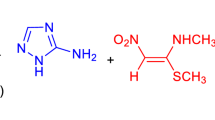
The possibility of preparation of juglone via Diels–Alder reaction of 3-hydroxypyridazine and 2-nitroquinone was analyzed on the basis of DFT computational study. It was found that key stage of the proposed transformation is head-to-head, polar cycloaddition process, which is realized without formation of zwitterionic intermediate. Further transformations of the intermediate exhibit nature of pseudocyclic reactions with nonconcerted reorganization of the electron density.




Similar content being viewed by others
References
(a) Domingo, L. R.; Ríos-Gutiérrez, M.; Chamorro, E.; Pérez, P. ChemistrySelect 2016, 1, 6026. (b) Jasiński, R. Comput. Theor. Chem. 2018, 1125, 77. (c) Jasiński, R.; Kula, K.; Kącka, A.; Mirosław, B. Monatsh. Chem. 2017, 148, 909. (d) Mirosław, B.; Babyuk, D.; Łapczuk-Krygier, A.; Kącka-Zych, A.; Demchuk, O. M.; Jasiński, R. Monatsh. Chem. 2018, 149, 1877.
(a) Lapinski, L.; Fulara, J.; Czerminski, R.; Nowak, M. J. Spectrochim. Acta, Part A 1990, 46, 1087. (b) Tavares, F. X.; Boucheron, J. A.; Dickerson, S. H.; Griffin, R. J.; Preugschat, F.; Thomson, S. A.; Wang, T. Y.; Zhou, H.-Q. J. Med. Chem. 2004, 47, 4716.
(a) Chen, Yu.; Ying, W.; Harmata, M. Tetrahedron Lett. 2011, 52, 480. (b) Polgatti, V.; Valderrama, J. A.; Tapia, R. Synth. Commun. 1990, 20, 1085.
(a) Jasiński, R. React. Kinet., Mech. Catal. 2016, 119, 49. (b) Houk, K. N.; Liu, F.; Yang, Z.; Seeman, J. I. Angew. Chem., Int. Ed. 2021, 60, 12660.
(a) Jasiński, R. J. Mol. Graphics Modell. 2017, 75, 55. (b) Jasiński, R. Comput. Theor. Chem. 2014, 1046, 93. (c) Jasiński, R. J. Fluorine Chem. 2018, 206, 1. (d) Ernd, M.; Heuschmann, M.; Zipse, H. Helv. Chim. Acta 2005, 88, 1491.
Jasiński, R. Symmetry 2021, 13, 1911.
Kula, K.; Dobosz, J.; Jasiński, R.; Kącka-Zych, A.; Łapczuk-Krygier, A.; Mirosław, B.; Demchuk, O. M. J. Mol. Struct. 2020, 1203, 127473.
Kącka-Zych, A.; Jasiński, R. New J. Chem. 2021, 45, 9491.
Jasiński, R.; Mirosław, B.; Demchuk, O. M.; Babyuk, D.; Łapczuk-Krygier, A. J. Mol. Struct. 2016, 1108, 689.
(a) Domingo, L. R. RSC Adv. 2014, 4, 32415. (b) Domingo, L. R.; Ríos-Gutiérrez, M. Sci. Rad. 2023, 2, 1.
Domingo, L. R. Molecules 2016, 21, 1319.
Jasiński, R. RSC Adv. 2015, 5, 50070.
Woliński, P.; Kącka-Zych, A.; Demchuk, O. M.; Łapczuk-Krygier, A.; Mirosław, B.; Jasiński, R. J. Clean. Product. 2020, 275, 122086.
Kącka, A.; Jasiński, R. Heteroat. Chem. 2016, 27, 279.
Domingo, L. R.; Sáez, J. A. Org. Biomol. Chem. 2009, 7, 3576.
Chai J.-D.; Head-Gordon, M. Phys. Chem. Chem. Phys. 2008, 10, 6615.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.; Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J. A., Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas, Ö.; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J.; Fox, D. J. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford, 2013.
Scalmani, G.; Frisch, M. J. J. Chem. Phys. 2010, 132, 114110.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2023, 59(3), 179–182
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jasiński, R. On the question of selective protocol for the preparation of juglone via (4+2) cycloaddition involving 3-hydroxypyridazine: DFT mechanistic study. Chem Heterocycl Comp 59, 179–182 (2023). https://doi.org/10.1007/s10593-023-03180-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-023-03180-4