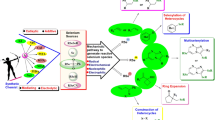
The photocyclization of stilbenes and their heterocyclic analogs has gained major importance in the synthesis of poly(hetero)aromatic compounds. This reaction generally leads to the formation of phenanthrene derivatives or their isosteric heterocyclic analogs. Over the last 15 years, examples of a new type of diarylethene reactions have been published, leading to bicyclic, instead of tricyclic aromatic products. These reactions are based on a sigmatropic hydrogen shift followed by the opening of one aromatic ring, and these transformations are applicable to the preparation of various aromatic compounds. Similar reactions are also highly relevant to the photostability of photochromic diarylethenes and terarylenes, thus the study of these compounds is important for the development of photo-controlled advanced materials and devices. In this review we provide the first analysis of scientific literature on this new type of diarylethene photoreactions.
Similar content being viewed by others
References
Handbook of Synthetic Photochemistry; Albini, A., Fagnoni, M., Eds.; Wiley-VCH: Weinheim, 2010.
Hoffmann, N. Chem. Rev. 2008, 108, 10523.
3. Bach, T.; Hehn, J. P. Angew. Chem., Int. Ed. 2011, 50, 1000.
Kärkäs, M. D.; Porco, J. A., Jr.; Stephenson, C. R. J. Chem. Rev. 2016, 116, 9683.
Bakulev, V. A. Russ. Chem. Rev. 1995, 64, 99. [Usp. Khim. 1995, 64, 107.]
Mallory, F. B.; Mallory, C. W. Photocyclization of Stilbenes and Related Molecules in Organic Reactions; Wiley: New York, 1984, Vol. 30, p. 1.
De Keukeleire, D.; He, S.-L. Chem. Rev. 1993, 93, 359.
Jørgensen, K. B. Molecules 2010, 15, 4334.
Parker, C. O.; Spoerri, P. E. Nature 1950, 166, 603.
Irie, M. Chem. Rev. 2000, 100, 1685.
Irie, M.; Fukaminato, T.; Matsuda, K.; Kobatake, S. Chem. Rev. 2014, 114, 12174.
Budyka, M. F. Russ. Chem. Rev. 2012, 81, 477. [Usp. Khim. 2012, 81, 477.]
Tian, H.; Yang, S.; Chem. Soc. Rev. 2004, 33, 85.
Nakamura, S.; Uchida, K.; Hatakeyama, M. Molecules 2013, 18, 5091.
Shirinian, V. Z.; Lonshakov, D. V.; Lvov, A. G.; Krayushkin, M. M. Russ. Chem. Rev. 2013, 82, 511. [Usp. Khim. 2013, 82, 511.]
Irie, M.; Lifka, T.; Uchida, K.; Kobatake, S.; Shindo, Y. Chem. Commun. 1999, 747.
Patel, P. D.; Mikhailov, I. A.; Belfield, K. D.; Masunov, A. E. Int. J. Quantum Chem. 2009, 109, 3711.
Herder, M.; Schmidt, B.; Grubert, L.; Pätzel, M.; Schwarz J.; Hecht, S. J. Am. Chem. Soc. 2015, 137, 2738.
Higashiuchi, K.; Matsuda, K.; Kobatake, S.; Yamada, T.; Kawai, T.; Irie, M. Bull. Chem. Soc. Jpn. 2000, 73, 2389.
Higashiuchi, K.; Matsuda, K.; Yamada, T.; Kawai, T.; Irie, M. Chem. Lett. 2000, 1358.
Shoji, H.; Kobatake, S. Chem. Commun. 2013, 49, 2362.
Shoji, H.; Kitagawa, D.; Kobatake, S. New J. Chem. 2014, 38, 933.
Tanaka, K.; Kitagawa, D.; Kobatake, S. Tetrahedron 2016, 72, 2364.
Nakagawa, H.; Kawai, S.; Nakashima, T.; Kawai, T. Org. Lett. 2009, 11, 1475.
Nakagawa, H.; Nakashima, T.; Kawai, T. Eur. J. Org. Chem. 2012, 4493.
Tietze, L. F. Chem. Rev. 1996, 96, 115.
Domino Reactions in Organic Synthesis; Tietze, L. F., Brasche, G., Gericke, K. M., Eds.; Wiley-VCH: Weinheim, 2006, p 337.
29. Ho, T.-I.; Wu, J.-Y.; Wang, S.-L. Angew. Chem., Int. Ed. 1999, 38, 2558.
Kellogg, R.; Groen, M. B;. Wynberg. H. J. Org. Chem. 1967, 32, 3093.
Wu, J.-Y.; Ho, J.-H.; Shih, S.-M.; Hsieh, T.-L.; Ho, T.-I. Org. Lett. 1999, 1, 1039.
Ho, T.-I.; Ho, J.-H.; Wu, J.-Y. J. Am. Chem. Soc. 2000, 122, 8575.
Ho, J.-H.; Ho, T.-I.; Liu, R. S. H. Org. Lett. 2001, 3, 409.
Ho, J.-H.; Lin, J.-H.; Ho, T.-I. J. Chin. Chem. Soc. 2005, 52, 805.
Ho, J.-H.; Ho, T.-I. Tetrahedron Lett. 2003, 44, 4669.
Samori, S.; Hara, M.; Ho, T.-I.; Tojo, S.; Kawai, K.; Endo, M.; Fujitsuka, M.; Majima, T. J. Org. Chem. 2005, 70, 2708.
Ho, J.-H.; Lee, Y.-W.; Chen, Y.-Z.; Chen, P.-S.; Liu, W.-Q.; Ding, Y.-S. Tetrahedron 2013, 69, 73252.
Ho, J.-H.; Lee, T.-H.; Lo, C.-K.; Chuang, C.-L. Tetrahedron Lett. 2011, 52, 7199.
Chen, Y.-Z.; Ni, C.-W.; Teng, F.-L.; Ding, Y.-S.; Lee, T.-H.; Ho, J.-H. Tetrahedron 2014, 70, 1748.
Shirinian, V. Z.; Shimkin, A. A.; Lonshakov, D. V.; Lvov, A. G.; Krayushkin, M. M. J. Photochem. Photobiol., A 2012, 233, 1.
Lvov, A. G.; Shirinian, V. Z.; Kachala, V. V.; Kavun, A. M.; Zavarzin, I. V.; Krayushkin, M. M. Org. Lett. 2014, 16, 4532.
Lvov, A. G.; Shirinian, V. Z.; Zakharov, A. V.; Krayushkin, M. M.; Kachala, V. V.; Zavarzin, I. V. J. Org. Chem. 2015, 80, 11491.
Hoffmann, R.; Woodward, R. B. Acc. Chem. Res. 1968, 1, 17.
Shirinian, V. Z.; Lonshakov, D. V.; Lvov, A. G.; Kavun, A. M.; Yadykov, A. V.; Krayushkin, M. M. Dyes Pigm. 2016, 124, 258.
Auzias, M.; Häussinger, D.; Neuburger, M.; Wegner, H. A. Org. Lett. 2011, 13, 474.
Galangau, O.; Nakashima, T.; Maurel, F.; Kawai, T. Chem. Eur. J. 2015, 21, 8471.
Lewis, F. D., Zuo, X.; Gevorgyan, V.; Rubin, M. J. Am. Chem. Soc. 2002, 124, 13664.
Alabugin, I. V.; Manoharan, M.; Breiner, B.; Lewis, F. D. J. Am. Chem. Soc. 2003, 125, 9329.
This work was supported by the Grants Council of the President of the Russian Federation (grant MK- 6190.2016.3).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2016, 52(9), 658–665
Rights and permissions
About this article
Cite this article
Lvov, A.G., Shirinyan, V.Z. Photoinduced Rearrangements of Diarylethenes. Chem Heterocycl Comp 52, 658–665 (2016). https://doi.org/10.1007/s10593-016-1946-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-016-1946-z