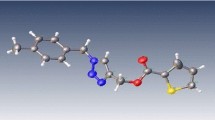
A novel series of 3,5-disubstituted isoxazoles bearing kojic acid moiety were synthesized via Cu(I)-catalyzed 1,3-dipolar cycloaddition reaction of terminal alkynes with nitrile oxide formed in situ from the corresponding hydroximoyl chloride. Structures of the synthesized compounds were characterized using spectroscopic analysis data. Antimicrobial activity of the isoxazole conjugates was determined as MIC values by broth microdilution method against different bacteria: Staphylococcus aureus, Bacillus subtilis, Escherichia coli, Salmonella typhi, and fungi Candida kefyr. The obtained results revealed notable antibacterial and antifungal activities of the compounds.
Similar content being viewed by others
References
Baraldi, P. G.; Barco, A.; Benetti, S.; Pollini, G. P.; Simoni, D. Synthesis 1987, 857.
Talley, J. J.; Brown, D. L.; Carter, J. S.; Graneto, M. J.; Koboldt, C. M.; Masferrer, J. L.; Perkins, W. E.; Rogers, R. S.; Shaffer, A. F.; Zhang, Y. Y.; Zweifel, B. S.; Seibert, K. J. Med. Chem. 2000, 43, 775.
Magar, B. K.; Bhosale, V. N.; Berad, B. N. Chem. Sin. 2011, 2, 147.
Basha, S. S.; Divya, K.; Padmaja, A.; Padmavathi, V. Res. Chem. Intermed. 2015, 41, 10067.
Kapoor, A.; Beniwal, R. Pharm. Lett. 2016, 8, 127.
Kankala, S.; Kankala, R. K.; Gundepaka, P.; Thota, N.; Nerella, S.; Gangula, M. R.; Guguloth, H.; Kagga, M.; Vadde, R.; Vasam, C. S. Bioorg. Med. Chem. Let. 2013, 23, 1306.
Rajanarendar, E.; Rama Krishna, S.; Nagaraju, D.; Govardhan Reddy, K.; Kishore, B.; Reddy, Y. N. Bioorg. Med. Chem. Lett. 2015, 25, 1630.
Hirpara, K.; Patel, S.; Joshi, A.; Parekh, H. Indian J. Heterocycl. Chem. 2004, 13, 221.
Tzanetou, E.; Liekens, S.; Kasiotis, K. M.; Melagraki, G.; Afantitis, A.; Fokialakis, N.; Haroutounian, S. A. Eur. J. Med. Chem. 2014, 81, 139.
Garg, H. G.; Singh, P. P. J. Med. Chem. 1970, 13, 1250.
Heasley, B. Angew. Chem., Int. Ed. 2011, 50, 8474.
Bode, J. W.; Carreira, E. M. Org. Lett. 2001, 3, 1587.
Yashiro, A.; Nishida, Y.; Kobayashi, K.; Ohno, M. Synlett 2000, 361.
Müller, I.; Jäger, V. Tetrahedron Lett. 1982, 23, 4777.
Lee, S. Y.; Lee, B. S.; Lee, C.-W.; Oh, D. Y. J. Org. Chem. 2000, 65, 256.
Grünanger, P.; Vita-Finzi, P. In Chemistry of Heterocyclic Compounds: Isoxazoles; Wiley Interscience: New York, 1991, Vol. 49, p. 1.
Nitta, M.; Kobayshi, T. J. Chem. Soc., Chem. Commun. 1982, 15, 877.
Eicher, T.; Hauptmann, S.; Speicher, A. The Chemistry of Heterocycles: Structure, Reactions, Synthesis, and Applications; Wiley-VCH: Weinheim, 2003, 2nd ed., p. 138.
Cuadrado, P.; González-Nogal, A. M.; Valero, R. Tetrahedron 2002, 58, 4975.
Pinho e Melo, T. M. V. D. Curr. Org. Chem. 2005, 9, 925.
Bondarenko, O. B.; Zyk, N. V. Chem. Heterocycl. Compd. 2020, 56, 694. [Khim. Geterotsikl. Soedin. 2020, 56, 694.]
Huisgen, R. In 1,3-Dipolar Cycloaddition Chemistry; Padwa, A., Ed.; Wiley: New York, 1984, Vol. 1, p. 1.
Xu, J.; Hamme, A. T. Synlett 2008, 919.
Kozikowski, A. P.; Adamczyk, M. J. Org. Chem. 1983, 48, 366.
Grundmann, C.; Richter, R. J. Org. Chem. 1968, 33, 476.
Himo, F.; Lovell, T.; Hilgraf, R.; Rostovtsev, V. V.; Noodleman, L.; Sharpless, K. B.; Fokin, V. V. J. Am. Chem. Soc. 2005, 127, 210.
Yabuta, T. J. Chem. Soc., Trans. 1924, 125, 575.
Mohamad, R.; Mohamed, M. S.; Suhaili, N.; Salleh, M. M.; Ariff, A. B. Biotechnol. Mol. Biol. Rev. 2010, 5, 24.
Mitani, H.; Koshiishi, I.; Sumita, T.; Imanari, T. Eur. J. Pharmacol. 2001, 411, 169.
Higa, Y.; Kawabe, M.; Nabae, K.; Toda, Y.; Kitamoto, S.; Hara, T.; Tanaka, N.; Kariya, K.; Takahashi, M. J. Toxicol. Sci. 2007, 32, 143.
Aytemir, M. D.; Özçelik, B. Eur. J. Med. Chem. 2010, 45, 4089.
Sansho Seiyaku Co. JP Patent 80154916.
Uher, M.; Konečný, V.; Rajniaková, O. Chem. Pap. 1994, 48, 282.
Khoeiniha, R.; Olyaei, A.; Saraei, M. J. Heterocycl. Chem. 2017, 54, 1746.
Saraei, M.; Eftekhari-Sis, B.; Faramarzi, M.; Hossienzadeh, R. J. Heterocycl. Chem. 2014, 51, 1500.
Masateru, M.; Shone, R. L. US Patent 4705871A.
Williams, H. W. R. Can. J. Chem. 1976, 54, 3377.
Bransová, J.; Uher, M.; Brtko, J. Chem. Pap. 1993, 47, 316.
Tehrani, M. B.; Emani, P.; Rezaei, Z.; Khoshneviszadeh, M.; Ebrahimi, M.; Edraki, N.; Mahdavi, M.; Larijani, B.; Ranjbar, S.; Foroumadi, A.; Khoshneviszadeh, M. J. Mol. Struct. 2019, 1176, 86.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2021, 57(7/8), 823–827
Supplementary Information
ESM 1
(PDF 5135 kb)
Rights and permissions
About this article
Cite this article
Khodabandlou, S., Saraei, M. Synthesis of novel isoxazole derivatives bearing kojic acid moiety and evaluation of their antimicrobial activity. Chem Heterocycl Comp 57, 823–827 (2021). https://doi.org/10.1007/s10593-021-02986-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-021-02986-4