Abstract
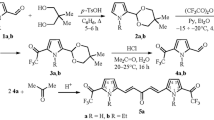
A study was carried out on the reaction of pyrrolo[1,2-a]pyrazines containing an alkyl, aryl, or aralkyl substituent at C-1 with trifluoroacetic anhydride. Trifluoroacetylation products may be formed either by reaction in the pyrrole ring or at the aryl or aralkyl groups at C-1. Products of electrophilic substitution at C-6 are formed in the trifluoroacetylation of pyrrolo[1,2-a]pyrazines containing at C-1 a substituent bulkier than a methyl group but lacking substituents at C-6 (the α-position of the pyrrole ring).
Similar content being viewed by others
References
R. Buchan, M. Fraser, and P. Kong Thoo Lin, J. Org. Chem., 54, 1074 (1989).
J. M. Minguez, M. I. Castellote, J. J. Vaquero, J. L. Garcia-Navio, J. Alvarez-Builla, and O. Castaño, J. Org. Chem., 61, 4655 (1996).
W. Paudler and D. Dunham, J. Heterocycl. Chem., 2, 410 (1965).
G. Maury, Chem. Heterocycl. Comp., 30, 223 (1977).
W. Cooper, J. Org. Chem., 23, 1382 (1958).
V. I. Terenin, E. L. Ruchkina, K. V. Karapetyan, V. M. Mamaev, and Yu. G. Bundel, Khim. Geterotsikl. Soedin., 1566 (1995). [Chem. Heterocycl. Comp., 31, 1360 (1995)].
V. I. Terenin, E. V. Kabanova, N. A. Tselishcheva, M. A. Kovalkina, A. P. Pleshkova, and N. V. Zyk, Khim. Geterotsikl. Soedin., 431 (2004). [Chem. Heterocycl. Comp., 40, 351 (2004)].
M. Kawase, M. Teshima, S. Saito, and S. Tani, Heterocycles, 48, 2103 (1998).
S. I. Bobrovskii, D. E. Lushnikov, and Yu. G. Bundel, Khim. Geterotsikl. Soedin., 1634 (1989). [Chem. Heterocycl. Comp., 25, 1360 (1989)].
V. I. Terenin, E. V. Kabanova, and Yu. G. Bundel, Khim. Geterotsikl. Soedin., 763 (1991). [Chem. Heterocycl. Comp., 27, 597 (1991)].
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1226–1233, August, 2007.
Rights and permissions
About this article
Cite this article
Terenin, V.I., Kabanova, E.V., Tselishcheva, N.A. et al. Trifluoroacetylation of pyrrolo[1,2-a]pyrazines. Chem Heterocycl Comp 43, 1038–1043 (2007). https://doi.org/10.1007/s10593-007-0162-2
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10593-007-0162-2