Abstract
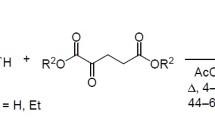
N-Substituted 5-pryazolones undergo thermal condensation with esters of β-keto acids, losing water and alcohol molecules, to form N-substituted pyrano[2,3-c]pyrazol-6-ones. The pyran ring in these products is readily cleaved by the action of alkali to give the corresponding salts of unsaturated acids.
Similar content being viewed by others
References
N. L. Nam and I. I. Grandberg, Khim. Geterotsikl. Soedin., 444 (2005).
Sheng-Chu Kuo, Li-Jian Huang, and H. Nakamura, J. Med. Chem., 27, 539 (1984).
R. Stolle, Ber., 38, 3023 (1905).
J. Elguero, R. Jacquier, and G. Tarrago, Bull. Soc. Chim. France, 3780 (1967).
M. Khan and A. Cosenza, J. Heterocycl. Chem., 19, 1077 (1982).
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 367–372, March, 2006.
Rights and permissions
About this article
Cite this article
Nam, N.L., Grandberg, I.I. Condensation of N-3-substituted 5-pyrazolones with esters of β-keto acids. Synthesis of pyrano[2,3-c]pyrazol-6-ones. Chem Heterocycl Compd 42, 326–330 (2006). https://doi.org/10.1007/s10593-006-0089-z
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s10593-006-0089-z