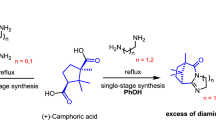
Several D-(+)-camphoric acid imides, not previously described in the literature, were synthesized with good yields. The synthesis of previously known imides was improved. The cytotoxicity and antiviral activity of all synthesized imides were studied.

Similar content being viewed by others
References
Salakhutdinov, N. F.; Volcho, K. P.; Yarovaya, O. I. Pure Appl. Chem. 2017, 89, 1105.
Sokolova, A. S.; Yarovaya, О. I.; Shtro, A. A.; Borisova, M. S.; Morozova, E. A.; Tolstikova, T. G.; Zarubaev, V. V; Salakhutdinov, N. F. Chem. Heterocycl. Compd. 2017, 53, 371. [Khim. Geterotsikl. Soedin. 2017, 53, 371.]
Sokolova, A. S.; Yarovaya, O. I.; Shernyukov, A. V.; Gatilov, Y. V.; Razumova, Y. V.; Zarubaev, V. V.; Tretiak, T. S.; Pokrovsky, A. G.; Kiselev, O. I.; Salakhutdinov, N. F. Eur. J. Med. Chem. 2015, 105, 263.
Sokolova, A. S.; Yarovaya, O. I.; Bormotov, N. I.; Shishkina, L. N.; Salakhutdinov, N. F. MedChemComm 2018, 9, 1746.
Chernyshov, V. V.; Yarovaya, O. I.; Fadeev, D. S.; Gatilov, Y. V.; Esaulkova, Y. L.; Muryleva, A. S.; Sinegubova, K. O.; Zarubaev, V. V.; Salakhutdinov, N. F. Mol. Diversity 2020, 24, 61.
Firstova, A. A.; Kofanov, E. R.; Zakshevskaya, V. M.; Kovaleva, M. I. Russ. J. Bioorg. Chem. 2019, 45, 204. [Bioorg. Khim. 2019, 45, 430.]
Xu, M.; Wang, Y.; Yang, F.; Wu, C.; Wang, Z.; Ye, B.; Jiang, X.; Zhao, Q.; Li, J.; Liu, Y.; Zhang, J.; Tian, G.; He, Y.; Shen, J.; Jiang, H. Eur. J. Med. Chem. 2018, 145, 74.
Köse, A.; Kaya, M.; Kishalı, N. H.; Akdemir, A.; Şahin, E.; Kara, Y.; Şanlı-Mohamed, G. Bioorg. Chem. 2020, 94, 103421.
Chen, H.; Song, W.; Huang, S. Chem. Heterocycl. Compd. 2006, 42, 726. [Khim. Geterotsikl. Soedin. 2006, 832.]
Novakov, I. A.; Sheikin, D. S.; Chapurkin, V. V.; Nawrozkij, M. B.; Babushkin, A. S.; Kuznetsov, Y. P.; Ruchko, E. A.; Kachala, V. V.; Maryshev, A. Yu.; Schols, D. Chem. Heterocycl. Compd. 2020, 56, 67. [Khim. Geterotsikl. Soedin. 2020, 56, 67.]
El-Etrawy, A. Sh.; Abdel-Rahman, A. A.-H. Chem. Heterocycl. Compd. 2010, 46, 1105. [Khim. Geterotsikl. Soedin. 2010, 1372.]
Komissarov, I. V.; Dulenko, V. I.; Voshchula, V. N.; Kharin, N. A.; Naletov, S. V.; Mamonov, A. B. Pharm. Chem. J. 1994, 28, 638. [Khim.-Farm. Zh. 1994, 28(9), 27.]
Takeda, K.; Tsuboyama, K.; Suzuki, A.; Ogura, H. Chem. Pharm. Bull. 1985, 33, 2545.
Wanner, K. T. J. Chem. Inf. Model. 1988, 321, 353.
Bell, K. H. Aust. J. Chem. 1981, 34, 665.
Rice, L. M.; Grogan, C. H. Hypotens. Agents 1957, 185.
Merzlikin, S. I.; Sal’nikova, S. I.; Yaremenko, F. G. Pharm. Chem. J. 2001, 35, 139.
Eagles, J. B.; Hitchcock, S. R. Tetrahedron: Asymmetry 2010, 21, 519.
Moloney, M. G.; Paul, D. R.; Thompson, R. M.; Wright, E. Tetrahedron: Asymmetry 1996, 7, 2551.
Hintermann, L.; Broggini, D.; Togni, A. Helv. Chim. Acta 2002, 85, 1597.
Boeckman, R. K., Jr.; Miller, Y.; Savage, D.; Summerton, J. E. Tetrahedron Lett. 2011, 52, 2243.
Kazemi, F.; Kiasat, A. R.; Mombaini, B. Synth. Commun. 2007, 37, 3219.
Kononova, A. A.; Sokolova, A. S.; Cheresiz, S. V.; Yarovaya, O. I.; Nikitina, R. A.; Chepurnov, A. A.; Pokrovsky, A. G.; Salakhutdinov, N. F. MedChemComm 2017, 8, 2233.
Gwon, Y.-D.; Strand, M.; Lindqvist, R.; Nilsson, E.; Saleeb, M.; Elofsson, M.; Överby, A. K.; Evander, M. Viruses 2020, 12, 351.
Singh, I. P.; Gupta, S.; Kumar, S. Med. Chem. 2020, 16, 4.
Saudi, M.; Zmurko, J.; Kaptein, S.; Rozenski, J.; Neyts, J.; Van Aerschot, A. Eur. J. Med. Chem. 2014, 87, 529.
Spizzichino, S.; Mattedi, G.; Lauder, K.; Valle, C.; Aouadi, W.; Canard, B.; Decroly, E.; Kaptein, S. J. F.; Neyts, J.; Graham, C.; Sule, Z.; Barlow, D. J.; Silvestri, R.; Castagnolo, D. ChemMedChem 2020, 15, 385.
Delang, L.; Li, C.; Tas, A.; Quérat, G.; Albulescu, I. C.; De Burghgraeve, T.; Segura Guerrero, N. A.; Gigante, A.; Piorkowski, G.; Decroly, E.; Jochmans, D.; Canard, B.; Snijder, E. J.; Pérez-Pérez, M. J.; van Hemert, M. J.; Coutard, B.; Leyssen, P.; Neyts, J. Sci. Rep. 2016, 6, 31819.
Lacroix, C.; Querol-Audí, J.; Roche, M.; Franco, D.; Froeyen, M.; Guerra, P.; Terme, T.; Vanelle, P.; Verdaguer, N.; Neyts, J.; Leyssen, P. J. Antimicrob. Chemother. 2014, 69, 2723.
Tănase, C. I.; Drăghici, C.; Hanganu, A.; Pintilie, L.; Maganu, M.; Volobueva, A.; Sinegubova, E.; Zarubaev, V. V.; Neyts, J.; Jochmans, D.; Slita, A. V. Molecules 2019, 24, 2446.
The authors would like to express their gratitude to the Collective Use Chemical Service Center of the Siberian Branch of Russian Academy of Sciences, for the obtained spectra and analytical data, as well as Dr. D. S. Fadeev for the obtained NMR spectra data. The authors also would like to thank Prof. J. Neyts, Dr. D. Jochmans, and C. Collard (Rega Institute for Medical Research, KU Leuven) for the obtained data on the antiviral activity.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information file containing 1H and 13C NMR spectra, IR spectra, and HRMS of all synthesized compounds is available at the journal website at http://link.springer.com/journal/10593.
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2020, 56(6), 763–768
Electronic supplementary material
ESM 1
(PDF 8296 kb)
Rights and permissions
About this article
Cite this article
Chernyshov, V.V., Yarovaya, O.I., Peshkov, R.Y. et al. Synthesis of cyclic D-(+)-camphoric acid imides and study of their antiviral activity. Chem Heterocycl Comp 56, 763–768 (2020). https://doi.org/10.1007/s10593-020-02728-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-020-02728-y