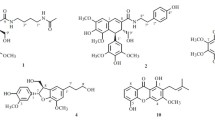
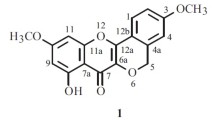
A new triterpenoid, methyl 2α-O-trans-feruloyl-3β-hydroxy-urs-12-en-28-oate (1), has been isolated from the leaves of Eriobotrya deflexa f. buisanensis, together with five known compounds, methyl ursolate (2), oleanolic acid (3), corosolic acid (4), 3-O-trans-caffeoyltormentic acid (5), and tormentic acid (6). The structure of the new compound 1 was determined through spectroscopic and MS analyses. Among the isolates, oleanolic acid (3), corosolic acid (4), and tormentic acid (6) showed potent inhibition with IC50 values of 16.3 ± 1.8, 20.4 ± 2.2, and 18.3 ± 1.4 μM, respectively, against LPS-induced NO generation. In addition, 3-O-trans-caffeoyltormentic acid (5) exhibited anti-Dengue virus activity with IC50 value of 12.4 ± 1.1 μM against Dengue virus serotype 2 strain PL046.
Similar content being viewed by others
References
H. Ohashi, Rosaceae in Flora of Taiwan, Vol. 3, 2nd Ed., Editorial Committee of the Flora of Taiwan, Taipei, Taiwan, 1993, pp. 69–157.
N. De Tommasi, F. De Simone, R. Aquino, C. Pizza, and Z. Z. Liang, J. Nat. Prod., 53, 810 (1990).
N. De Tommasi, R. Aquino, F. De Simone, and C. Pizza, J. Nat. Prod., 55, 1025 (1992).
N. De Tommasi, F. De Simone, C. Pizza, N. Mahmood, N. Orsi, and M. L. Stein, J. Nat. Prod., 55, 1067 (1992).
T. Kikuchi, H. Akazawa, K. Tabata, A. Manosroi, J. Manosroi, T. Suzuki, and T. Akihisa, Chem. Pharm. Bull., 59, 378 (2011).
T. Uto, A. Sakamoto, N. H. Tung, T. Fujiki, K. Kishihara, S. Oiso, H. Kariyazono, O. Morinaga, and Y. Shoyama, Int. J. Mol. Sci., 14, 4106 (2013).
D. O. Collins, P. L. D. Ruddock, J. C. de Grasse, W. F. Reynolds, and P. B. Reese, Phytochemistry, 59, 479 (2002).
A. A. Bimurzaev and G. K. Nikonov, Chem. Nat. Compd., 20, 505 (1984).
G. Uddin, W. Aliullah, B. S. Siddiqui, M. Alam, A. Sadat, A. Ahmad, and A. Uddin, Middle East J. Sci. Res., 8, 85 (2011).
Y. H. Shen, T. Lu, J. Tang, R. H. Liu, H. L. Li, and W. D. Zhang, Chem. Nat. Compd., 46, 305 (2010).
S. Taniguchi, Y. Imayoshi, E. Kobayashi, Y. Takamatsu, H. Ito, T. Hatano, H. Sakagami, H. Tokuda, H. Nishino, D. Sugita, S. Shimura, and T. Yoshida, Phytochemistry, 59, 315 (2002).
M. Johansson, B. Kopcke, H. Anke, and O. Sterner, J. Antibiot., 55, 104 (2002).
Y. L. Lin, C. L. Liao, L. K. Chen, C. T. Yeh, C. I. Liu, S. H. Ma, Y. Y. Huang, Y. L. Huang, C. L. Kao, and C. C. King, J. Virol., 72, 9729 (1998).
S. F. Wu, C. J. Lee, C. L. Liao, R. A. Dwek, N. Zitzmann, and Y. L. Lin, J. Virol., 76, 3596 (2002).
Acknowledgment
This research was supported by a grant from the Ministry of Science and Technology, Taiwan (MOST 106-2320-B-010-033-MY3), awarded to Prof. J.-J. Chen. This work was also supported by grants from Pingtung Christian Hospital (PS 106022), Kaohsiung Municipal United Hospital (KMUH-10612), and Zuoying Branch of Kaohsiung Armed Forces General Hospital (ZBH 106-24 and ZBH 106-25). Jih-Jung Chen, Ru-Fang Wu, Jen-Wen Hsiao, and Li-Chai Chen have contributed equally in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2019, pp. 65–68.
Rights and permissions
About this article
Cite this article
Chen, JJ., Wu, RF., Hsiao, JW. et al. A New Triterpenoid and Bioactive Constituents of Eriobotrya deflexa f. buisanensis. Chem Nat Compd 55, 74–78 (2019). https://doi.org/10.1007/s10600-019-02616-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-019-02616-8