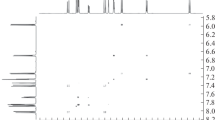
A new spiropyran containing a cationic 3Н-indolium substituent and methoxy groups at positions 5 and 5” of the indoline cycles was synthesized. Hydrolysis of this compound was observed during crystallization from EtOH, leading to the formation of protonated merocyanine form of spiropyran containing a free formyl group. The hydrolysis product was characterized by X-ray structural analysis, the intermolecular interactions in crystal were studied with the CrystalExplorer 21.5 software suite. Quantum-chemical modeling based on the Fukui function distribution was used to establish the preferred site for nucleophilic attack, and a mechanism for hydrolysis was proposed.









Similar content being viewed by others
References
Zhao, W.; Quan, M.; Cao, Z.; Zhang, Y.; Wen, J.; Pan, D.; Dong, Z.; Yang, Z.; Wang, D.; Cao, H.; He, W. Colloids Surf., A 2018, 554, 93.
Xia, Z.; Alphonse, V. D.; Trigg, D. B.; Harrigan, T. P.; Paulson, J. M.; Luong, Q. T.; Lloyd, E. P.; Barbee, M. H.; Craig, S. L. Molecules 2019, 24, 542.
Bispo, M.; van Dijl, J. M.; Szymanski, W. In Molecular Photoswitches: Chemistry, Properties, and Applications; Pianowski, Z. L., Ed.; WILEY-VCH GmbH: Weinheim, 2022, p. 843.
Sauvage, J.-P. Angew. Chem., Int. Ed. 2017, 56, 11080.
Feringa, B. L. Angew. Chem., Int. Ed. 2017, 56, 11060.
Stoddart, J. F. Angew. Chem., Int. Ed. 2017, 56, 11094.
Klajn, R. Chem. Soc. Rev. 2014, 43, 148.
Kozlenko, A. S.; Pugachev, A. D.; Ozhogin, I. V.; El-Sewify, I. M.; Lukyanov, B. S. Chem. Heterocycl. Compd. 2021, 57, 984.
Lukyanov, B. S.; Lukyanova, M. B. Chem. Heterocycl. Compd. 2005, 41, 281.
Wizinger, R.; Wenning, H. Helv. Chim. Acta 1940, 43, 247.
Kortekaas, L.; Browne, W. R. Chem. Soc. Rev. 2019, 48, 3406.
Pugachev, A. D.; Mukhanov, E. L.; Ozhogin, I. V.; Kozlenko, A. S.; Metelitsa, A. V.; Lukyanov, B. S. Chem. Heterocycl. Compd. 2021, 57, 122.
Laptev, A. V.; Lukin, A. Y.; Belikov, N. E.; Zvezdin, K. V.; Demina, O. V.; Barachevsky, V. A.; Varfolomeev, S. D.; Khodonov, A. A.; Shvets, V. I. Russ. Chem. Bull. 2014, 63, 2026.
Nikolaeva, O. G.; Metelitsa, A. V.; Cheprasov, A. S.; Karlutova, O. Y.; Starikov, A. G.; Dubonosov, A. D.; Bren', V. A.; Minkin, V. I. Russ. Chem. Bull. 2016, 65, 944.
Frolova, L. A.; Rezvanova, A. A.; Lukyanov, B. S.; Sanina, N. A.; Troshin, P. A.; Aldoshin, S. M. J. Mater. Chem. C 2015, 3, 11675.
Bénard, S.; Rivière, E.; Yu, P.; Nakatani, K.; Delouis, J. F. Chem. Mater. 2001, 13, 159.
Bazzan, I.; Bolle, P.; Oms, O.; Salmi-Mani, H.; Aubry-Barroca, N.; Dolbecq, A.; Serier-Brault, H.; Dessapt, R.; Philippe R.; Mialane, P. J. Mater. Chem. C 2017, 5, 6343.
Kida, N.; Hikita, M.; Kashima, I.; Okubo, M.; Itoi, M.; Enomoto, M.; Kato, K.; Takata, M.; Kojima, N. J. Am. Chem. Soc. 2009, 131, 212.
Pugachev, A. D.; Ozhogin, I. V.; Lukyanova, M. B.; Lukyanov, B. S.; Rostovtseva, I. A.; Dorogan, I. V.; Makarova, N. I.; Tkachev, V. V.; Metelitsa, A. V.; Aldoshin, S. M. Spectrochim. Acta, Part A 2020, 230, 118041.
Khalanskiy, K. N.; Alekseenko, Y. S.; Lukyanov, B. S.; Borodkin, G. S.; Bezuglyi, S. O. Chem. Heterocycl. Compd. 2012, 48, 1090.
Funasako, Y.; Miyazaki, H.; Sasaki, T.; Goshima, K.; Inokuchi, M. J. Phys. Chem. B 2020, 124, 7251.
Pugachev, A. D.; Lukyanova, M. B.; Lukyanov, B. S.; Ozhogin, I. V.; Kozlenko, A. S.; Rostovtseva, I. A.; Makarova, N. I.; Tkachev, V. V.; Aksenov, N. A. J. Mol. Struct. 2019, 1178, 590.
Kozlenko, A. S.; Makarova, N. I.; Ozhogin, I. V.; Pugachev, A. D.; Lukyanova, M. B.; Rostovtseva, I. A.; Borodkin, G. S.; Stankevich, N. V.; Metelitsa, A. V.; Lukyanov, B. S. Mendeleev Commun. 2021, 31, 403.
Pugachev, A. D.; Ozhogin, I. V.; Makarova, N. I.; Rostovtseva, I. A.; Lukyanova, M. B.; Kozlenko, A. S.; Borodkin, G. S.; Tkachev, V. V.; El-Sewify, I. M.; Dorogan, I. V.; Metelitsa, A. V.; Aldoshin, S. M.; Lukyanov, B. S. Dyes Pigm. 2022, 199, 110043.
Pugachev, A. D.; Ozhogin, I. V.; Lukyanova, M. B.; Lukyanov, B. S.; Kozlenko, A. S.; Rostovtseva, I. A.; Makarova, N. I.; Tkachev, V. V.; Aldoshin, S. M.; Metelitsa, A. V. J. Mol. Struct. 2021, 1229, 129615.
Lukyanova, M. B.; Tkachev, V. V.; Lukyanov, B. S.; Pugachev, A. D.; Ozhogin, I. V.; Komissarova, O. A.; Aldoshin, S. M.; Minkin, V. I. J. Struct. Chem. 2018, 59, 565.
Balmond, E. I.; Tautges, B. K.; Faulkner, A. L.; Or, V. W.; Hodur, B. M.; Shaw, J. T.; Louie, A. Y. J. Org. Chem. 2016, 81, 8744.
Tkachev, V. V.; Lukyanova, M. B.; Lukyanov, B. S.; Pugachev, A. D.; Aldoshin, S. M.; Minkin, V. I. J. Struct. Chem. 2016, 57, 1270.
Allen, F. H.; Watson, D. G.; Brammer, L.; Orpen, A. G.; Taylor, R. In International Tables for Crystallography. Volume C: Mathematical, Physical and Chemical Tables; Prince, E., Ed.; Springer: Dordrecht, 2006, p. 803.
Jelsch, C.; Ejsmont, K.; Huder, L. IUCrJ 2014, 1, 119.
Seiler, V. K.; Callebaut, K.; Robeyns, K.; Tumanov, N.; Wouters, J.; Champagne, B.; Leyssens, T. CrystEngComm 2018, 20, 3318.
Brieke, C.; Heckel, A. Chem.–Eur. J. 2013, 19, 15726.
Hammarson, M.; Nilsson, J. R.; Li, S.; Beke-Somfai, T.; Andréasson, J. J. Phys. Chem. B 2013, 117, 13561.
Stafforst, T.; Hilvert, D. Chem. Commun. 2009, 3, 287.
Cho, Y. J.; Lee, S. H.; Bae, J. W.; Pyun, H.-J.; Yoon, C. M. Tetrahedron Lett. 2000, 41, 3915.
Luk'yanova, M. B.; Pugachev, A. D.; Tkachev, V. V.; Luk'yanov, B. S.; Shilov, G. V.; Kozlenko, A. S.; Rostovtseva, I. A.; Minkin, V. I.; Aldoshin, S. M. Dokl. Chem. 2018, 482, 220.
Pugachev, A. D.; Lukyanova, M. B.; Lukyanov, B. S.; Ozhogin, I. V.; Kozlenko, A. S.; Tkachev, V. V.; Chepurnoi, P. B.; Shilov, G. V.; Minkin, V. I.; Aldosin, S. M. Dokl. Chem. 2020, 492, 76.
Tian, W.; Tian, J. Dyes Pigm. 2014, 105, 66.
Kim, D.; Zhang, Z.; Xu, K. J. Am. Chem. Soc. 2017, 139, 9447.
Sheng, Y.; Leszczynski, J.; Garcia, A. A.; Rosario, R.; Gust, D.; Springer, J. J. Phys. Chem. B 2004, 108, 16233.
Yang, W.; Parr, R. G.; Pucci, R. J. Chem. Phys. 1984, 81, 2862.
Hirshfeld, F. L. Theor. Chim. Acta 1977, 44, 129.
Wang, B.; Rong, C.; Chattaraj, P. K.; Liu, S. Theor. Chem. Acc. 2019, 138, 123.
Sheldrick, G. M. SHELXTL v. 6.14, Structure Determination Software Suite; Bruker AXS: Madison, 2000.
Macrae, C. F.; Sovago, I.; Cottrell, S. J.; Galek, P. T. A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G. P.; Stevens, J. S.; Towler, M.; Wood, P. A. J. Appl. Crystallogr. 2020, 53, 226.
Spackman, P. R.; Turner, M. J.; McKinnon, J. J.; Wolff, S. K.; Grimwood, D. J.; Jayatilaka, D.; Spackman, M. A. J. Appl. Crystallogr. 2021, 54, 1006.
Neese, F.; Wennmohs, F.; Becker, U.; Riplinger, C. J. Chem. Phys. 2020, 152, 224108.
Neese, F. Wiley Interdiscip. Rev.: Comput. Mol. Sci. 2012, 2, 73.
Becke, A. D. J. Chem. Phys. 1993, 98, 5648.
Hehre, W. J.; Random, L.; Schleyer, P. v. R.; Pople, J. A. Ab Initio Molecular Orbital Theory; Wiley: New York, 1986.
Lu, T.; Chen, F. J. Comput. Chem. 2012, 33, 580.
Chemcraft – Graphical Software for Visualization of Quantum Chemistry Computations. https://www.chemcraftprog.com
This study was supported by a grant from the Ministry of Science and Higher Education of the Russian Federation within the framework of State Contract for scientific research (Southern Federal University, No. 0852-2020-00-19). The X-ray structural analysis was performed in accordance with State Contract, registry No. АААА-А19-119092390076-7 (V. V. Tkachev, S. M. Aldoshin).
1Н and 13С NMR spectra were acquired at the Molecular Spectroscopy Collective Use Center of the Southern Federal University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2022, 58(12), 712–720
Supplementary Information
ESM 1
(PDF 801 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kozlenko, A.S., Pugachev, A.D., Ozhogin, I.V. et al. Synthesis and structural characterization of new spiropyran containing conjugated vinyl-3Н-indolium moiety and its hydrolysis product. Chem Heterocycl Comp 58, 712–720 (2022). https://doi.org/10.1007/s10593-023-03147-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-023-03147-5