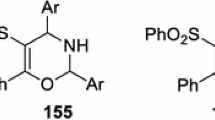
Diversity-oriented synthesis embodied in a strategy of pairing vinyl sulfonamide moiety with other functional groups resulted in an array of skeletally diverse 5–8-membered sultams (cyclic sulfonamides) in the fewest possible steps. In particular, this strategy allows utilization of a number of cyclization protocols including intramolecular Michael, Diels–Alder, Heck, Pauson–Khand, Baylis–Hillman reactions, ring-closing metathesis (RCM) and others.

Similar content being viewed by others
References
Okwuchukwu, P. M.; Bandyopadhyay, D. Mini-Rev. Med. Chem. 2020, 20, 2193.
Rassadin, V. A.; Grosheva, D. S.; Tomashevskii, A. A.; Sokolov, V. V. Chem. Heterocycl. Compd. 2013, 49, 39. [Khim. Geterotsikl. Soedin. 2013, 47.]
Popova, M. V.; Dobrydnev, A. V. Chem. Heterocycl. Compd. 2017, 53, 492. [Khim. Geterotsikl. Soedin. 2017, 53, 492.]
Debnath, S.; Mondal, S. Eur. J. Org. Chem. 2018, 933.
Grygorenko, O. O.; Vashchenko, B. V.; Blahun, O. P.; Zhersh, S. Eur. J. Org. Chem. 2020, 5787.
Dobrydnev, A. V.; Marco-Contelles, J. Eur. J. Org. Chem. 2021, 1229.
Tong, K.; Tu, J.; Qi, X.; Wang, M.; Wang, Y.; Fu, H.; Pittman, C. U., Jr.; Zhou, A. Tetrahedron 2013, 69, 2369.
Mulet, C.; Escolano, M.; Llopis, S.; Sanz, S.; Ramírez de Arellano, C.; Sánchez-Roselló, M.; Fustero, S.; del Pozo, C. Adv. Synth. Catal. 2018, 360, 2885.
Zhou, A.; Rayabarapu, D.; Hanson, P. R. Org. Lett. 2009, 11, 531.
Zhou, A.; Hanson, P. R. Org. Lett. 2008, 10, 2951.
Ji, T.; Wang, Y.; Wang, M.; Niu, B.; Xie, P.; Pittman, C. U., Jr.; Zhou, A. ACS Comb. Sci. 2013, 15, 595.
Wang, M.; Wang, Y.; Qi, X.; Xia, G.; Tong, K.; Tu, J.; Pittman, C. U., Jr.; Zhou, A. Org. Lett. 2012, 14, 3700.
Rolfe, A.; Samarakoon, T. B.; Hanson, P. R. Org. Lett. 2010, 12, 1216.
Charrier, N.; Clarke, B.; Demont, E.; Dingwall, C.; Dunsdon, R.; Hawkins, J.; Hubbard, J.; Hussain, I.; Maile, G.; Matico, R.; Mosley, J.; Naylor, A.; O'Brien, A.; Redshaw, S.; Rowland, P.; Soleil, V.; Smith, K. J.; Sweitzer, S.; Theobald, P.; Vesey, D.; Walter, D. S.; Wayne, G. Bioorg. Med. Chem. Lett. 2009, 19, 3669.
Grubbs, R. H. Angew. Chem., Int. Ed. 2006, 45, 3760.
Hanessian, S.; Sailes, H.; Therrien, E. Tetrahedron 2003, 59, 7047.
Mondal, S.; Debnath, S. Synthesis 2014, 368.
Bungard, C. J.; Williams, P. D.; Schulz, J.; Wiscount, C. M.; Holloway, M. K.; Loughran, H. M.; Manikowski, J. J.; Su, H.-P.; Bennett, D. J.; Chang, L.; Chu, X.-J.; Crespo, A.; Dwyer, M. P.; Keertikar, K.; Morriello, G. J.; Stamford, A. W.; Waddell, S. T.; Zhong, B.; Hu, B.; Ji, T.; Diamond, T. L.; Bahnck-Teets, C.; Carroll, S. S.; Fay, J. F.; Min, X.; Morris, W.; Ballard, J. E.; Miller, M. D.; McCauley, J. A. ACS Med. Chem. Lett. 2017, 8, 1292.
Ricker, J. D.; Geary, L. M. Top. Catal. 2017, 60, 609.
Astruc, D. Anal. Bioanal. Chem. 2011, 399, 1811.
Ueda, M.; Miyabe, H.; Nishimura, A.; Miyata, O.; Takemoto, Y.; Naito, T. Org. Lett. 2003, 5, 3835.
Rogachev, V. O.; Filimonov, V. D.; Fröhlich, R.; Kataeva, O.; Metz, P. Heterocycles 2006, 67, 589.
Plietker, B.; Seng, D.; Fröhlich, R.; Metz, P. Tetrahedron 2000, 56, 873.
Jeon, K. O.; Rayabarapu, D.; Rolfe, A.; Volp, K.; Omar, I.; Hanson, P. R. Tetrahedron 2009, 65, 4992.
Kelleher, S.; Muldoon, J.; Müller-Bunz, H.; Evans, P. Tetrahedron Lett. 2007, 48, 4733.
Rogatchov, V. O.; Bernsmann, H.; Schwab, P.; Fröhlich, R.; Wibbeling, B.; Metz, P. Tetrahedron Lett. 2002, 43, 4753.
Liang, H.; Ciufolini, M. A. Org. Lett. 2010, 12, 1760.
Liang, H.; Ciufolini, M. A. Chem.–Eur. J. 2010, 16, 13262.
Liang, H.; Ciufolini, M. A. Tetrahedron 2010, 66, 5884.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Yulian M. Volovenko on the occasion of his 71st birthday
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2021, 57(6), 630–632
Rights and permissions
About this article
Cite this article
Dodrydnev, A.V., Omelian, T.V.
 Ring-closure reactions of functionalized vinyl sulfonamides (microreview).
Chem Heterocycl Comp 57, 630–632 (2021). https://doi.org/10.1007/s10593-021-02960-0
Ring-closure reactions of functionalized vinyl sulfonamides (microreview).
Chem Heterocycl Comp 57, 630–632 (2021). https://doi.org/10.1007/s10593-021-02960-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-021-02960-0