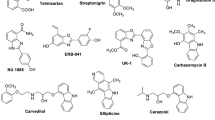
The review summarizes data on the synthesis of fused heterocyclic aromatic compounds by the Diels–Alder reaction, including as part of cascade reactions, and the dehydro-Diels–Alder reaction published over the past five years. In all the examples presented, the Diels–Alder reaction was used to assemble the polycyclic core of the considered molecules.
Similar content being viewed by others
References
Stępień, M.; Gońka, E.; Żyła, M.; Sprutta, N. Chem. Rev. 2017, 117, 3479.
Zhao, X.; Chaudhry, S. T.; Mei, J. Adv. Heterocycl. Chem. 2017, 121, 133.
Kondratov, I. S.; Tolmachova, N. A.; Haufe, G. Eur. J. Org. Chem. 2018, 3618.
Dyan, O. T.; Borodkin, G. I.; Zaikin, P. A. Eur. J. Org. Chem. 2019, 7271.
Yu, F.; Li, D.; Wei, Y.; Kang, R. M.; Guo, Q. X. Tetrahedron 2018, 74, 1965.
Fang, R.-J.; Yan, C.; Sun, J.; Han, Y.; Yan, C.-G. Beilstein J. Org. Chem. 2021, 17, 2425.
Gulbrandsen, H. S.; Serigstad, H.; Lovell Read, M.; Joos, I.; Gundersen, L.-L. Eur. J. Org. Chem. 2019, 6044.
Manikandan, P.; Karunakaran, J.; Varathan, E.; Schreckenbach, G.; Mohanakrishnan, A. K. Chem. Commun. 2020, 56, 15317.
Zhang, Z.; Zhang, Q. Mater. Chem. Front. 2020, 4, 3419.
Chou, T.-C.; Cheng, J.-F.; Gholap, A. R.; Huang, J. J.-K.; Chen, J.-C.; Huang, J.-K.; Tseng, J.-C. Tetrahedron 2019, 75, 130689.
Zhang, P.-F.; Zhuang,-F. D.; Sun, Z.-H.; Lu, Y.; Wang, J.-Y.; Pei, J. J. Org. Chem. 2020, 85, 241.
Kotha, S.; Aswar, V. R.; Chinnam, A. K. Tetrahedron Lett. 2017, 58, 4360.
Bornadiego, A.; Diáz, J.; Marcos, C. F. J. Org. Chem. 2019, 84, 7426.
Hu, Z.; Dong, J.; Li, Z.; Yuan, B.; Wei, R.; Xu, X. Org. Lett. 2018, 20, 6750.
Sun, X.-S.; Diao, X.-Y.; Dong, X.-Q.; Wang, C.-J. Chem. Sci. 2022, 10448.
Vázquez-Vera, Ó.; Segura-Olvera, D.; Rincón-Guevara, M. A.; Gutiérrez-Carrillo, A.; García-Sánchez, M. A.; Ibarra, I. A.; Lomas-Romero, L.; Islas-Jácome, A.; González-Zamora, E. Molecules 2018, 23, 2029.
Kappe, C. O.; Pieber, B.; Dallinger, D. Angew. Chem., Int. Ed. Engl. 2013, 52, 1088.
Arepalli, S. K.; Park, B.; Jung, J.-K.; Lee, K.; Lee, H. Tetrahedron Lett. 2017, 58, 449.
Raju, S.; Annamalai, P.; Chen, P.-L.; Liu, Y.-H.; Chuang, S.-C. Chem. Commun. 2017, 53, 6247.
Talukdar, R.; Singh, V.; Mourya, H.; Nasibullah, M.; Tiwari, B. J. Org. Chem. 2021, 86, 12277.
Wu, F.; Lu, S.; Zhu, S. Adv. Synth. Catal. 2020, 362, 5632.
Kudoh, T.; Fujisawa, S.; Kitamura, M.; Sakakura, A. Synlett 2017, 2189.
Wang, H.; Wang, S.; Qin, T.; Zi, W. Chem.–Eur. J. 2018, 24, 17911.
Zhang, L.; Jin, T.; Guo, Y.; Martin, A. C.; Sun, K.; Dudley, G. B.; Yang, J. J. Org. Chem. 2021, 86, 16716.
Shibata, T.; Sekine, A.; Mitake, A.; Kanyiva, K. S. Angew. Chem., Int. Ed. 2018, 57, 15862.
Hu, Y.; Ma, J.; Li, L.; Hu, Q.; Lv, S.; Liu, B.; Wang, S. Chem. Commun. 2017, 53, 1542.
Lee, D.; Ross, S. P.; Xiao, X.; Hoye, T. R. Chem 2021, 7, 2527.
Jassas, R. S.; Mughal, E. U.; Sadiq, A.; Alsantali, R. I.; Al-Rooqi, M. M.; Naeem, N.; Moussa, Z.; Ahmed, S. A. RSC Adv. 2021, 11, 32158.
Ammon, F.; Sauer, S. T.; Lippert, R.; Lungerich, D.; Reger, D.; Hampel, F.; Jux, N. Org. Chem. Front. 2017, 4, 861.
Reger, D.; Haines, P.; Heinemann, F. W.; Guldi, D. M.; Jux, N. Angew. Chem., Int. Ed. 2018, 57, 5938.
Yin, J.; Hu, Y.; Zhang, D.; Li, X.; Jin, W. Tetrahedron 2017, 73, 5794.
Urieta-Mora, J.; Krug, M.; Wiebke, A.; Perles, J.; Fernández, I.; Molina-Ontoria, A.; Guldi, D. M.; Martín, N. J. Am. Chem. Soc. 2020, 142, 4162.
Shi, Y.; Li, C.; Ma, S.; Zhang, Y. Synthesis 2018, 102.
Dusold, C.; Haines, P.; Platzer, B.; Guldi, D. M.; Hirsch, A. Chem.–Eur. J. 2021, 27, 6511.
Zink-Lorre, N.; Doncel-Giménez, A.; Font-Sanchis, E.; Calbo, J.; Sastre-Santos, Á.; Ortí, E.; Fernández-Lázaro, F. Org. Chem. Front. 2019, 6, 2860.
Hahn, U.; Maisonhaute, E.; Nierengarten, J. Angew. Chem., Int. Ed. 2018, 57, 10635.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2023, 59(4/5), 201–216
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dyan, O.T., Zaikin, P.A. The Diels–Alder reaction in the synthesis of fused heterocyclic aromatic compounds. Chem Heterocycl Comp 59, 201–216 (2023). https://doi.org/10.1007/s10593-023-03183-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-023-03183-1