Abstract
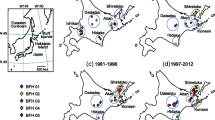
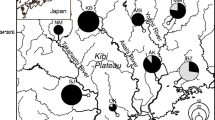
Demographic events can shape genetic diversity through genetic drift, often leaving a persistent signal in the genetic characteristics of species. Rhodeus atremius suigensis is an endangered bitterling fish endemic to the Okayama Plain, Japan. In this study, we inferred its demographic history and genetic structure using a comprehensive analysis of the mtDNA ND1 gene, microsatellite markers (MS) and MHC class IIB gene. Based on mtDNA, R. a. suigensis included two sublineages; A and B. While the former was widely distributed, the latter was restricted to eastern populations that were monomorphic in MHC. Phylogenetic analysis revealed that R. a. suigensis, together with R. a. atremius, experienced a substantial bottleneck in the middle Pleistocene. In MS and MHC, genetic diversity was low in all populations; ranked as the lowest among bitterling species. Bayesian clustering suggested that two clusters of MS had been widely introgressed in the centre of its distribution. These clusters seem to have been formed by the disruption of the distribution in the last Pleistocene, and later admixed by a large-scale reclamation in the Okayama Plain since the sixteenth century, which triggered a decline in effective population size (Ne) in many populations. Based on coalescence analysis, all populations reached their lowest Ne around the middle of the twentieth century. Accordingly, R. a. suigensis seems to have experienced two large bottlenecks in the past. While the first bottleneck was probably due to climatic changes in the middle Pleistocene, the second is due to anthropogenic degradation and fragmentation of habitats in recent years.






Similar content being viewed by others
Data availability
The datasets generated and analysed in this study are in the Dryad Digital Repository (microsatellite and MHC data: https://doi.org/10.5061/dryad.k98sf7m8h), and in DDBJ/GenBank/EMBL (mtDNA and MHC sequence data).
References
Abe T, Onikura N, Nakajima J (2015) Current status and conservation problems of two Rhodeus fishes in Japan. Jpn J Ichthyol 62:65–69. https://doi.org/10.11369/jji.11362.11365
Agbali M, Reichard M, Bryjová A, Bryja J, Smith C (2010) Mate choice for nonadditive genetic benefits correlate with MHC dissimilarity in the rose bitterling (Rhodeus ocellatus). Evolution 64:1683–1696. https://doi.org/10.1111/j.1558-5646.2010.00961.x
Allendorf FW, Luikart G, Aitken SN (2013) conservation and the genetics of populations, 2nd edn. Wiley–Blackwell, West Sussex
Banarescu P (1990) Zoogeography of fresh waters. Aula-Verlag, Wiesbaden
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Ann Statist 29:1165–1188. https://doi.org/10.1214/aos/1013699998
Bernatchez L, Wilson CC (1998) Comparative phylogeography of Nearctic and Palearctic fishes. Mol Ecol 7:431–452. https://doi.org/10.1046/j.1365-1294x.1998.00319.x
Bouckaert R, Vaughan TG, Barido-Sottani J, Duchêne S, Fourment M, Gavryushkina A, Heled J, Jones G, Kühnert D, De Maio N (2019) BEAST 2.5: an advanced software platform for Bayesian evolutionary analysis. PLoS Comput Biol 15:e1006650
Byrne M (2008) Evidence for multiple refugia at different time scales during Pleistocene climatic oscillations in southern Australia inferred from phylogeography. Quat Sci Rev 27:2576–2585. https://doi.org/10.1016/j.quascirev.2008.2508.2032
Chen M, Fontaine MC, Chehida YB, Zheng J, Labbe F, Mei Z, Hao Y, Wang K, Wu M, Zhao Q (2017) Genetic footprint of population fragmentation and contemporary collapse in a freshwater cetacean. Sci Rep 7:1–12. https://doi.org/10.1038/s41598-41017-14812-z
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659. https://doi.org/10.1046/j.1365-1294x.2000.01020.x
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772–772. https://doi.org/10.1038/nmeth.2109
Dawson DA, Burland TM, Douglas A, Le Comber SC, Bradshaw M (2003) Isolation of microsatellite loci in the freshwater fish, the bitterling Rhodeus sericeus (Teleostei: Cyprinidae). Mol Ecol Notes 3:199–202. https://doi.org/10.1046/j.1471-8286.2003.00395.x
DeWoody JA, Harder AM, Mathur S, Willoughby JR (2021) The long-standing significance of genetic diversity in conservation. Mol Ecol 30:4147–4154. https://doi.org/10.1111/mec.16051
Drummond AJ, Rambaut A, Shapiro B, Pybus OG (2005) Bayesian coalescent inference of past population dynamics from molecular sequences. Mol Biol Evol 22:1185–1192. https://doi.org/10.1093/molbev/msi1103
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-12011-19548-12687
El Mousadik A, Petit RJ (1996) High level of genetic differentiation for allelic richness among populations of the argan tree [Argania spinosa (L.) Skeels] endemic to Morocco. Theor Appl Genet 92:832–839. https://doi.org/10.1007/BF00221895
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-2294X.2005.02553.x
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Falush D, Stephens M, Pritchard JK (2003) Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics 164:1567–1587. https://doi.org/10.1093/genetics/1164.1564.1567
Frankham R, Ballow JD, Briscoe DA (2002) Introduction to conservation genetics. Cambridge University Press, Cambridge
Freeland J (2020) Molecular ecology. Wiley, Hoboken
Froese R, Pauly D (2021) FishBase. http://www.fishbase.org/search.php. Accessed on 10 Jan 2022
Fu YX (1997) Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 147:915–925. https://doi.org/10.1093/genetics/1147.1092.1915
Fujii H, Watanabe T, Yamashita Y, Tajiri N (1990) Chapter 2. Geotechnical engineering problems in Chugoku Area. 3 Reclamation in water area of Chugoku Area−specially remarked on Okayama. Plain Soil Mech Found Eng 38:59–64
Garza JC, Williamson EG (2001) Detection of reduction in population size using data from microsatellite loci. Mol Ecol 10:305–318. https://doi.org/10.1046/j.1365-1294X.2001.01190.x
Goudet J (2001) FSTAT, Version 2.9.3: a program to estimate and test gene diversities and fixation indices. Available at http://www.unil.ch/izea/software/fstat.html. Accessed on 28 Dec 2021
Harpending H (1994) Signature of ancient population growth in a low-resolution mitochondrial DNA mismatch distribution. Hum Biol 66:591–600
Hedrick WP (2010) Genetics of populations, 4th edn. Jones & Bartlett Publishers, Sudbury
Hoelzel AR, Bruford MW, Fleischer RC (2019) Conservation of adaptive potential and functional diversity. Conserv Genet 20:1–5. https://doi.org/10.1007/s10592-10019-01151-x
Höglund J (2009) Evolutionary conservation genetics. Oxford University Press, Oxford
Holderegger R, Kamm U, Gugerli F (2006) Adaptive vs. neutral genetic diversity: implications for landscape genetics. Landsc Ecol 21:797–807. https://doi.org/10.1007/s10980-10005-15245-10989
Jeon H-B, An J, Kweon S-M, Kim S, Yu J-N, Kim B-J, Kawase S, Suk HY (2016) Development of novel microsatellite loci and analyses of genetic diversity in the endangered Tanakia somjinensis. Biochem Syst Ecol 66:344–350. https://doi.org/10.1016/j.bse.2016.1005.1006
Jeon H-B, Won H, Suk HY (2019) Polymorphism of MHC class IIB in an acheilognathid species, Rhodeus sinensis shaped by historical selection and recombination. BMC Genet 20:1–12. https://doi.org/10.1186/s12863-12019-10775-12863
Jombart T (2008) adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405. https://doi.org/10.1093/bioinformatics/btn1129
Jombart T, Pontier D, Dufour A-B (2009) Genetic markers in the playground of multivariate analysis. Heredity 102:330–341. https://doi.org/10.1038/hdy.2008.1130
Jones OR, Wang J (2010) COLONY: a program for parentage and sibship inference from multilocus genotype data. Mol Ecol Resour 10:551–555. https://doi.org/10.1111/j.1755-0998.2009.02787.x
Kalinowski ST (2005) HP–RARE 1.0: a computer program for performing rarefaction on measures of allelic richness. Mol Ecol Notes 5:187–189. https://doi.org/10.1111/j.1471-8286.2004.00845.x
Kalkvik HM, Stout IJ, Parkinson CL (2012) Unraveling natural versus anthropogenic effects on genetic diversity within the southeastern beach mouse (Peromyscus polionotus niveiventris). Conserv Genet 13:1653–1664. https://doi.org/10.1007/s10592-10012-10417-z
Kawabe T (1989) Stratigraphy of the lower part of the Kobiwako Group around the Ueno basin, Kinki district, Japan. J Geosci Osaka City Univ 32:39–52
Kawamura K, Ueda T, Arai R, Nagata Y, Saitoh K, Ohtaka H, Kanoh Y (2001) Genetic introgression by the rose bitterling, Rhodeus ocellatus ocellatus, into the Japanese rose bitterling, R. o. kurumeus (Teleostei: Cyprinidae). Zool Sci 18:1027–1039. https://doi.org/10.2108/zsj.1018.1027
Kawamura K, Ueda T, Arai R, Smith C (2014) Phylogenetic relationships of bitterling fishes (Teleostei: Cypriniformes: Acheilognathinae), inferred from mitochondrial cytochrome b sequences. Zool Sci 31:321–329. https://doi.org/10.2108/zs130233
Kim W, Shin E, Kong H, Kim H, Kim B, Nam B, Kim Y, Kim C, Jung H, An C (2014) Characterization of novel microsatellite markers derived from Korean rose bitterling (Rhodeus uyekii) genomic library. Genet Mol Res 13:8147–8152. https://doi.org/10.4238/2014.october.8147.8149
Kirk H, Freeland JR (2011) Applications and implications of neutral versus non-neutral markers in molecular ecology. Int J Mol Sci 12:3966–3988. https://doi.org/10.3390/ijms12063966
Kitagawa T, Jeon S-R, Kitagawa E, Yoshioka M, Kashiwagi M, Okazaki T (2005) Genetic relationships among the Japanese and Korean striated spined loach complex (Cobitidae: Cobitis) and their phylogenetic positions. Ichthyol Res 52:111–122. https://doi.org/10.1007/s10228-10004-10261-10223
Kitamura J, Uchiyama R (2020) Bitterling fishes of japan-natural history and culture. Yamakei Press, Tokyo
Koizumi I, Usio N, Kawai T, Azuma N, Masuda R (2012) Loss of genetic diversity means loss of geological information: the endangered Japanese crayfish exhibits remarkable historical footprints. PLoS ONE 7:e33986. https://doi.org/10.31371/journal.pone.0033986
Kondo T, Nakagoshi N, Isagi Y (2009) Shaping of genetic structure along Pleistocene and modern river systems in the hydrochorous riparian azalea, Rhododendron ripense (Ericaceae). Am J Bot 96:1532–1543. https://doi.org/10.3732/ajb.0800273
Kubota H, Watanabe K (2013) Loss of genetic diversity at an MHC locus in the endangered Tokyo bitterling Tanakia tanago (Teleostei: Cyprinidae). Zool Sci 30:1092–1101. https://doi.org/10.2108/zsj.1030.1092
Kubota H, Watanabe K, Suguro N, Tabe M, Umezawa K, Watanabe K (2010) Genetic population structure and management units of the endangered Tokyo bitterling, Tanakia tanago (Cyprinidae). Conserv Genet 11:2343–2355. https://doi.org/10.1007/s10592-10010-10120-x
Kuwashiro I (1959) Submarine topography of Japanese inlandsea Setonaikai. Geogr Rev Jpn 32:24–35
Leigh JW, Bryant D (2015) popart: full-feature software for haplotype network construction. Methods Ecol Evol 6:1110–1116. https://doi.org/10.1111/2041-1210X.12410
Luikart G, Allendorf FW, Cornuet JM, Sherwin WB (1998) Distortion of allele frequency distributions provides a test for recent population bottlenecks. J Hered 89:238–247. https://doi.org/10.1093/jhered/1089.1093.1238
Lynch M, Ritland K (1999) Estimation of pairwise relatedness with molecular markers. Genetics 152:1753–1766
Machida H, Matsuda T, Umitsu M, Koizumi T (2006) Geomorphology of Chubu Regional. Geomorphology of the Japanese islands, vol 5. University of Tokyo Press, Tokyo
Mattucci F, Oliveira R, Lyons LA, Alves PC, Randi E (2016) European wildcat populations are subdivided into five main biogeographic groups: consequences of Pleistocene climate changes or recent anthropogenic fragmentation? Ecol Evol 6:3–22. https://doi.org/10.1002/ece1003.1815
Mills LS (2013) Conservation of wildlife populations: demography, genetics, and management, 2nd edn. Wiley-Blackwell, West Sussex
Milot E, Béchet A, Maris V (2020) The dimensions of evolutionary potential in biological conservation. Evol Appl 13:1363–1379. https://doi.org/10.1111/eva.12995
Ministry of the Environment Government of Japan (MEGJ) (2015) Threatened wildlife of Japan, red data book 2014: pisces-brackish and fresh water fishes. Gyosei, Tokyo
Ministry of the Environment Government of Japan (MEGJ) (2020) Publication of the Japanese red list 2020. https://www.env.go.jp/press/107905.html. Accessed on 20 Dec 2021
Ministry of the Environment Government of Japan (MEGJ) (2021) List of national endangered species of wild fauna and flora. https://www.env.go.jp/nature/kisho/domestic/list.html#gyorui. Accessed on 20 Dec 2021
Miyake T, Kawamura K, Hosoya K, Okazaki T, Kitagawa T (2007) Discovery of the Japanese rosy bitterling, Rhodeus ocellatus kurumeus, in Nara Prefecture. Jpn J Ichthyol 54:139–148. https://doi.org/10.11369/jji11950.11354.11139
Miyake T, Nakajima J, Onikura N, Ikemoto S, Iguchi K, Komaru A, Kawamura K (2011) The genetic status of two subspecies of Rhodeus atremius, an endangered bitterling in Japan. Conserv Genet 12:383–400. https://doi.org/10.1007/s10592-10010-10146-10590
Miyake T, Nakajima J, Umemura K, Onikura N, Ueda T, Smith C, Kawamura K (2021) Genetic diversification of the Kanehira bitterling Acheilognathus rhombeus inferred from mitochondrial DNA, with comments on the phylogenetic relationship with its sister species Acheilognathus barbatulus. J Fish Biol 99:1677–1695. https://doi.org/10.1111/jfb.14876
Nakamura M (1969) Cyprinid fishes of Japan: studies on the life history of cyprinid fishes of Japan. Research Institute for National Resources, Tokyo
Nakano T, Kobayashi K (1959) Nature of Japan. Iwanami Shoten, Tokyo
Narum SR (2006) Beyond Bonferroni: less conservative analyses for conservation genetics. Conserv Genet 7:783–787. https://doi.org/10.1007/s10592-10005-19056-y
Nei M (1978) Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89:583–590. https://doi.org/10.1093/genetics/1089.1093.1583
Neki A, Neki O (1997) Small ecology system of fishes in the area of water-way for irrigation, and the pernicious influence to this system by the improvement and its restoration: as a case study of the Okayama plains. Bull Nagaoka Univ Technol 19:141–150
Nikolic N, Chevalet C (2014) Detecting past changes of effective population size. Evol Appl 7:663–681. https://doi.org/10.1111/eva.12170
O’Brien SJ, Wildt DE, Bush M, Caro TM, FitzGibbon C, Aggundey I, Leakey RE (1987) East African cheetahs: evidence for two population bottlenecks? Proc Natl Acad Sci USA 84:508–511. https://doi.org/10.1073/pnas.1084.1072.1508
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research-an update. Bioinformatics 28:2537–2539
Piry S, Luikart G, Cornuet JM (1999) BOTTLENECK: a computer program for detecting recent reductions in the effective size using allele frequency data. J Hered 90:502–503. https://doi.org/10.1093/jhered/1090.1094.1502
Provan J, Bennett K (2008) Phylogeographic insights into cryptic glacial refugia. Trends Ecol Evol 23:564–571. https://doi.org/10.1016/j.tree.2008.1006.1010
Radwan J, Biedrzycka A, Babik W (2010) Does reduced MHC diversity decrease viability of vertebrate populations? Biol Conserv 143:537–544. https://doi.org/10.1016/j.biocon.2009.1007.1026
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Syst Biol 67:901–904. https://doi.org/10.1093/sysbio/syy1032
Raymond M, Rousset F (1995) GENEPOP (Version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249. https://doi.org/10.1093/oxfordjournals.jhered.a111573
Ritchie AM, Lo N, Ho SY (2017) The impact of the tree prior on molecular dating of data sets containing a mixture of inter-and intraspecies sampling. Syst Biol 66:413–425. https://doi.org/10.1093/sysbio/syw1095
Rogers AR (1995) Genetic evidence for a Pleistocene population explosion. Evolution 49:608–615. https://doi.org/10.1111/j.1558-5646.1995.tb02297.x
Rougemont Q, Moore J-S, Leroy T, Normandeau E, Rondeau EB, Withler RE, Van Doornik DM, Crane PA, Naish KA, Garza JC (2020) Demographic history shaped geographical patterns of deleterious mutation load in a broadly distributed Pacific Salmon. PLoS Genet 16:e1008348
Sambrook JG, Figueroa F, Beck S (2005) A genome-wide survey of major histocompatibility complex (MHC) genes and their paralogues in zebrafish. BMC Genomics 6:1–10. https://doi.org/10.1186/1471-2164-1186-1152
Schuelke M (2000) An economic method for the fluorescent labeling of PCR fragments. Nat Biotechnol 18:233–234. https://doi.org/10.1038/72708
Shen X, Wan S, Colin C, Tada R, Shi X, Pei W, Tan Y, Jiang X, Li A (2018) Increased seasonality and aridity drove the C4 plant expansion in Central Asia since the Miocene-Pliocene boundary. Earth Planet Sci Lett 502:74–83. https://doi.org/10.1016/j.epsl.2018.1008.1056
Shirai Y, Ikeda S, Tajima S (2009) Isolation and characterization of new microsatellite markers for rose bitterlings, Rhodeus ocellatus. Mol Ecol Resour 9:1031–1033. https://doi.org/10.1111/j.1755-0998.2009.02566.x
Slatkin M (1985) Rare alleles as indicators of gene flow. Evolution 39:53–65. https://doi.org/10.1111/j.1558-5646.1985.tb04079.x
Smith C, Reichard M, Jurajda P, Przybylski M (2004) The reproductive ecology of the European bitterling (Rhodeus sericeus). J Zool 262:107–124. https://doi.org/10.1017/S0952836903004497
Sommer S (2005) The importance of immune gene variability (MHC) in evolutionary ecology and conservation. Front Zool 2:1–18. https://doi.org/10.1186/1742-9994-1182-1116
Stockwell CA, Hendry AP, Kinnison MT (2003) Contemporary evolution meets conservation biology. Trends Ecol Evol 18:94–101. https://doi.org/10.1016/S0169-5347(1002)00044-00047
Suzuki N (1995) Rhodeus atremius suigensis. In: Fisheries Agencyin Japan (ed) Basic data of rare wild aquatic organisms in Japan (II). Japan Fisheries Resource Conservation Association, Tokyo, pp 308–313
Tabata R, Kakioka R, Tominaga K, Komiya T, Watanabe K (2016) Phylogeny and historical demography of endemic fishes in Lake Biwa: the ancient lake as a promoter of evolution and diversification of freshwater fishes in western Japan. Ecol Evol 6:2601–2623. https://doi.org/10.1002/ece2603.2070
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595. https://doi.org/10.1093/genetics/1123.1093.1585
Teixeira JC, Huber CD (2021) The inflated significance of neutral genetic diversity in conservation genetics. Proc Natl Acad Sci USA 118:e2015096118
Templeton A, Crandall K, Sing C (1992) A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping and DNA sequence data. III. Cladogram estimation. Genetics 132:619–633. https://doi.org/10.1093/genetics/1132.1092.1619
Tominaga K, Nakajima J, Watanabe K (2016) Cryptic divergence and phylogeography of the pike gudgeon Pseudogobio esocinus (Teleostei: Cyprinidae): a comprehensive case of freshwater phylogeography in Japan. Ichthyol Res 63:79–93. https://doi.org/10.1007/s10228-10019-00715-10228
Tominaga K, Nagata N, Kitamura J-i, Watanabe K, Sota T (2020) Phylogeography of the bitterling Tanakia lanceolata (Teleostei: Cyprinidae) in Japan inferred from mitochondrial cytochrome b gene sequences. Ichthyol Res 67:105–116. https://doi.org/10.1007/s10228-10015-10478-10223
van Oosterhout C, Hutchinson WF, Wills DP, Shipley P (2004) MICRO–CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538. https://doi.org/10.1111/j.1471-8286.2004.00684.x
Wang J (2009) A new method for estimating effective population sizes from a single sample of multilocus genotypes. Mol Ecol 18:2148–2164. https://doi.org/10.1111/j.1365-2294X.2009.04175.x
Watanabe K, Takahashi H (2010) Natural history of freshwater fish geography. Hokkaido University Press, Sapporo
Wiepkema PR (1961) An ethological analysis of the reproductive behaviour of the bitterling (Rhodeus amarus Bloch). Arch Neerl Zool 14:103–199
Won H, Jeon H-B, Suk HY (2020) Evidence of an ancient connectivity and biogeodispersal of a bitterling species, Rhodeus notatus, across the Korean Peninsula. Sci Rep 10:1–13. https://doi.org/10.1038/s41598-41020-57625-41593
Won H, Jeon H-B, Kim D-Y, Suk HY (2021) Differential patterns of diversity at neutral and adaptive loci in endangered Rhodeus pseudosericeus populations. Sci Rep 11:1–12. https://doi.org/10.1038/s41598-41021-95385-w
Xiong LW, Wang Q, Wang SB, Wang JG, Feng Q, Yue LJ (2017) Description of 21 microsatellites for the Chinese bitterling, Rhodeus sinensis Günther, 1868. J Appl Ichthyol 33:940–942. https://doi.org/10.1111/jai.13380
Yamazaki Y, Uehara K, Ikeya K, Nishio M (2020) Interpopulational and intrapopulational genetic diversity of the endangered Itasenpara bitterling (Acheilognathus longipinnis) with reference to its demographic history. Conserv Genet 21:55–64. https://doi.org/10.1007/s10592-10019-01232-x
Young AG, Clarke GM (2000) Genetics, demography and viability of fragmented populations. Cambridge University Press, Cambridge
Yu D, Chen M, Tang Q, Li X, Liu H (2014) Geological events and Pliocene climate fluctuations explain the phylogeographical pattern of the cold water fish Rhynchocypris oxycephalus (Cypriniformes: Cyprinidae) in China. BMC Evol Biol 14:1–12. https://doi.org/10.1186/s12862-12014-10225-12869
Zheng H, Powell CM, Rea DK, Wang J, Wang P (2004) Late Miocene and mid-Pliocene enhancement of the East Asian monsoon as viewed from the land and sea. Glob Planet Change 41:147–155. https://doi.org/10.1016/j.gloplacha.2004.1001.1003
Acknowledgements
We thank Shigetoyo Ikemoto, Hiroshi Aoe, Tetsushi Furumoto, Yuji Ohkita, Yasunori Tanaka, Masakazu Ao, Takayuki Muro, Dr Kazuhiko Uehara and Prof. Shuji Kobayashi for collecting samples of R. a. suigensis. This study was carried out as part of a conservation project for R. a. suigensis organized by Ministry of the Environment Government of Japan, the Fisheries Agency in Japan and Okayama Prefectural Office. We express sincere thanks to two anonymous reviewers for the critical comments and useful suggestions that have helped us improve our paper considerably.
Funding
This study was supported by Ministry of the Environment Government of Japan, the Fisheries Agency in Japan and Okayama Prefectural Office.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conceptions and design. Manuscript preparation and sample analyses were done by KK and TM, and all authors commented on the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent for publication
All authors approved the submission and subsequent publication of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kawamura, K., Miyake, T. & Smith, C. Population genetic structure and demographic history of Rhodeus atremius suigensis, an endangered bitterling in Japan. Conserv Genet 23, 885–901 (2022). https://doi.org/10.1007/s10592-022-01461-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-022-01461-7