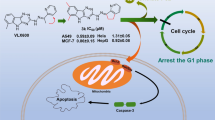
Two series of fluorinated camptothecin derivatives had been designed and synthesized via the Mannich reaction. The hexacyclic fluorinated camptothecin derivatives were found to be more active. (9S,10S)-2-Cyclopropyl-9-ethyl-10-fluoro-9-hydroxy-2,3,9,10,12,15-hexahydro-1H,13H-[1,3]oxazino[5,6-f]pyrano[3',4':6,7]indolizino[1,2-b]quinolin-13-one showed potent antitumor activity against A549 and T24 cell lines with effective topoisomerase I inhibitory activity.



Similar content being viewed by others
References
Wang, J.; Sánchez-Roselló, M.; Aceña, J. L.; del Pozo, C.; Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev. 2014, 114, 2432.
Richardson, P. Expert Opin. Drug Discovery 2016, 11, 983.
Yerien, D. E.; Bonesi, S.; Postigo, A. Org. Biomol. Chem. 2016, 14, 8398.
Wang, B.-C.; Wang, L.-J.; Jiang, B.; Wang, S.-Y.; Wu, N.; Li, X.-Q.; Shi, D.-Y. Mini-Rev. Med. Chem. 2017, 17, 683.
Mei, H.; Han, J.; Klika, K. D.; Izawa, K.; Sato, T.; Meanwell, N. A.; Soloshonok, V. A. Eur. J. Med. Chem. 2020, 186, 111826.
Kuznecovs, J.; Baskevics, V.; Orlova, N.; Belyakov, S.; Domračeva, I.; Vorona, M.; Veinberg, G. Chem. Heterocycl. Compd. 2020, 56, 1297. [Khim. Geterotsikl. Soedin. 2020, 56, 1297.]
Zhuang, C.; Wu, Y.; Zhang, W.; Miao, Z. In Natural Products in Clinical Trials; Atta-ur-Rahman; Anjum, S.; El-Seedi, H. R., Eds.; Bentham Science Publishers: Sharjah, 2020, Vol. 2, p. 123.
Müller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
Gillis, E. P.; Eastman, K. J.; Hill, M. D.; Donnelly, D. J.; Meanwell, N. A. J. Med. Chem. 2015, 58, 8315.
Meanwell, N. A. J. Med. Chem. 2018, 61, 5822.
Miao, Z.; Zhu, L.; Dong, G.; Zhuang, C.; Wu, Y.; Wang, S.; Guo, Z.; Liu, Y.; Wu, S.; Zhu, S.; Fang, K.; Yao, J.; Li, J.; Sheng, C.; Zhang, W. J. Med. Chem. 2013, 56, 7902.
Wu, Y.; Zhu, L.; Sheng, C.; Yao, J.; Miao, Z.; Zhang, W.; Wei, Y.; Shi, R. J. Fluorine Chem. 2014, 157, 48.
Ma, H.; Chen, B.; Wang, Y.; Wang, C.; Yao, J.; Zhang, W.; Miao, Z. Chem. Biodiversity 2020, 17, e2000068.
Yang, C.-J.; Song, Z.-L.; Goto, M.; Hsu, P.-L.; Zhang, X.-S.; Yang, Q.-R.; Liu, Y.-Q.; Wang, M.-J.; Morris-Natschke, S. L.; Shang, X.-F.; Lee, K.-H. Bioorg. Med. Chem. Lett. 2017, 27, 4694.
Biersack, B.; Ahmed, K.; Padhye, S.; Schobert, R. Expert Opin. Drug Discovery 2018, 13, 39.
Roman, G. Eur. J. Med. Chem. 2015, 89, 743.
Lukashenko, A. V.; Osipov, D. V.; Osyanin, V. А.; Klimochkin, Yu. N. Chem. Heterocyclic Compd. 2020, 56, 529. [Khim. Geterotsikl. Soedin. 2020, 56, 529.]
Xu, T.; Zheng, Z.; Guo, Y.; Bai, L.-P. Eur. J. Med. Chem. 2020, 205, 112663.
Bracher, F.; Tremmel, T. Arch. Pharm. 2017, 350, e1600236.
The PyMOL Molecular Graphics System, Version 2.5; Schrödinger, LLC. https://www.schrodinger.com/products/pymol
AutoDock, Version 4.2.6; The Scripps Research Institute. http://autodock.scripps.edu/
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2021, 57(6), 704–708
Supplementary Information
ESM 1
(PDF 1696 kb)
Rights and permissions
About this article
Cite this article
Wang, C., Wang, Y., Ma, H. et al. Synthesis via the Mannich Reaction and Biological Activity of Novel Fluorinated Camptothecin Derivatives. Chem Heterocycl Comp 57, 704–708 (2021). https://doi.org/10.1007/s10593-021-02971-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-021-02971-x