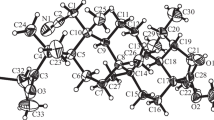
Caryophylla-4,8-dien-5-one (betulenone) was synthesized from α-betulenol in 96% yield. Thioacetates and sulfides with aromatic and heterocyclic fragments were newly synthesized from it in yields up to 95% and diastereomeric excesses (de) up to 80% via addition of thioacetic acid and thiols.

Similar content being viewed by others
References
M. A. Getman and J. Gertsch, Pat. WO2013184036 A3, Dec. 12, 2013.
M. J. Chavan, P. S. Wakte, and D. B. Shinde, Phytomedicine, 17, 149 (2010).
W. Zhang, Z. Yao, Y. W. Zhang, X. X. Zhang, Y. Takaishi, and H. Q. Duan, Planta Med., 76, 1882 (2010).
L. Siaz-Urra, J. C. Racero, A. J. Macias-Sanchez, R. Hernandez-Galan, J. R. Hanson, M. Perez-Gonzalez, and I. G. Collado, J. Agric. Food Chem., 57, 2420 (2009).
X. Cheng, N. Harzdorf, Z. Khaing, D. Kang, A. M. Camelio, T. Shaw, C. E. Schmidt, and D. Siege, Org. Biomol. Chem., 10, 383 (2012).
M. Xian, K. Ito, T. Nakazato, T. Shimizu, C.-K. Chen, K. Yamato, A. Murakami, H. Ohigashi, Y. Ikeda, and M. Kizaki, Cancer Sci., 98, 118 (2007).
Q.-X. Wu, Y.-P. Shi, and L. Yang, Org. Lett., 6, 2313 (2004).
E. P. Romanenko and A. V. Tkachev, Chem. Sustainable Develop., 15, 571 (2007).
U. Vogt, U. Eggert, A. M. Z. Slawin, D. J. Williams, and H. M. R. Hoffmann, Angew. Chem., Int. Ed. Engl., 29 (12), 1456 (1990).
S. Sobhani and S. Rezazadeh, Phosphorus Sulfur Silicon Relat. Elem., 185, 2076 (2010).
D. P. Nair, S. Chatani, T. Gong, W. Xi, C. R. Fenoli, and C. N. Bowman, Chem. Mater., 26, 724 (2014).
A. V. Tkachev, Chem. Nat. Compd., 23, 393 (1987).
R. Kaiser and D. Lamparsky, Helv. Chim. Acta, 66 (6), 1843 (1983).
A. De Mico, R. Margarita, L. Parlanti, A. Vescovi, and G. Piancatelli, J. Org. Chem., 62, 6974 (1997).
R. N. Salvatore, R. A. Smith, A. K. Nischwitz, and T. Gavin, Tetrahedron Lett., 46, 8931 (2005).
J. K. Shneine and Y. H. Alaraji, Int. J. Sci. Res. (IJSR), 5 (3), 1411 (2016).
D. A. Otte, D. E. Borchmann, C. Lin, M. Weck, and K. A. Woerfel, Org. Lett., 16, 1566 (2014).
CrysAlisPro, Agilent Technologies, Version 1.171.36.28 (release 01-02-2013 CrysAlis171.NET).
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. Howard, and H. Puschmann, J. Appl. Crystallogr., 42, 339 (2009).
G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 64, 112 (2008).
Acknowledgment
The work was supported financially by the RFBR (Project No. 16-03-01064) using equipment at the Khimiya Center for Collective Use (CCU), Inst. Chem., Komi SC, UB, RAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2017, pp. 56–60.
Rights and permissions
About this article
Cite this article
Gyrdymova, Y.V., Sudarikov, D.V., Rubtsova, S.A. et al. Synthesis of New Sesquiterpenoid Thio-Derivatives Based on Betulenone. Chem Nat Compd 53, 66–71 (2017). https://doi.org/10.1007/s10600-017-1913-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-017-1913-7