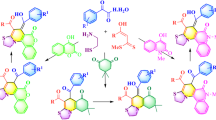
In this work, we developed a straightforward and convenient method for the synthesis of substituted pyrazoloquinolines – highly desirable compounds for many high-tech and biomedical applications. On the basis of intramolecular cyclization of 5-(N-aryl)aminopyrazoles in the presence of DMF diethyl acetal and POCl3, a series of differently substituted pyrazoloquinolines were synthesized. The synthetic procedure was thoroughly optimized taking into account key factors such as the type of formylating agent (DMF vs. DMF diethyl diacetal), reaction temperature, and stoichiometry of the reagents used. Because of its versatility, simplicity, and ease in purification of the obtained products, this synthetic approach provides a valuable guideline in the rational design of pyrazoloquinoline-based functional materials.


Similar content being viewed by others
References
Michaelis, A. Justus Liebigs Ann. Chem. 1911, 385, 1.
Musierowicz, A.; Niementowski, S.; Tomasik, Z. Rocz. Chem. 1928, 8, 325.
Wolfrum, G.; Putter, R.; Hanke, H.-G.; Menzel, K.-H. Patent US3234142.
Stein, R. G.; Biel, J. H.; Singh, T. J. Med. Chem. 1970, 13, 153.
Crenshaw, R. R.; Luke, G. M.; Siminoff, P. J. Med. Chem. 1976, 19, 262.
Rádl, S.; Zikán, V.; Šmejkal, F. Czesk. Farm. 1984, 33, 429.
Jitender, D. G.; Poornachandra, Y.; Reddy, K. R.; Kumar, N. R.; Ravikumar, N.; Swaroop, D. K.; Ranjithreddy, P.; Kumar, S. G.; Nanubolu, B. J.; Kumar, C. G.; Narsaiah, B. Eur. J. Med. Chem. 2017, 130, 223.
Brack, A. Liebigs Ann. Chem. 1965, 681, 105.
Parusel, A. B. J.; Rechthaler, K.; Piorun, D.; Danel, A.; Khatchatryan, K.; Rotkiewicz, K.; Köhler, G. J. Fluoresc. 1998, 8, 375.
Rechthaler, K.; Rotkiewicz, K.; Danel, A.; Tomasik, P.; Khatchatryan, K.; Kohler, G. J. Fluoresc. 1997, 7, 301.
Rurack, K.; Danel, A.; Rotkiewicz, K.; Grabka, D.; Spieles, M.; Rettig, W. Org. Lett. 2002, 4, 4647.
Mac, M.; Uchacz, T.; Danel, A.; Musiolik, H. J. Fluoresc. 2013, 23, 1207.
Danel, A.; Kolbus, A.; Grabka, D.; Kucharek, M.; Pokładko- Kowar, M. Dyes Pigm. 2021, 195, 109713.
Chen, C.-H.; Wu, F.-I.; Shu, C.-F.; Chien, C.-H; Tao, Y.-T. J. Mater. Chem. 2004, 14, 1585.
Wan, W.; Wang, H.; Lin, H.; Wang, J.; Jiang, Y.; Jiang, H.; Zhu, S.; Wang, Z.; Hao, J. Dyes Pigm. 2015, 121, 138.
He, Z.; Milburn, G. H. W.; Danel, A.; Puchała, A.; Tomasik, P.; Rasała, D. J. Mater. Chem. 1997, 7, 2323.
Danel, A. PhD Thesis; Krakow, 1996.
Tomasik, P.; Tomasik, D.; Abramovitch, R. J. Heterocycl. Chem. 1983, 20, 1539.
Marjani, A. P.; Khalafy, J.; Salami, F.; Mohammadlou, M. Synthesis 2015, 47, 1656.
Ezzati, M.; Khalafy, J.; Marjani, A. P.; Prager, R. H. Tetrahedron 2017, 73, 6587.
Marjani, A. P.; Khalafy, J.; Akbarzadeh, S. Green Process. Synth. 2019, 8, 533.
Yadav, P.; Awasthi, A.; Gokulnath, S.; Tiwari, D. K. J. Org. Chem. 2021, 86, 2658.
Danel, A.; Gondek, E.; Kucharek, M.; Szlachcic, P.; Gut, A. Molecules 2022, 27, 2775.
Purnaprajna, V.; Seshadri, S. Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem. 1976, 14B, 971.
Kucharek, M.; Danel, A. Chem. Heterocycl. Compd. 2021, 57, 633.
Szlachcic, P.; Kucharek, M.; Jarosz, B.; Danel, A.; Stadnicka, K. J. Heterocycl. Chem. 2017, 54, 1729.
Całus, S.; Gondek, E.; Pokladko, M.; Kulig, E.; Jarosz, B.; Kityk, A. V. Spectrochim. Acta, Part A 2007, 67, 1007.
Gondek, E.; Danel, A.; Nizioł, J.; Armatys, P.; Kityk, I. V.; Szlachcic, P.; Karelus, M.; Uchacz, T.; Chwast, J.; Lakshminarayana, G. J. Lumin. 2010, 130, 2093.
Mac, M.; Uchacz, T.; Andrzejak, M.; Danel, A.; Szlachcic, P. J. Photochem. Photobiol., A 2007, 187, 78.
Gondek, E.; Nosidlak, N.; Kulig, E.; Danel, A.; Al Zayed, N.; Kityk, I. V.; Karasinki, P. J. Mater. Sci.: Mater. Electron. 2013, 24, 613.
Brack, A. BE Patent 617180.
Shiba, S. A.; Harb, N. M. S.; Hassan, M. A.; El-Kassaby, M. A.; Abou-El-Regal, M. M. K. Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem. 1996, 35B, 426.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2022, 58(11), 639–645
Supplementary Information
ESM 1
(PDF 3117 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kucharek, M., Danel, A. & Gut, A. A simple and efficient synthesis of 1,3,6-trisubstituted pyrazoloquinolines via intramolecular cyclization – optimization studies. Chem Heterocycl Comp 58, 639–645 (2022). https://doi.org/10.1007/s10593-022-03127-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-022-03127-1