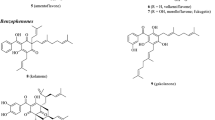
A new phenolic ester, viridone (1), has been isolated from the EtOH soluble part of Ocimum viride, along with previously reported known compounds: ursolic acid (2), oleanolic acid (3), stigmasterol (4), β-sitosterol-glucoside (5), 7-hydroxycoumarin (6), jacarandic acid (7), pedunculin (8), and crismaritin (9). Their structures were elucidated by spectroscopic techniques including two-dimensional NMR. Viridone (1) showed significant inhibitory activity against butyrylcholinesterase.

Similar content being viewed by others
References
A. Paton, R. M. Harley, and M. M. Harley, Medicinal and Aromatic Plants–Industrial Profile, Harwood Academic, Amsterdam, 1999, p. 1.
D. Tewari, H. K. Pandey, A. N. Sah, H. S. Meena, and A. Manchanda, Asian J. Plant Sci. Res., 2, 446 (2012).
S. Verma and P. Kothiyal, Int. J. Biopharm. Phytochem. Res., 1, 21 (2012).
S. Singh, M. Taneja, and D. K. Majumdar, Ind. J. Exp. Biol., 45, 403 (2007).
L. Ntezurubanza, J. J. Scheffer, and A. B. Svendsen, Planta Med., 53, 421 (1987).
A. Fleisher, J. Sci. Foot. Agric., 32, 1119 (1981).
P. Pushpangadan and S. Sobti, Cytologia, 47, 575 (1982).
S. M. Keita, C. Vincent, J. P. Schmit, and A. Belanger, Flav. Frag., 15, 339 (2000).
G. L. Ellman, D. Courtney, V. Andres, and R. M. Featherstone, Biochem. Pharmacol., 7, 88 (1961).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 4, July–August, 2018, pp. 564–566.
Rights and permissions
About this article
Cite this article
Hashmi, A., Ali, M.S., Latif, M. et al. Viridone, a New Butyrylcholinesterase Inhibitory Phenolic Ester from Leaves of Ocimum viride. Chem Nat Compd 54, 665–668 (2018). https://doi.org/10.1007/s10600-018-2441-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-018-2441-9