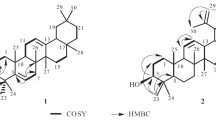
Methyl esters of 3β-O-acetoxy-18βH-olean-12-en-30-oic, 3β-hydroxy-9(11),12(13)-dien-30-oic, and 3β-O-acetoxy-18βH-olean-12-en-30-oic acids were oxidized by meta-chloroperbenzoic acid (m-CPBA) to produce methyl esters of 3β-O-acetoxy-12α,13α-epoxy-18β-oleanan-30-oic and 3β-O-acetoxy-18β-oleanan-12-on-30-oic acids in addition to 3β-hydroxy-12-oxo-18βH-olean-9(11)-en-30-oic and 3β-O-acetoxy-18β-oleanan-12-on-30-oic acids in 70–75% yields. The structures of the synthesized compounds were confirmed by PMR and 13C NMR spectra.
Similar content being viewed by others
References
X. Wang, H. Zhang, L. Chen, L. Shan, G. Fan, and X. Gao, J. Ethnopharmacol., 150, 781 (2013).
C. S. Graebin, H. Verli, and J. A. Guimaraes, J. Braz. Chem. Soc., 21, 1595 (2010).
M. Nassiri and H. Hosseinzadeh, Phytother. Res., 22, 709 (2008).
H. Liu, J. Wang, W. Zhou, Y. Wang, and L. Yang, J. Ethnopharmacol., 146, 773 (2013).
G. A. Tolstikov, L. A. Baltina, V. P. Grankina, R. M. Kondratenko, and T. G. Tolstikova, Licorice: Biodiversity, Chemistry and Use in Medicine [in Russian], Akademicheskoe Izd. Geo, Novosibirsk, 2007.
X. Su, L. Wu, M. Hu, W. Dong, M. Xu, and P. Zhang, Biomed. Pharmacother., 95, 670 (2017).
R. Czuk, Mini-Rev. Org. Chem., 11, 253 (2014).
A. Roohbakhsh, M. Iranshahy, and M. Iranshahi, Curr. Med. Chem., 23, 498 (2016).
L. R. Mikhailova, M. V. Khudobko, L. A. Baltina, Jr., L. V. Spirikhin, R. M. Kondratenko, and L. A. Baltina, Chem. Nat. Compd., 45, 393 (2009).
C. Farina and M. Pinza, Gazz. Chim. Ital., 117, 561 (1987).
R. B. Boar, L. Jokhadar, M. De Luque, and J. F. Mc Ghie, J. Chem. Soc., Perkin Trans. 1, 2104 (1977).
Y.-M. Zhu, J.-K. Shen, H.-K. Wang, L. M. Cosentino, and K.-H. Lee, Bioorg. Med. Chem. Lett., 11, 3115 (2001).
A. A. Ryabinin and N. E. Konovalova, Zh. Obshch. Khim., 32, 644 (1962).
E. B. Logashenko, O. V. Salomatina, A. V. Markov, D. V. Korchagina, N. F. Salakhutdinov, and G. A. Tolstikov, ChemBioChem, 12, 784 (2011).
L. R. Mikhailova, M. V. Khudobko, L. A. Baltina, O. S. Kukovinets, V. K. Mavrodiev, and F. Z. Galin, Chem. Nat. Compd., 43, 571 (2007).
A. S. Budaev, L. R. Mikhailova, L. V. Spirikhin, and L. A. Baltina, Chem. Nat. Compd., 52, 448 (2016).
A.Garcia-Granados, P. E. Lopez, E. Melguizo, A. Parra, and Y. Simeo, Tetrahedron, 60, 3831 (2004).
A. J. Gordon and R. A. Ford, The Chemist’s Companion, Wiley-Interscience, New York (1972).
Acknowledgment
The work was performed under State Task topic AAAA-A17-117011910025-6 and used equipment of Khimiya CUC, UfIC, UfSC, RAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2019, pp. 76–79.
Rights and permissions
About this article
Cite this article
Mikhailova, L.R., Budaev, A.S., Spirikhin, L.V. et al. Oxidation of Licorice-Root Triterpene-Acid Derivatives by m-Chloroperbenzoic Acid. Chem Nat Compd 55, 88–91 (2019). https://doi.org/10.1007/s10600-019-02619-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-019-02619-5