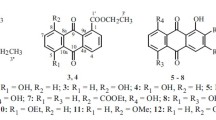
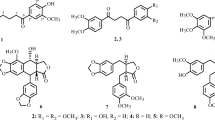
Two new anthraquinones, siameaquinones A (1) and B (2), together with seven known anthraquinones (3–9), were isolated from the stems of Cassia siamea. The structure of 1–9 was elucidated by spectroscopic methods, including extensive 1D and 2D NMR techniques. Compounds 1 and 2 were also evaluated for their cytotoxicities against five human tumor cell lines. The results revealed that compound 1 showed moderate cytotoxicities against A549 and SHSY5Y cells with IC50 values of 5.2 and 6.3 μM, and compound 2 showed moderate cytotoxicities against NB4 and SHSY5Y cells with IC50 values of 8.8 and 7.5 μM, respectively.

Similar content being viewed by others
References
V. Duraipandiyan and S. Ignacimuthu, J. Ethnopharmacol., 112, 590 (2007).
S. Rajan, D. S. Baburaj, M. Sethuraman, and S. Parimala, Ethnobotany, 6, 19 (2001).
J. Ma, L. X. Zhang, and Y. H. Guan, Chin. J. Ethnomed. Ethnopharm., 5, 178 (2004).
K. A. Abo, A. A. Adeyemi, and A. O. Sobowale, Afr. J. Med. Med. Sci., 30, 9 (2001).
S. Aurapa and G. Wandee, Int. J. Biomed. Pharm. Sci., 3, 42 (2009).
P. Sartorelli, S. P. Andrade, M. S. Melhem, F. O. Prado, and A. G. Tempone, Phytother. Res., 21, 644 (2007).
Y. H. Kuo, P. H. Lee, and Y. S. Wein, J. Nat. Prod., 65, 1165 (2002).
Z. Zuraini, C. Yeng, L. L. Yee, Y. L. Lachimanan, N. S. Lai, and S. Sreenivasan, Molecules, 16, 7583 (2011).
Q. F. Hu, B. Zhou, X. M. Gao, L. Y. Yang, L. D. Shu, Y. Q. Shen, G. P. Li, C. T. Che, and G. Y. Yang, J. Nat. Prod., 75, 1909 (2012).
B. Theeshan, S. N. Vidushi, and I. A. Okezie, Afr. J. Biotechnol., 4, 1530 (2005).
X. M. Gao, L. D. Shu, L. Y. Yang, Y. Q. Shen, M. Z. Cui, X. M. Li, and Q. F. Hu, Heterocycles, 87, 125 (2013).
D. Q. Yu and J. S. Yang, Handbook of Analytical Chemistry, Vol. 7, Nuclear Magnetic Resonance spectroscopy, 2nd ed., Chemical Industry Press, Beijing, 1999, p. 892–896.
I. Kubo and Y. Murai, Phytochemistry, 31, 1063 (1992).
T. S. Lv and D. Z. Zhou, Chin. J. Pharm. Pract., 19, 107 (2001).
Y. Igarashi, M. E. Trujillo, E. Martinez-Molina, S. Yanase, S. Miyanaga, T. Obata, H. Sakurai, I. Saiki, T. Fujita, and T. Furumai, Bioorg. Med. Chem. Lett., 17, 3702 (2007).
L. F. Tietze, K. M. Gericke, and I. Schuberth, Eur. J. Org. Chem., 4563 (2007).
Y. C. Hu, E. D. Martinez, and J. B. MacMillan, J. Nat. Prod., 75, 1759 (2012).
D. R. Mou, W. Zhao, T. Zhang, L. Wan, G. Y. Yang, Y. K. Chen, Q. F. Hu, and M. M. Miao, Heterocycles, 85, 2485 (2012).
D. Jeevitha and K. Amarnath, Colloids Surfaces B, 101, 126 (2013).
M. Endale, A. Ekberg, J. P. Alao, H. M. Akala, A. Ndakala, P. Sunnerhagen, M. Erdelyi, and A. Yenesew, Molecules, 18, 311 (2013).
T. Mosmann, J. Immunol. Methods, 65, 55 (1983).
Acknowledgment
This research was supported by the National Natural Science Foundation of China (No. 21032085), the Excellent Scientific and Technological Team of Yunnan High School (2010CI08), the Yunnan University of Nationalities Green Chemistry and Functional Materials Research for Provincial Innovation Team (2011HC008), the National Undergraduates Innovating Experimentation Project (2011HX18), and start-up funds of the Yunnan University of Nationalities.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2014, pp. 711–714.
Rights and permissions
About this article
Cite this article
Ye, YQ., Xia, CF., Li, YK. et al. Anthraquinones from Cassia siamea and Their Cytotoxicity. Chem Nat Compd 50, 819–822 (2014). https://doi.org/10.1007/s10600-014-1091-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-014-1091-9