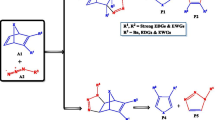
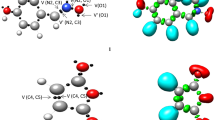
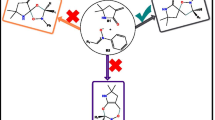
A molecular electron density theory study of the [3+2] cycloaddition reaction of 5-oxo-2-(phenylmethylidene)pyrazolidin-2-ium-1-ide, a simple azomethine imine, with 3-nitro-2-phenyl-2H-chromene was carried out at the B3LYP/6-311G(d,p) computational level in order to unravel the origin of the stereoselectivity experimentally observed. Electron localization function and natural population analysis showed that the azomethine imine is a zwitterionic three-atom component. Analysis of the conceptual density functional theory reactivity indices indicates that azomethine imine is a strong nucleophile, while 3-nitro-2-phenyl-2H-chromene is a good electrophile, which accounts for a polar process confirmed by the high global electron density transfer values. The Parr function indices explain well the experimentally obtained ortho regioselectivity. Analysis of the energy profiles of the possible reactive pathways in gas phase and in solution of i-PrOH points to high exo stereoselectivity and complete ortho regioselectivity of the reaction in excellent agreement with the experimental findings. Analysis of the transition state structures indicates a very asynchronous molecular mechanism for the favored orthoregioisomeric reaction channels. Analysis using noncovalent interactions, quantum theory of atoms in molecules, and independent gradient model based on Hirshfeld distribution indicates that the presence of several hydrogen bonds and van der Waals intermolecular noncovalent interactions are the factors favoring the ortho-exo selectivity. The bonding evolution theory study of the most favorable pathway reveals a two-stage one-step molecular mechanism.










Similar content being viewed by others
References
Kaur, N. J. Heterocycl. Chem. 2015, 52, 953.
Indelicato, J. M.; Pasini, C. E. J. Med. Chem. 1988, 31, 1227.
Ternansky, R. J.; Draheim, S. E.; Pike, A. J.; Counter, F. T.; Eudaly, J. A.; Kasher, J. S. J. Med. Chem. 1993, 36, 3224.
Dorn, H. Chem. Heterocycl. Compd. 1981, 17, 1.
Varvounis, G.; Fiamegos, Y.; Pilidis, G. Adv. Heterocycl. Chem. 2001, 80, 73.
Eicher, T.; Hauptmann, S. The Chemistry of Heterocycles: Structure, Reactions, Syntheses, and Applications; Wiley-VCH: Weinheim, 2003, 2nd ed., Vol. 1–2.
Recent Progress in the Chemical Synthesis of Antibiotics; Lukacs, G.; Ohno, M., Eds.; Springer: Heidelberg, 2012.
Konaklieva, M. I.; Plotkin, B. J. Curr. Med. Chem.: Anti-Infect. Agents 2003, 2, 287.
White, H. L.; Howard, J. L.; Cooper, B. R.; Soroko, F. E.; McDermed, J. D.; Ingold, K. J.; Maxwell, R. A. J. Neurochem. 1982, 39, 271.
Grošelj, U.; Svete, J. ARKIVOC 2015, (vi), 175.
Panfil, I.; Urbańczyk-Lipkowska, Z.; Suwińska, K.; Solecka, J.; Chmielewski, M. Tetrahedron 2002, 58, 1199.
Claramunt, R. M.; Elguero, J. Org. Prep. Proced. Int. 1991, 23, 273.
Jungheim, L. N.; Sigmund, S. K. J. Org. Chem. 1987, 52, 4007.
Kula, K.; Łapczuk, A.; Sadowski, M.; Kras, J.; Zawadzińska, K.; Demchuk, O. M.; Gaurav, G. K.; Wróblewska, A.; Jasiński, R. Molecules 2022, 27, 8409.
Kula, K.; Kącka-Zych, A.; Łapczuk-Krygier, A.; Wzorek, Z.; Nowak, A. K.; Jasiński, R. Molecules 2021, 26, 1364.
Nájera, C.; Sansano, J. M.; Yus, M. Org. Biomol. Chem. 2015, 13(32), 8596.
Emamian, S.; Lu, T.; Domingo, L. R.; Heidarpoor Saremi, L.; Ríos-Gutiérrez, M. Chem. Phys. 2018, 501, 128.
Domingo, L. R.; Ríos-Gutiérrez, M. Molecules 2017, 22, 750.
Domingo, L. R. Molecules 2016, 21, 1319.
Chafaa, F.; Nacereddine, A. K.; Djerourou, A. Theor. Chem. Acc. 2019, 123.
Chouit, H.; Sobhi, C.; Bouasla, S.; Messikh, S.; Kheribeche, A.; Khorief Nacereddine, A. J. Mol. Model. 2022, 28, 104.
Ríos-Gutiérrez, M.; Nasri, L.; Khorief Nacereddine, A.; Djerourou, A.; Domingo, L. R. A J. Phys. Org. Chem. 2018, 31, e3830.
Nacereddine, A. K. J. Mol. Model. 2020, 26, 328.
Khorief Nacereddine, A.; Chafaa, F. Organics 2022, 3, 281.
Nasri, L.; Ríos-Gutiérrez, M.; Nacereddine, A. K.; Djerourou, A.; Domingo, L. R. Theor. Chem. Acc. 2017, 136, 104.
Domingo, L. R.; Kula, K.; Ríos-Gutiérrez, M.; Jasiński, R. J. Org. Chem. 2021, 86, 12644.
Kula, K.; Zawadzińska, K. Curr. Chem. Lett. 2021, 10, 9.
Żmigrodzka, M.; Sadowski, M.; Kras, J.; Dresler, E.; Demchuk, O. M.; Kula, K. Sci. Rad. 2022, 1, 26.
Barkov, A. Y.; Zimnitskiy, N. S.; Kutyashev, I. B.; Korotaev, V. Y.; Sosnovskikh, V. Y. Tetrahedron Lett. 2017, 58, 3989.
Babazadeh, S. M.; Emamian, S.; Zahedi, E. J. Phys. Org. Chem. 2019, 32, e3925.
Reed, A. E.; Weinstock, R. B.; Weinhold, F. J. Chem. Phys. 1985, 83, 735.
Reed, A. E.; Curtiss, L. A.; Weinhold, F. Chem. Rev. 1988, 88, 899.
Huisgen, R. Angew. Chem., Int. Ed. 1963, 2, 565.
Geerlings, P.; De Proft, F.; Langenaeker, W. Chem. Rev. 2003, 103, 1793.
Domingo, L. R.; Ríos-Gutiérrez, M.; Pérez, P. Molecules 2016, 21, 748.
Domingo, L. R.; Ríos-Gutiérrez, M.; Pérez, P. J. Org. Chem. 2018, 83, 2182.
Pérez, P.; Domingo, L. R.; Aurell, M. J.; Contreras, R. Tetrahedron 2003, 59, 3117.
Domingo, L. R.; Aurell, M. J.; Pérez, P.; Contreras, R. Tetrahedron 2002, 58, 4417.
Domingo, L. R.; Ríos-Gutiérrez, M.; Pérez, P. Tetrahedron 2016, 72, 1524.
Sanderson, R. T. Science 1955, 121, 207.
Domingo, L. R.; Pérez, P.; Sáez, J. A. RSC Adv. 2013, 3, 1486.
Yahia, W.; Nacereddine, A. K.; Djerourou, A. Intl. J. Quantum Chem. 2017, 118, e25540.
Chafaa, F.; Hellel, D.; Nacereddine, A. K.; Djerourou, A. Mol. Phys. 2015, 114, 663.
Khorief Nacereddine, A.; Merzoud, L.; Morell, C.; Chermette, H. J. Comput. Chem. 2021, 42, 1296.
Lamri, S.; Heddam, A.; Kara, M.; Yahia, W.; Khorief Nacereddine, A. Organics 2021, 2, 57.
Sadi, S.; Khorief Nacereddine, A.; Djerourou, A. J. Phys. Org. Chem. 2022, 35, e4311.
Ríos-Gutiérrez, M.; Domingo, L. R. Eur. J. Org. Chem. 2019, 267.
Domingo, L. R. A RSC Adv. 2014, 4, 32415.
Johnson, E. R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen, A. J.; Yang, W. J. Am. Chem. Soc. 2010,132, 6498.
Nacereddine, A. K.; Sobhi, C.; Djerourou, A.; Ríos-Gutiérrez, M.; Domingo, L. R. RSC Adv. 2015, 5, 99299.
Ríos-Gutiérrez, M.; Domingo, L. R.; Pérez, P. RSC Adv. 2015, 5, 84797.
Bayoud, B.; Barama, L.; Nacereddine, A. K.; Djerourou, A. Mol. Phys. 2020, 119, e1828635.
Lane, J. R.; Contreras-García, J.; Piquemal, J.-P.; Miller, B. J.; Kjaergaard, H. G. J. Chem. Theory Comput. 2013, 9, 3263.
Bader, R. F. W. Atoms in Molecules. A Quantum Theory; Clarendon Press: Oxford, 1990.
Bader, R. F. W.; Essén, H. J. Chem. Phys. 1984, 80, 1943.
Rozas, I.; Alkorta, I.; Elguero, J. J. Am. Chem. Soc. 2000, 122, 11154.
Lefebvre, C.; Khartabil, H.; Boisson, J.-C.; Contreras-García, J.; Piquemal, J.-P.; Hénon, E. ChemPhysChem 2018, 19, 724.
Klein, J.; Khartabil, H.; Boisson, J.-C.; Contreras-García, J.; Piquemal, J.-P.; Hénon, E. J. Phys. Chem. A 2020, 124, 1850.
Krokidis, X.; Noury, S.; Silvi, B. J. Phys. Chem. A 1997, 101, 7277.
Domingo, L. R.; Ríos-Gutiérrez, M.; Sáez, J. A. RSC Adv. 2015, 5, 37119.
Stephens, P. J.; Devlin, F. J.; Chabalowski, C. F.; Frisch, M. J. J. Phys. Chem. 1994, 98, 11623.
Hehre, W. J.; Radom, L. Schleyer, P. v. R.; Pople, J. A. AB INITIO Molecular Orbital Theory; John Wiley & Sons: New York, 1986, p. 82.
Schlegel, H. B. J. Comput. Chem. 1982, 3, 214.
Cui, C. X.; Liu, Y. J. J. Phys. Org. Chem. 2015, 28, 281.
Tomasi, J.; Persico, M. Chem. Rev. 1994, 94, 2027.
Simkin, B. Y.; Sheikhet, I. I. Quantum Chemical and Statistical Theory of Solutions. A Computational Approach; Ellis Horwood: London, 1995.
Cancès, E.; Mennucci, B.; Tomasi, J. J. Chem. Phys. 1997, 107, 3032.
Barone, V.; Cossi, M.; Tomasi, J. J. Comput. Chem. 1998, 19, 404.
Fukui, K. A J. Phys. Chem. 1970, 74, 4161.
Domingo, L. R.; Pérez, P.; Sáez, J. A. Tetrahedron 2013, 69, 107.
Becke, A. D.; Edgecombe, K. E. J. Chem. Phys. 1990, 92, 5397.
Lu, T.; Chen, F. J. Comput. Chem. 2012, 33, 580.
Pettersen, E. F.; Goddard, T. D.; Huang, C. C.; Couch, G. S.; Greenblatt, D. M.; Meng, E. C.; Ferrin, T. E. J. Comput. Chem. 2004, 25, 1605.
Parr, R. G.; Szentpály, L. V.; Liu, S. J. Am. Chem. Soc. 1999, 12, 1922.
Parr, R. G.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: New York, 1989, p. 70.
Parr, R. G.; Pearson, R. G. J. Am. Chem. Soc. 1983, 105, 7512.
Domingo, L. R.; Chamorro, E.; Pérez, P. J. Org. Chem. 2008, 73, 4615.
Domingo, L. R.; Pérez, P. Org. Biomol. Chem. 2011, 9, 7168.
Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. A.; Nakatsuji, H.; Li, X.; Caricato, M.; Marenich, A.; Bloino, J.; Janesko, B. G.; Gomperts, R.; Mennucci, B.; Hratchian, H. P.; Ortiz, J. V.; Izmaylov, A. F.; Sonnenberg, J. L.; Williams-Young, D.; Ding, F.; Lipparini, F.; Egidi, F.; Goings, J.; Peng, B.; Petrone, A.; Henderson, T.; Ranasinghe, D.; Zakrzewski, V. G.; Gao, J.; Rega, N.; Zheng, G.; Liang, W.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Throssell, K.; Montgomery, J. A., Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Keith, T.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Millam, J. M.; Klene, M.; Adamo, C.; Cammi, R.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Farkas, O.; Foresman, J. B.; Fox, D. J. Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford, 2016.
This work was supported by the Ministry of Higher Education and Scientific Research of the Algerian Government (project PRFU Code: B00L01EN210120220001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2023, 59(3), 128–137
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Messaadia, S., Nacereddine, A.K. & Djerourou, A. Exploring the factors controlling the mechanism and the high stereoselectivity of the polar [3+2] cycloaddition reaction of the N,N'-cyclic azomethine imine with 3-nitro-2-phenyl-2H-chromene. A molecular electron density theory study. Chem Heterocycl Comp 59, 128–137 (2023). https://doi.org/10.1007/s10593-023-03174-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-023-03174-2