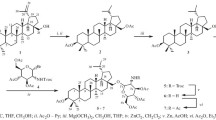
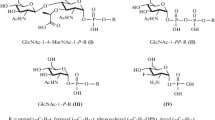
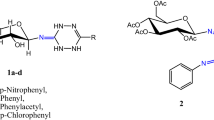
Glucuronosyl derivatives of the glycoside steviolbioside from Stevia rebaudiana were synthesized for the first time. Steviolbioside exhibited selective bacteriostatic and bactericidal activity against S. aureus ATCC 209p with minimum inhibitory concentration (MIC) of 62.5 μg/mL, which corresponded to the activity of the antibiotic chloramphenicol.
Similar content being viewed by others
References
A. V. Stachulski and X. Meng, Nat. Prod. Rep., 30, 806 (2013).
A. Wheeler, A. C. Boileau, P. C. Winkler, J. C. Compton, I. Prakash, X. Jiang, and D. A. Mandarino, Food Chem. Toxicol., 46, S54 (2008).
M. Wang, H. Qi, J. Li, Y. Xu, and H. Zhang, Food Chem. Toxicol., 86, 217 (2015).
R. R. Sharipova, I. Yu. Strobykina, V. E. Kataev, O. A. Lodochnikova, A. T. Gubaidullin, and A. A. Stomakhin, Zh. Obshch. Khim., 79, 2041 (2009).
R. R. Sharipova, I. Yu. Strobykina, G. G. Mordovskoi, R. V. Chestnova, V. F. Mironov, and V. E. Kataev, Chem. Nat. Compd., 46, 902 (2011).
R. R. Sharipova, B. F. Garifullin, O. V. Andreeva, I. Yu. Strobykina, O. B. Bazanova, and V. E. Kataev, Zh. Obshch. Khim., 85, 989 (2015).
R. R. Sharipova, B. F. Garifullin, O. V. Andreeva, I. Yu. Strobykina, O. B. Bazanova, and V. E. Kataev, Zh. Org. Khim., 51, 437 (2015).
R. R. Sharipova, B. F. Garifullin, O. V. Andreeva, I. Yu. Strobykina, and V. E. Kataev, Chem. Nat. Compd., 52, 870 (2016).
T. C. Cheng, S. R. Roffler, S. C. Tzou, K. H. Chuang, Y. C. Su, C. H. Chuang, C. H. Kao, C. S. Chen, I. H. Harn, K. Y. Liu, T. L. Cheng, and Y. L. Leu, J. Am. Chem. Soc., 134, 3103 (2012).
O. V. Andreeva, R. R. Sharipova, I. Yu. Strobykina, M. A. Kravchenko, A. S. Strobykina, A. D. Voloshina, R. Z. Musin, and V. E. Kataev, Zh. Org. Khim., 51, 1349 (2015).
S. A. K. Johgkees and S. G. Withers, J. Am. Chem. Soc., 133, 19334 (2011).
O. V. Andreeva, R. R. Sharipova, B. F. Garifullin, I. Yu. Strobykina, and V. E. Kataev, Chem. Nat. Compd., 51, 689 (2015).
M. Upreti, G. Dubois, and I. Prakash, Molecules, 17, 4186 (2012).
K. Hirose, Y. Kosuge, T. Otomatsu, K. Endo, and K. Hasegawa, Tetrahedron Lett., 44, 2171 (2003).
J. Kiss, Adv. Carbohydr. Chem. Biochem., 25, 229 (1974).
H. B. Wood, R. Allerton, H. W. Diehl, and H. G. Fletcher, J. Org. Chem., 20, 875 (1955).
G. N. Bollenback, J. W. Long, D. G. Benjamin, and J. A. Lindquist, J. Am. Chem. Soc., 77, 3310 (1955).
National Committee for Clinical Laboratory Standards, Methods for Dilution Antimicrobial Susceptibility. Tests for Bacteria That Grow Aerobically, 6th Ed.: Approved standard. M7-A5, NCCLS, Wayne, Pa., USA, 2000.
National Committee for Clinical Laboratory Standards, Reference Method for Broth Dilution Antifungal Susceptibility Testing of Conidium-Forming Filamentous Fungi: Proposed Standard, M38-P, NCCLS, Wayne, Pa., USA, 1998.
Acknowledgment
The work was supported financially by the Russian Science Foundation (Grant No. 14-50-00014).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2017, pp. 941–944.
Rights and permissions
About this article
Cite this article
Sharipova, R.R., Andreeva, O.V., Strobykina, I.Y. et al. Synthesis and Antimicrobial Activity of Glucuronosyl Derivatives of Steviolbioside from Stevia rebaudiana . Chem Nat Compd 53, 1107–1111 (2017). https://doi.org/10.1007/s10600-017-2211-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-017-2211-0