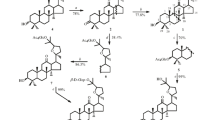
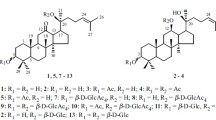
Glycosylation of 6α,12β-dihydroxy-20R,25-epoxydammaran-3-one (panaxatriol 3-ketone, 2) under Koenigs–Knorr reaction conditions and the Helferich modification was studied. Condensation of 2 with 2,3,4,6-tetra-O-acetyl-α-D-glucopyranosylbromide (5) in the presence of Ag2O and 4-Å molecular sieves in CH2Cl2 gave acetylated 6α,12β-dihydroxy-20R,25-epoxydammaran-3-one 12-O-β-D-glucopyranoside whereas in the presence of Hg(CN)2 in nitromethane at 25°C the tetrahydropyran ring opened and, depending on the ratio of reagents, acetylated 12-, 12,25-di-, or 6,12,25-tri-O-β-D-glucopyranosides of 6α,12β,25-trihydroxydammar-20(22)E-en-3-one formed. 6α,12β-Dihydroxy-20R,25-epoxydammaran-3-one 12-O-β-D-glucopyranoside and 12-, 12,25-di-, and 6,12,25-tri-O-β-D-glucopyranosides of 6α,12β,25-trihydroxydammar-20(22)E-en-3-one were synthesized for the first time.
Similar content being viewed by others
References
L. N. Atopkina, N. F. Samoshina, and N. I. Uvarova, Chem. Nat. Compd., 25, 690 (1989).
L. N. Atopkina, N. I. Uvarova, and G. B. Elyakov, Carbohydr. Res., 303, 449 (1997).
L. N. Atopkina and V. A. Denisenko, Chem. Nat. Compd., 42, 452 (2006).
L. N. Atopkina and V. A. Denisenko, Chem. Nat. Compd., 45, 664 (2009).
N. F. Samoshina, V. L. Novikov, V. A. Denisenko, and N. I. Uvarova, Chem. Nat. Compd., 19, 299 (1983).
L. N. Atopkina and V. A. Denisenko, Chem. Nat. Compd., 48, 1111 (2012).
N. K. Kochetkov, A. F. Bochkov, T. A. Sokolovskaya, and N. I. Snyatkova, Carbohydr. Res., 16, 17 (1971).
S. Shibata, T. Ando, and O. Tanaka, Chem. Pharm. Bull., 14, 1157 (1966).
S. Shibata, O. Tanaka, K. Soma, Y. Iida, T. Ando, and H. Nakamura, Tetrahedron Lett., 207 (1965).
Acknowledgment
The work was supported financially in part by the Interdisciplinary Integrated Project for Basic Research of the Siberian, Far-East, and Ural Branches, RAS (No. 12-II-SU-05-006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2014, pp. 751–757.
Rights and permissions
About this article
Cite this article
Atopkina, L.N., Denisenko, V.A. Glycosylation of 6α,12β-dihydroxy-20R,25-epoxydammaran-3-one. Chem Nat Compd 50, 868–875 (2014). https://doi.org/10.1007/s10600-014-1103-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-014-1103-9
Keywords
- dammarane-type triterpenoids
- glycosylation
- panaxatriol 3-ketone
- 6α,12β-dihydroxy-20R,25-epoxydammaran-3-one
- 6α,12β-dihydroxy-20R,25-epoxydammaran-3-one 12-O-β-D-glucopyranoside
- 6α,12β,25-trihydroxydammar-20(22)E-en-3-one 12-O-β-D-glucopyranoside
- 6α,12β,25-trihydroxydammar-20(22)E-en-3-one 12,25-di-O-β-D-glucopyranoside
- 6α,12β,25-trihydroxydammar-20(22)E-en-3-one 6,12,25-tri-O-β-D-glucopyranoside