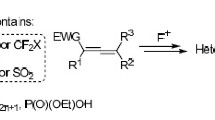
3-Aryl-2-chloropropanal was obtained by the reaction of arenediazonium chlorides with acrolein in the presence of copper(II) chloride. N-Aryl(2-pyridyl)-5-(R-benzyl)-1,3-thiazole-2-amines were formed in high yield by the reaction of these aldehydes with aryl- and 2-pyridylthioureas. The same compounds were obtained by the reaction of 5-(R-benzyl)-1,3-thiazole-2-amines with aniline.
Similar content being viewed by others
References
M. D. Obushak, V. S. Matiychuk, and V. V. Turytsa, Tetrahedron Lett., 50, 6112 (2009).
S. Ueda, H. Terauchi, M. Kawasaki, A. Yano, and M. Ido, Chem. Pharm. Bull., 52, 634 (2004).
D. Zuev, J. A. Michne, S. S. Pin, J. Zhang, M. T. Taber, and G. M. Dubowchik, Bioorg. Med. Chem. Lett., 15, 431 (2005).
A. Dondoni and A. Marra, Chem. Rev., 104, 2557 (2004).
R. M. Borzilleri, R. S. Bhide, J. C. Barrish, C. J. D'Arienzo, G. M. Derbin, J. Fargnoli, J. T. Hunt, R. Jeyaseelan, A. Kamath, D. W. Kukral, P. Marathe, S. Mortillo, L. Qian, J. S. Tokarski, B. S. Wautlet, X. Zheng, and L. J. Lombardo, J. Med. Chem., 49, 3766 (2006).
Y. Sagara, M. Mitsuya, M. Uchiyama, Y. Ogino, T. Kimura, N. Ohtake, and T. Mase, Chem. Pharm. Bull., 53, 437 (2005).
S. Wang, C. Meades, G. Wood, A. Osnowski, S. Anderson, R. Yuill, M. Thomas, M. Mezna, W. Jackson, C. Midgley, G. Griffiths, J. Fleming, S. Green, I. McNae, S.-Y. Wu, C. McInnes, D. Zheleva, M. D. Walkinshaw, and P. M. Fischer, J. Med. Chem., 47, 1662 (2004).
B. S. Zimenkovskii, R. V. Kutsyk, R. B. Lesyk, V. S. Matiychuk, N. D. Obushak, and T. I. Klofinskaya, Khim.-Farm. Zh., 40, No.6, 57 (2006).
J. V. Metzger (editor), Thiazole and Its Derivatives. John Wiley and Sons, London (1979), Vol. 3, p.23.
A. R. Katritzky, C. W. Rees, and E. F. V. Scriven (editors), Comprehensive Heterocyclic Chemistry, Elsevier (1997), Vol. 6, p. 326.
I. K. Moiseev, M. N. Zemtsova, and N. V. Makarova, Khim. Geterotsikl, Soed., 867 (1994). [Chem. Heterocycl. Comp., 30, 745 (1994)].
J. V. Metzger (editor), Thiazole and Its Derivatives. John Wiley and Sons, London (1979), Vol. 1, p. 165.
J. M. Sprague and A. H. Land, in: R. Elderfield (editor), Heterocyclic Compounds (Russian translation). Izd-vo inostr. lit., Moscow (1961), Vol. 5, p.395.
N. D. Obushak, V. S. Matiichuk, and N. I. Ganushchak, Zh. Org. Khim., 33, 1081 (1997).
N. D. Obushak, V. S. Matiichuk, R. Ya. Vasyl'shin, and Yu. V. Ostashok, Zh. Org. Khim., 40, 412 (2004).
D. S. Emolat'ev, V. L. Alifanov, V. B. Rybakov, E. V. Babaev, and E. V. Van der Eycken, Synthesis, 2083 (2008).
A. P. Engoyan, K. A. Eliazyan, V. A. Pivazyan, E. A. Kazaryan, and V. V. Dovlatyan, Khim. Geterotsikl. Soedin., 1240 (2005). [Chem. Heterocycl. Comp., 41, 1062 (2005)].
M. Annese, A. B. Corradi, L. Forlani, C. Rizzoli, and P. Sgarabotto, J. Chem. Soc., Perkin Trans 2, 615 (1994).
T. N. Birkinshaw, S. A. Harkin, P. T. Kaye, G. D. Meakins, and A. K. Smith, J. Chem. Soc., Perkin Trans 1, 939 (1982).
G. C. Barrett, Tetrahedron, 36, 2023 (1980).
Author information
Authors and Affiliations
Corresponding author
Additional information
For Communication 21, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No.4, 624-629, April 2010.
Rights and permissions
About this article
Cite this article
Matiychuk, V.S., D.Obushak, N., Pidlypnyi, N.I. et al. Synthesis of heterocycles on the basis of products of arylation of unsaturated compounds 22.* 3-Aryl-2-chloropropanal in the synthesis of N-Aryl-5-(R-benzyl)-1,3-thiazole-2-amines. Chem Heterocycl Comp 46, 495–499 (2010). https://doi.org/10.1007/s10593-010-0537-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-010-0537-7