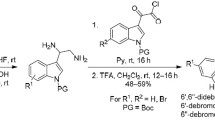
Iotrochamide B is the first cinnamoyl amino acid reported from the marine sponge Iotrochota sp. The total synthesis of the marine indole alkaloid iotrochamide B was achieved by condensation of 6-bromo-L-tryptophan (3) and (Z)-2-methoxy-3-phenylacrylic acid (6). The key step was the synthesis of 6-bromo-L-tryptophan ((S)-3) from racemic N-acetyltryptophan by optical resolution using (S)-(–)-1-phenylethylamine. This work provides an efficient method for future synthesis of iotrochamide B derivatives.
Similar content being viewed by others
References
A. J. Kochanowska-Karamyan and M. T. Hamann, Chem. Rev., 110, 4489 (2010).
S. R. Ibrahim, G. A. Mohamed, and M. A. Fouad, Nat. Prod. Res., 23, 86 (2009).
Y. J. Feng, R. A. Davis, and M. L. Sykes, Bioorg. Med. Chem. Lett., 22, 4873 (2012).
M. P. Moyer, F. John, J. F. Shiurba, and H. Rapoport, J. Org. Chem., 51, 5106 (1986).
Y. Konda-Yamada, C. Okada, and K. Yoshida, Tetrahedron, 58, 7851 (2002).
X. B. Su, Q. H. Zhang, and Y. Q. Wu, Chin. J. Org. Chem., 22, 496 (2002).
L. Shi, L. Wang, and Z. Wang, Eur. J. Med. Chem., 47, 585 (2012).
Y. Z. Liu, L. Q. Shang, and H. Fang, Bioorg. Med. Chem., 17, 7398 (2009).
Acknowledgment
This work was supported by Scientific Research Fund of Xinxiang Medical University grants [2013QN121].
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 3, May–June, 2019, pp. 428–430.
Rights and permissions
About this article
Cite this article
Wang, S.Y., Zhao, J. & Que, H.D. Synthesis of the Natural Product Iotrochamide B. Chem Nat Compd 55, 499–501 (2019). https://doi.org/10.1007/s10600-019-02724-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-019-02724-5