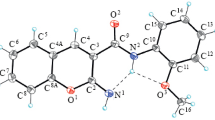
By the action of acetic acid on 2-morpholinochromanes, a series of chroman-2-ols were obtained in which the hydroxyl group is easily substituted by a primary or secondary amine or benzyl carbamate fragments. In the case of hydrazines and hydrazides of carboxylic acids, the corresponding hydrazones are isolated.


Similar content being viewed by others
References
(a) Ahmed, A. A.; Mohamed, A. El-H. H.; Karchesy, J.; Asakawa, Y. Phytochemistry2006, 67, 424. (b) Kwon, J.; Lee, H.; Ko, W.; Kim, D.-C.; Kim, K.-W.; Kwon, H. C.; Guo, Y.; Sohn, J. H.; Yim, J. H.; Kim, Y.-C.; Oh, H.; Lee, D. Tetrahedron2017, 73, 3905. (c) Boonsri, S.; Karalai, C.; Ponglimanont, C.; Chantrapromma, S.; Kanjana-opas, A. J. Nat. Prod. 2008, 71, 1173. (d) Song, J.; Manir, M. M.; Moon, S.-S. Chem. Biodiversity2009, 6, 1435. (e) Li, J.-T.; Fu, X.-L.; Tan, C.; Zeng, Y.; Wang, Q.; Zhao, P.-J. Molecules2011, 16, 686. (f) Stobiecki, M.; Popenda, M. Phytochemistry1994, 37, 1707.
(a) Wu, X.-N.; You, Z.-H.; Liu, Y.-K. Org. Biomol. Chem. 2018, 16, 6507. (b) Mori, T.; Akioka, Y.; Onodera, G.; Kimura, M. Molecules2014, 19, 9288. (c) Li, J.-Y.; Li, Z.-L.; Zhao, W.-W.; Liu, Y.-K.; Tong, Z.-P.; Tan, R. Org. Biomol. Chem. 2016, 14, 2444. (d) You, Z.-H.; Chen, Y.-H.; Tang, Y.; Liu, Y.-K. Org. Lett. 2018, 20, 6682. (e) Sun, X.-L.; Chen, Y.-H.; Zhu, D.-Y.; Zhang, Y.; Liu, Y.-K. Org. Lett. 2016, 18, 864. (f) You, Z.-H.; Chen, Y.-H.; Tang, Y.; Liu, Y.-K. Org. Lett. 2019, 21, 8358. (g) Chen, Y.-H.; Li, D.-H.; Liu, Y.-K. ACS Omega2018, 3, 16615. (h) Chen, Y.-H.; Lv, X.-J.; You, Z.-H.; Liu, Y.-K. Org. Lett. 2019, 21, 5556. (i) Mattalia, J.-M. R.; Attolini, M. M. ARKIVOC2013, (i), 101.
(a) Osipov, D. V.; Mel'nikova, I. V.; Klimochkin, Yu. N.; Osyanin, V. А. RF patent 2652119. (b) Lukashenko, A. V.; Osyanin, V. A.; Osipov, D. V.; Klimochkin, Yu. N. J. Org. Chem.2017, 82, 1517.
(a) Golfi, С. A.; Domenico, R. D.; Spinelli, S.; Gallico, L.; Fiocchi, L.; Lotto, A.; Menta, E.; Borghi, A.; Rosa, C. D.; Tognella, S. J. Med. Chem.1995, 38, 508. (b) Jurd, L. J. Heterocycl. Chem. 1996, 33, 1919. (c) Liu, Y.-K.; Li, Z.-L.; Li, J.-Y.; Feng, H.-X.; Tong, Z.-P. Org. Lett. 2015, 17, 2022. (d) Hewage, A. N. D. P; Yao, H.; Nammalwar, B.; Gnanasekaran, K. K.; Lovell, S.; Bunce, R. A.; Eshelman, K.; Phaniraj, S. M.; Lee, M. M.; Peterson, B. R.; Battaile, K. P.; Reitz, A. B.; Rivera, M. J. Am. Chem. Soc. 2019, 141, 8171. (e) Eggler, J. F.; Johnson, M. R.; Melvin, L. S., Jr. US Patent 4486428A. (f) Nakazawa, T.; Furukawa, H.; Torii, K.; Itabashi, K. Nippon Kagaku Kaishi1989, 244.
This study was supported by the Russian Science Foundation (grant 19-13-00421).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2020, 56(3), 311–319
Rights and permissions
About this article
Cite this article
Semenova, I.А., Korzhenko, K.S., Osipov, D.V. et al. Synthesis and reactions of chroman-2-ols and their benzo analogs with N-nucleophiles. Chem Heterocycl Comp 56, 311–319 (2020). https://doi.org/10.1007/s10593-020-02661-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-020-02661-0