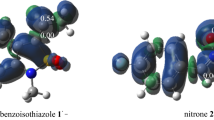
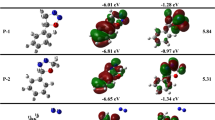
This theoretical study, performed using density functional theory at the B3LYP/6-311+G(d,p) level, shows that the most likely route to obtain 3-phenyl[1,2,4]oxadiazolo[4,5-a]benzimidazole from the reaction of 2-chlorobenzimidazole with benzonitrile oxide implies the presence of anionic species. A concerted [3+2] cycloaddition reaction on the imidoyl group of 2-chlorobenzimidazole is not possible. The presence of a polar protic solvent (MeOH) favors the reaction. The analysis of the Wiberg indices shows that the transition states are earlier in MeOH than in THF or gas phase. Finally, topological analysis of the electron localization function indicates that the formation of the N4–C3 and C5–O bonds and the breaking of the C5–Cl bond in the preparation of 3-phenyl[1,2,4]oxadiazolo[4,5-a]benzimidazole are marked for each by the presence of an asynaptic basin V(Asyn) and proceed through tetrahedral intermediates, indicating the nonconcerted nature of the mechanism. These results are in good agreement with the experimental results.












Similar content being viewed by others
References
Çevik, U. A.; Osmaniye, D.; Çavuşoğlu, B. K.; Sağlik, B. N.; Levent, S.; Ilgin, S.; Can, N. Ö.; Özkay, Y.; Kaplancıklı, Z. A. Med. Chem. Res. 2019, 28, 2252.
Gupta, V. K.; Kaur, B.; Kaur, A.; Kaur, A.; Gupta, M. Ind. J. Pharm. Biol. Res. 2018, 6(2), 64.
Rashid, M.; Husain, A.; Mishra, R. Eur. J. Med. Chem. 2012, 54, 855.
Acar Çevik, U.; Sağlık, B. N.; Osmaniye, D.; Levent, S.; Kaya Çavuşoğlu, B.; Karaduman, A. B.; Eklioğlu, Ö. A.; Özkay, Y.; Kaplancıklı, Z. A. J. Enzyme Inhib. Med. Chem. 2020, 35, 1657.
Rathore, A.; Rahman, M. U.; Siddiqui, A. A.; Ali, A.; Shaharyar, M. Arch. Pharm. 2014, 347, 923.
Kamal, A.; Reddy, T. S.; Vishnuvardhan, M. V. P. S.; Nimbarte, V. D.; Subba Rao, A. V.; Srinivasulu, V.; Shankaraiah, N. Bioorg. Med. Chem. 2015, 23, 4608.
Palumbo Piccionello, A. Int. J. Mol. Sci. 2021, 22, 3988.
Jiang, K.-M.; Zhang, J.-Q.; Jin, Y.; Lin, J. Asian J. Org. Chem. 2017, 6, 1620.
Jawalekar, A. M.; Reubsaet, E.; Rutjes, F. P. J. T.; van Delft, F. L. Chem. Commun. 2011, 47, 3198.
Zhao, G.; Liang, L.; Wen, C. H. E.; Tong, R. Org. Lett. 2019, 21, 315.
Lin, B.; Yu, P.; He, C. Q.; Houk, K. N. Bioorg. Med. Chem. 2016, 24, 4787.
Ouahdi, Z.; Ourhriss, N.; Oualed Aitouna, A.; Barhoumi, A.; Belghiti, M. E.; Moubarik, A.; El Abdallaoui, H. E. A.; El Idrissi, M.; Zeroual, A. Theor. Chem. Acc. 2022, 141, 50.
Zeroual, A.; Ríos-Gutiérrez, M.; El Idrissi, M.; El Abdallaoui, H. E. A.; Domingo, L. R. Int. J. Quantum Chem. 2019, 119, e25980.
Domingo, L. R.; Chamorro, E.; Pérez, P. Eur. J. Org. Chem. 2009, 3036.
El Idrissi, M.; El Ghozlani, M.; Eşme, A.; Ríos-Gutiérrez, M.; Ouled Aitouna, A.; Salah, M.; El Abdallaoui, H. E. A.; Zeroual, A.; Mazoir, N. Organics 2021, 2, 1.
Zahnoune, R.; Asserne, F.; Ourhriss, N.; Ouled Aitouna, A.; Barhoumi, A.; Hakmaoui Y.; Belghiti, M. E.; Abouricha, S.; El ajlaoui, R.; Zeroual, A. J. Mol. Struct. 2022, 1269, 133630.
Domingo, L. R.; Aurell, M. J.; Pérez, P. Tetrahedron 2014, 70, 4519.
Domingo, L. R.; Emamian, S. R. Tetrahedron 2014, 70, 1267.
Ríos-Gutiérez, M.; Domingo, L. R. Eur. J. Org. Chem. 2018, 267.
Domingo, L. R.; Sáez, J. A. Org. Biomol. Chem. 2009, 7, 3576.
Domingo, L. R.; Aurell, M. J.; Jalal, R.; Esseffar, M. J. Mol. Struct.: THEOCHEM 2010, 942, 26.
Mohammad-Salim, H. A.; Ahmed Basheer, H.; Abdallah, H. H.; Zeroual, A.; Abdi Jamil, L. New J. Chem. 2021, 45, 262.
Salah, M.; Belghiti, M. E.; Aitouna, A. O.; Zeroual, A.; Jorio, S.; El Alaoui Abdellaoui, H.; El Hadki, H.; Marakchi, K.; Komiha, N. J. Mol. Graphics Modell. 2021, 102, 107763.
Harrison, D.; Ralph, J. T. J. Chem. Soc. 1965, 236.
Lunn, W. H. W.; Harper, R. W. J. Heterocycl. Chem. 1971, 8, 141.
Ponomar', V. S.; Kochergin, P. M. Chem. Heterocycl. Compd. 1972, 8, 229.
Ajay, A.; Gupt, M. P.; Devender, N.; Tripathi, R. P. Mol. Diversity 2012, 16, 335.
Tanaka, M.; Ubukata, M.; Matsuo, T.; Yasue, K.; Matsumoto, K.; Kajimoto, Y.; Ogo T.; Inaba, T. Org. Lett. 2007, 9, 3331.
Scrocco, E.; Tomasi, J. Adv. Quantum Chem. 1978, 11, 115.
Zeroual, A.; Ríos-Gutiérrez, M.; El Ghozlani, M.; El Idrissi, M.; Ouled Aitouna, A.; Salah, M.; El Alaoui, El Abdallaoui, H.; Domingo, L. R. Theor. Chem. Acc. 2020, 139, 37.
Fukui, K. Acc. Chem. Res. 1981, 14, 363.
Sun, P.; Liu, M.; Pu, W.; Jin, Y.; Liu, S.; Cao, Q.; Ding, Z. Authorea 2020. DOI: https://doi.org/10.22541/au.159078914.44654675.
Reed, A. E.; Weinstock, R. B.; Weinhold, F. J. Chem. Phys. 1985, 83, 735.
Fukui, K. J. Phys. Chem. 1970, 74, 4161.
Wiberg, K. B. Tetrahedron 1968, 24, 1083.
Reed, A. E.; Curtiss, L. A.; Weinhold, F. Chem. Rev. 1988, 88, 899.
Glendening, E. D.; Landis, C. R.; Weinhold, F. Wiley Interdiscip. Rev.: Comput. Mol. Sci. 2012, 2, 1.
Becke, A. D.; Edgecombe, K. E. J. Chem Phys. 1990, 92, 5397.
Cossi, M.; Rega, N.; Scalmani, G.; Barone, V. J. Comput. Chem. 2003, 24(6), 669.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H. P.; Izmaylov, A. F.; Bloino, J.; Zheng, G.; Sonnenberg, J. L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J. A.; Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Rega, N.; Millam, J. M.; Klene, M.; Knox, J. E.; Cross, J. B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R. E.; Yazyev, O.; Austin, A. J.; Cammi, R.; Pomelli, C.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Zakrzewski, V. G.; Voth, G. A.; Salvador, P.; Dannenberg, J. J.; Dapprich, S.; Daniels, A. D.; Farkas, Ö.; Foresman, J. B.; Ortiz, J. V.; Cioslowski, J.; Fox, D. J. Gaussian 09, Revision E.01; Gaussian, Inc.: Wallingford, 2009.
Lee, C.; Yang, W.; Parr, R. G. Phys. Rev. B: Condens. Matter Mater. Phys. 1988, 37, 785.
Becke, A. D. J. Chem. Phys. 1993, 98, 5648.
Parr, R. G.; Szentpály, L. v.; Liu, S. J. Am. Chem. Soc. 1999, 121, 1922.
Domingo, L. R.; Chamorro, E.; Pérez, P. J. Org. Chem. 2008, 73, 4615.
Domingo, L. R.; Pérez, P. Org. Biomol. Chem. 2011, 9, 7168.
Kohn, W.; Sham, L. J. Phys. Rev. 1965, 140, A1133.
Domingo, L. R.; Pérez, P.; Sáez, J. A. RSC Adv. 2013, 3, 1486.
Domingo, L. R. RSC Adv. 2014, 4, 32415.
Reed, A. E.; Curtiss, L. A.; Weinhold, F. Chem. Rev. 1988, 88, 899.
Reed, A. E.; Weinstock, R. B.; Weinhold, F. J. Chem. Phys. 1985, 83, 735.
Zeroual, A.; Ríos-Gutiérrez, M.; Salah, M.; El Alaoui El Abdallaoui, H.; Domingo, L. R. J. Chem. Sci. (Berlin, Ger.) 2019, 131, 75.
El Ghozlani, M.; Barhoumi, A.; Elkacmi, R.; Ouled Aitouna, A.; Zeroual, A.; El Idrissi, M. Chem. Afr. 2020, 3, 901.
Tamilmani, V.; Daul, C. A.; Lage Robles, J.; Bochet, C. G.; Venuvanalingam, P. Chem. Phys. Lett. 2005, 406, 355.
Noury, S.; Krokidis, X.; Fuster, F.; Silvi, B. Comput. Chem. (Oxford, U. K.) 1999, 23, 597.
Ouled Aitouna, Ab.; Barhoumi, A.; El Idrissi, M.; Ouled Aitouna, A.; Zeroual, A.; Mazoir, N.; Chakroun, A.; Benharref, A. Mor. J. Chem. 2021. DOI: https://doi.org/10.48317/IMIST.PRSM/morjchem-v9i1.20462.
Wiberg, K. B.; Rablen, P. R. J. Comput. Chem. 1993, 14, 1504.
Divya, P.; Bena Jothy, V. Chem. Phys. Lett. 2018, 695, 1.
Merino, P.; Chiacchio, M.; Legnani, L.; Tejero, T. Molecules 2017, 22, 1371.
Abbiche, K.; Mohammad-Salim, H.; Salah, M.; Mazoir, N.; Zeroual, A.; El Alaoui El Abdallaoui, H.; El Hammadi, A.; Hilali, M.; Abdallah, H. H.; Hochlaf, M. Theor. Chem. Acc. 2020, 139, 148.
Ouled Aitouna, A.; Belghiti, M. E.; Eşme, A.; Ouled Aitouna, A.; Salah, M.; Chekroun, A.; El Alaoui El Abdallaoui, H.; Benharref, A.; Mazoir, N.; Zeroual, A.; Nejjari, C. J. Mol. Struct. 2021, 1244, 130864.
Zawadzińska, K.; Ríos-Gutiérrez, M.; Kula, K.; Woliński, P.; Mirosław, B.; Krawczyk, T.; Jasiński, R. Molecules 2021, 26, 6774.
Jasiński, R.; Jasińska, E.; Dresler, E. J. Mol. Model. 2017, 23, 13.
Zawadzińska, K.; Kula, K. Organics 2021, 2, 26.
Du, P.; Zhao, J.; Liu, S.; Yue, Z. RSC Adv. 2021, 11, 24238.
Li, H. G.; Kim, C. K.; Lee, B.-S.; Kim, C. K.; Rhee, S. K.; Lee, I. J. Am. Chem. Soc. 2001, 123, 2326.
Zeroual, A.; El Hajbi, A. Can. Chem. Trans. 2015, 3, 430.
Salah, M.; Zeroual, A.; Jorio, S.; El Hadki, H.; Kabbaj, O.; Marakchi, K.; Komiha, N. J. Mol. Graphics Modell. 2020, 94, 107458.
Melin, J.; Seabra, G.; Ortiz, J. V. In Theoretical Aspects of Chemical Reactivity; Toro-Labbé, A., Ed.; Elsevier: Amsterdam, 2007, p. 87.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2023, 59(3), 155–164
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abdoul-Hakim, M., Idrissi, K.E., Zeroual, A. et al. Investigation of the solvent effect, regioselectivity, and the mechanism of the cycloaddition reaction between 2-chlorobenzimidazole and benzonitrile oxide. Chem Heterocycl Comp 59, 155–164 (2023). https://doi.org/10.1007/s10593-023-03177-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-023-03177-z