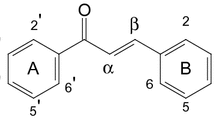
Two new natural products, 3-carbonyl-4-androstene-17-carboxylic acid (1) and trans-4-hydroxy-2-nonenoic acid (4), along with ten known compounds, were isolated from the genus Pottsia (Apocynaceae). Their structures were established on the basis of spectroscopic analysis, especially by means of 1D, 2D NMR and HR-ESI-MS analyses. All the compounds were isolated from the genus Pottsia for the first time. (+)Catechin (6), (–)epicatechin (7), cinnamtannin B-1 (11), and aesculitannin B (12) showed neuroprotective effects against corticosterone–induced neuronal SH-SY5Y cells and DPPH free-radical scavenging activity with EC50 of 16.2,12.7, 5.6, and 4.8 μM.

Similar content being viewed by others
References
Jiangsu New Medical College, Chinese Materia Medica Dictionary, Shanghai People Publishing House, Shanghai, 1977, 1062 pp.
S. J. Wei, X. Y. Huang, and H. Chen, J. Chin. Nat. Med., 3, 41 (2008).
M. M. M. Pinto, A. Kijjoa, I. Mondranondra, A. B. Gutierrez, and W. Herz, Phytochemistry, 29, 1985 (1990).
H. Masao, H. Sumitra, Y. Kavata, Y. Tezuka, T. Kikuchi, and T. Namba, Chem. Pharm. Bull., 35, 3315 (1987).
E. Kiehlmann and A. S. Tracey, Magn. Reson. Chem., 26, 204 (1988).
A. L. Davis, Y. Cai, A. P. Davies, and J. R. Lewis, Magn. Reson. Chem., 34, 887 (1996).
D. Jacques and E. Haslam, Chem. Soc. Perkin Trans. I, 2663 (1974).
K. Ohashi, W. Hisashi, and O. Yasuaki, Chem. Pharm. Bull., 42, 1924 (1994).
H. Achendach, M. Lowel, R.Waibel, M. Gupta, and P. Solis, Planta Med., 58, 270 (1992).
K. Kamiya, C. Watanabe, H. Endang, M. Umar, and T. Satake, Chem. Pharm. Bull., 49, 551 (2001).
G. G. Mellado, E. Zubia, M. J. Ortega, and P. J. Lopez-Gonzalez, Steroids, 69, 291 (2004).
F. Q. Wang and Y. J. Yuan, Chin. Pharm. J., 5, 313 (2001).
C. J. Smith, D. Abbana, V. S. Bernan, W. M. Maiese, M. Greenstein, J. Jompa, A. Tahir, and C. M. Ireland, J. Nat. Prod., 63, 142 (2000).
J. Brichac, A. Honzatko, and M. J. Picklo, J. Chromatogr. A, 1149, 305 (2007).
P. Liu, Y. Hu, D. H. Guo, D. X. Wang, H. H. Tu, L. Ma, T. T. Xie, and L. Y. Kong, Phytomedicine, 17, 794 (2010).
M. S. Blois, Nature, 181, 1199 (1958).
Acknowledgment
We are grateful to Mr. Li-Ping Kang for the NMR and HR-EI-MS measurement in the Instrumentation Center of the Academy of Military Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2012, pp. 890–892.
Rights and permissions
About this article
Cite this article
Mu, LH., Qiang-Feng, J., Liu, P. et al. Chemical constituents of the roots of Pottsia laxiflora . Chem Nat Compd 48, 1004–1007 (2013). https://doi.org/10.1007/s10600-013-0450-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-013-0450-2