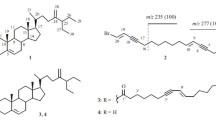
The new compound isoacremine D (1) and acremine A (2) that was previously unknown for marine organisms were isolated from the marine isolate of the fungus Myceliophthora lutea. It was found that acremine A in CHCl3 was converted through the action of light into spirocompounds called by us spiroacremines A (3) and B (4). The structures of the compounds were elucidated based on NMR spectroscopic and high-resolution mass spectrometric data. The absolute stereochemistry of the spiroacremines was determined by Mosher’s method. It was shown that isoacremine D exhibited antimicrobial activity against Staphylococcus aureus at a concentration of 200 μg/mL and cytotoxic activity against embryos of the sea urchin Strongylocentrotus nudus. It was found that 1–4 exhibited cytotoxic activity against sea urchin sperm cells.



Similar content being viewed by others
References
J. W. Blunt, B. R. Copp, W.-P. Hu, M. H. G. Munro, P. T. Northcote, and M. R. Prinsep, Nat. Prod. Rep., 24, 31 (2007).
K. Koyama, M. Ishino, K. Takatori, T. Sugita, K. Kinoshita, and K. Takahashi, Tetrahedron Lett., 45, 6947 (2004).
Z. H. Hin, Y. Fang, L. Du, T. Zhu, L. Duan, J. Chen, Q.-Q. Gu, and W.-M. Zhu, J. Nat. Prod., 70, 853 (2007).
C. A. N. Van Oorschot, Persoonia, 9, 401 (1977).
O. Nozawa, T. Okazaki, N. Sakai, T. Komurasaki, K. Hanada, S. Morimoto, Z. X. Chen, B. M. He, and K. Mizoue, J. Antibiot., 48, 113 (1995).
O. Nozawa, T. Okazaki, S. Morimoto, Z. X. Chen, B. M. He, and K. Mizoue, J. Antibiot., 53, 1296 (2000).
X. Gao, M. Nakadai, and B. Snider, Org. Lett., 5, 451 (2003).
Yu. L. Yang, C. P. Lu, M. Y. Chen, K. Yu. Chen, Y. C. Wu, and S. H. Wu, Chem. Eur. J., 13, 6985 (2007).
G. Assante, S. Dallavalle, L. Malpezzi, G. Nasini, S. Burruano, and L. Torta, Tetrahedron, 61, 7686 (2005).
A. Arnone, G. Assante, A. Bava, S. Dallavalle, and G. Nasini, Tetrahedron, 65, 786 (2009).
N. S. Bhacca and D. H. Williams, Application of NMR Spectroscopy in Organic Chemistry, Helden-Day Inc. (1964).
T. Kuzumi, T. Ooi, Y. Ohkubo, and T. Yabuuchi, Bull. Chem. Soc. Jpn., 79, 965 (2006).
V. I. Bilai, Methods of Experimental Mycology [in Russian], Naukova Dumka, Kiev, 1982, p. 5525.
W. J. Shim, S. H. Hong, I. G. Agafonova, and D. L. Aminin, Bull. Environ. Contam. Toxicol., 77, 755 (2006).
Acknowledgment
The work was supported financially by the Russian Foundation for Basic Research (Projects Nos. 09-04-00388 and 08-04-00289), the RAS Presidium (Molecular and Cellular Biology Program), the RF Federal Agency for Science and Innovation (State Contract No. 02.518.11.7169), and a Grant of the RF President (Project NSh-3531.2010.4).
Author information
Authors and Affiliations
Corresponding author
Additional information
T. A. Kuznetsova (Deceased).
Translated from Khimiya Prirodnykh Soedinenii, No. 3, pp. 345–349, May–June, 2011.
Rights and permissions
About this article
Cite this article
Smetanina, O.F., Yurchenko, A.N., Kalinovskii, A.I. et al. Biologically active metabolites from the marine isolate of the fungus Myceliophthora lutea . Chem Nat Compd 47, 385–390 (2011). https://doi.org/10.1007/s10600-011-9940-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-011-9940-2