang, Z. G. Du, L. Wu, and W. C. Hu, Chem. Nat. Compd., 52, 132 (2016).
C. N. Zhao, Z. L. Yao, D. Yang, J. Ke, Q. L. Wu, J. K. Li, and X. D. Zhou, Biomolecules, 10, 74 (2020).
C. L. Si, J. Z. Jiang, S. C. Liu, H. Y. Hu, X. D. Ren, G. J. Yu, and G. H. Xu, Holzforschung, 67, 357 (2013).
P. K. Agrawal, Carbon-13 NMR of Flavanoids, Elsevier, New York, 1989.
L. Q. Hu, K. Wang, G. B. Li, R. Y. Zhang, Y. Y. Luo, C. L. Si, and J. H. Wang, Holzforschung, 71, 785 (2017).
L. Wu, W. Xiong, J. W. Hu, J. Wu, Z. J. Li, Y. Gao, D. Liu, Y. Liu, W. Liu, M. Liang, C. L. Si, and Y. S. Bae, Chem. Nat. Compd., 55, 345 (2019).
J. B. Harborne and T. J. Mabry, The Flavonoids: Advances in Research, Chapman and Hall Ltd., London, 1982.
Acknowledgment
This work was financially supported by the Fundamental Research Funds for the Central Non-profit Research Institute of CAF (CAFYBB2016ZD009), Guangxi Key Laboratory of Clean Pulp & Papermaking and Pollution Control, College of Light Industry and Food Engineering, Guangxi University (KF201801-5), Key Technology Research and Development Program of Tianjin (19YFZCSN00950), and Tianjin Enterprise Technology Commissioner Project (19JCTPJC52800).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2021, pp. 96–99.
Rights and permissions
About this article
Cite this article
Liu, K., Liu, HY., Tao, X. et al. A New Triterpene Glycoside from Pinus pumila. Chem Nat Compd 57, 115–119 (2021). https://doi.org/10.1007/s10600-021-03294-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-021-03294-1