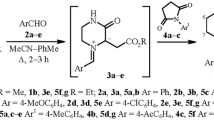
The reaction of aryl isothiocyanates with nonstabilized azomethine ylides generated in situ by various methods was studied. It was established that the use of N-(methoxymethyl)-N-(trimethylsilylmethyl)benzylamine in the presence of trifluoroacetic acid in the role of a catalyst led to the formation of (Z)-N-aryl-3-benzylthiazolidin-5-imines in 22–47% yields.

Similar content being viewed by others
References
(a) Nájera, C.; Sansano, J. M. Curr. Org. Chem. 2003, 7, 1105. (b) Coldham, I.; Hufton, R. Chem. Rev. 2005, 105, 2765. (c) Pandey, G.; Banerjee, P.; Gadre, S. R. Chem. Rev. 2006, 106, 4484.
(a) Padwa, A.; Dent, W. J. Org. Chem. 1987, 52, 235. (b) Beugelmans, R.; Chastanet, J.; Roussi, G. Heterocycles 1987, 26, 3197. (c) Chastanet, J.; Roussi, G. J. Org. Chem. 1988, 53, 3808. (d) Mykhaylychenko, S. S.; Siryi, S. A.; Pikun, N. V.; Shermolovich, Y. G. Chem. Heterocycl. Compd. 2015, 51, 861. [Khim. Geterotsikl. Soedin. 2015, 51, 861.] (e) Meyer, A. G.; Ryan, J. H. Molecules 2016, 21, 935. e Izquierdo, C.; Esteban, F.; Ruano, J. L. G.; Fraile, A.; Alemán, J. Org. Lett. 2016, 18, 92. f Laha, J. K.; Jethava, K. P.; Tummalapalli, K. S. S.; Sharma, S. Eur. J. Org. Chem. 2017, 4617. g Beuvin, M.; Manneveau, M.; Diab, S.; Picard, B.; Sanselme, M.; Piettre, S. R.; Legros, J.; Chataigner, I. Tetrahedron Lett. 2018, 59, 4487.
(a) Korotaev, V. Y.; Barkov, A. Y.; Moshkin, V. S.; Matochkina, E. G.; Kodess, M. I.; Sosnovskikh, V. Y. Tetrahedron 2013, 69, 8602. (b) Sosnovskikh, V. Y.; Kornev, M. Y.; Moshkin, V. S.; Buev, E. M. Tetrahedron 2014, 70, 9253. (c) Buev, E. M.; Moshkin, V. S.; Sosnovskikh, V. Ya. Chem. Heterocycl. Compd. 2017, 53, 167. [Khim. Geterotsikl. Soedin. 2017, 53, 167.] (d) Gorbunova, E. V.; Buev, E. M.; Moshkin, V. S.; Sosnovskikh, V. Y. Synlett 2020, 343.
Buev, E. M.; Moshkin, V. S.; Sosnovskikh, V. Y. Tetrahedron Lett. 2019, 60, 773.
(a) Ozaki, S. Chem. Rev. 1972, 72, 457. (b) Mukerjee, A. K.; Ashare, R. Chem. Rev. 1991, 91, 1. (c) Kim, J. N.; Ryu, E. K. Tetrahedron Lett. 1993, 34, 8283. (d) Trofimov, B. A. J. Heterocycl. Chem. 1999, 36, 1469.
(a) Mali, P. R.; Khomane, N. B.; Sridhar, B.; Meshram, H. M.; Likhar, P. R. New J. Chem. 2018, 42, 13819.
This study received financial support from the Russian Foundation for Basic Research within the framework of scientific project No. 18-33-00042 and the State Assignment with the Ministry of Science and Higher Education of the Russian Federation (project FEUZ-2020-0052).
Analytical studies were performed at the Center for Collective Use “Spectroscopy and Analysis of Organic Compounds” on the basis of the Postovsky Institute of Organic Synthesis, Ural Branch of the Russian Academy of Sciences and at the Laboratory of Complex Investigations and Expert Evaluation of Organic Materials, Ural Federal University named after the first President of Russia B. N. Yeltsin.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2020, 56(9), 1222–1225
Electronic supplementary material
ESM 1
(PDF 947 kb)
Rights and permissions
About this article
Cite this article
Buev, E.M., Osintseva, A.P., Moshkin, V.S. et al. Reaction of N-benzyl azomethine ylide with aryl isothiocyanates: synthesis of (Z)-N-aryl-3-benzylthiazolidine-5-imines. Chem Heterocycl Comp 56, 1222–1225 (2020). https://doi.org/10.1007/s10593-020-02802-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-020-02802-5