Abstract
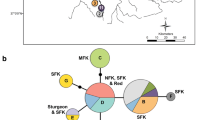
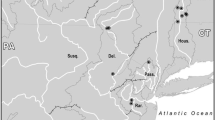
The eastern hellbender has declined across much of its range and is a candidate for listing under the Endangered Species Act. Some of the most viable remaining populations exist in the Southern Appalachian Region in the Tennessee Valley watershed; however these populations are highly isolated and fragmented, and occupy several physiographic provinces, suggesting they may exhibit significant levels of genetic differentiation. We investigated genetic and phylogeographic relationships among eastern hellbender populations across the Tennessee Valley, using nuclear microsatellite markers and mitochondrial sequence data. Our population genetic analyses of microsatellite data revealed a strong pattern of isolation by stream distance, and 4 genetically distinct populations. These four populations were mainly associated with major watersheds, although middle Tennessee samples were difficult to assign to any particular population. Our phylogeographic analysis of mtDNA resulted in a strongly supported monophyletic ingroup containing nine largely allopatric clades, which also largely corresponded to major watersheds. Our findings suggest that hellbenders from different watersheds in the Tennessee Valley should be recognized as genetically distinct populations, and care should be taken to balance the needs of rescuing declining populations with translocation or headstart programs, while also preserving genetic diversity across the region.






Similar content being viewed by others
References
Ahlstedt SA, Powell JR, Butler RS, Fagg MT, Hubbs DW, Novak SF, Palmer SR, Johnson PD (2004) Historical and current examination of freshwater mussels (Bivalvia: Margaritiferidae, Unionidae) in the Duck River basin, Tennessee. Tennessee Wildlife Resources Agency, Nashville. Available from https://www.researchgate.net/profile/Jeff_Powell2/publication/260176288_Historical_and_Current_Examination_of_Freshwater_Mussels_Bivalvia_Margaritiferidae_Unionidae_in_the_Duck_River_Basin_Tennessee/links/0a85e52fe3a78b5fd8000000/Historical-and-Current-Examination-of-Freshwater-Mussels-Bivalvia-Margaritiferidae-Unionidae-in-the-Duck-River-Basin-Tennessee.pdf. Accessed 10 June 2017
Bodinof C, Briggler J, Duncan M, Milspaugh J (2011) Historic occurrence of the amphibian chytrid fungus Batrachochytrium dendrobatidis in hellbender Cryptobranchus alleganiensis populations from Missouri. Dis Aquat Org 96:1–7. https://doi.org/10.3354/dao02380
Bodinof CM, Briggler JT, Junge RE et al (2012) Postrelease movements of captive-reared Ozark hellbenders (Cryptobranchus alleganiensis bishopi). Herpetologica 68:160–173
Bothner R, Gottlieb JA (1991) A study of the New York state populations of the hellbender, Cryptobranchus alleganiensis alleganiensis (Daudin). Proc Rochester Acad Sci 17:41–54
Bouzat JL, Johnson JA, Toepfer JE et al (2009) Beyond the beneficial effects of translocations as an effective tool for the genetic restoration of isolated populations. Conserv Genet 10:191–201. https://doi.org/10.1007/s10592-008-9547-8
Briggler JT, Ultrup J, Davidson C et al (2007) Hellbender population and habitat viability assessment. Apple Valley, Minnesota
Burgmeier NG, Unger SD, Meyer JL et al (2011a) Health and habitat quality assessment for the eastern hellbender (Cryptobranchus alleganiensis alleganiensis) in Indiana, USA. J Wildl Dis 47:836–848
Burgmeier NG, Unger SD, Sutton TM, Williams RN (2011b) Population status of the eastern hellbender (Cryptobranchus alleganiensis alleganiensis) in Indiana. J Herpetol 45:195–201
Caruso NM, Sears MW, Adams DC, Lips KR (2014) Widespread rapid reductions in body size of adult salamanders in response to climate change. Glob Change Biol 20:1751–1759. https://doi.org/10.1111/gcb.12550
Chhatre VE, Emerson KJ (2017) StrAuto: automation and parallelization of STRUCTURE analysis. BMC Bioinform 18:192. https://doi.org/10.1186/s12859-017-1593-0
Crowhurst RS, Faries KM, Collantes J et al (2011) Genetic relationships of hellbenders in the Ozark highlands of Missouri and conservation implications for the Ozark subspecies (Cryptobranchus alleganiensis bishopi). Conserv Genet 12:637–646. https://doi.org/10.1007/s10592-010-0170-0
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-011-9548-7
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Foster RL, McMillan AM, Roblee KJ (2009) Population status of hellbender salamanders (Cryptobranchus alleganiensis) in the Allegheny River drainage of New York State. J Herpetol 43:579–588.. https://doi.org/10.1670/08-156.1
Freake MJ, DePerno CS (2017) Importance of demographic surveys and public lands for the conservation of eastern hellbenders Cryptobranchus alleganiensis alleganiensis in southeast USA. PLoS ONE 12:e0179153. https://doi.org/10.1371/journal.pone.0179153
GADNR (2015) Georgia State Wiuldlife Action Plan. Available from http://georgiawildlife.com/sites/default/files/wrd/pdf/swap/SWAP2015MainReport_92015.pdf. Accessed 16 June 2017
Gilpin S, Soule M (1986) Minimum viable populations: processes of species extinction. In: Soule ME, Sinauer (eds) Conservation biology: the science of scarcity and diversity. Sinauer Associates, Sunderland, pp 19–34
Hecht-Kardasz KA, Nickerson MA, Freake M, Colclough P (2012) Population structure of the Hellbender (Cryptobranchus alleganiensis) in a Great Smoky Mountains stream. Bull Fla Mus Nat Hist 51:227–241
Hedrick PW (2001) Conservation genetics: where are we now? Trends Ecol Evol 16:629–636
Hendry AP, Lohmann LG, Conti E et al (2010) Evolutionary biology in biodiversity science, conservation, and policy: a call to action. Evolution 64(5):1517–1528. https://doi.org/10.1111/j.1558-5646.2010.00947.x
Hubisz MJ, Falush D, Stephens M, Pritchard JK (2009) Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour 9:1322–1332. https://doi.org/10.1111/j.1755-0998.2009.02591.x
Hudson RR, Coyne JA (2002) Mathematical consequences of the genealogical species concept. Evolution 56:1557–1565. https://doi.org/10.1111/j.0014-3820.2002.tb01467.x
Kearse M, Moir R, Wilson A et al (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Lanfear R, Calcott B, Ho SYW, Guindon S (2012) PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol 29:1695–1701. https://doi.org/10.1093/molbev/mss020
Lankau R, Jørgensen PS, Harris DJ, Sih A (2011) Incorporating evolutionary principles into environmental management and policy: evolutionary environmental management. Evol Appl 4:315–325. https://doi.org/10.1111/j.1752-4571.2010.00171.x
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Martel A, Blooi M, Adriaensen C et al (2014) Recent introduction of a chytrid fungus endangers Western Palearctic salamanders. Science 346:630–631
Mast MA, Turk JT (1999) Environmental characteristics and water quality of hydrologic benchmark network stations in the eastern United States, 1963–1995. U.S. G.P.O.; Free on application to the U.S. Geological Survey, Information Services, Washington, DC, Denver
Mayasich J, Grandmaison D, Phillips C (2003) Eastern hellbender status assessment report. Nat Resour Res Inst Tech Rep 9:1–41
Meirmans PG, Hedrick PW (2011) Assessing population structure: FST and related measures: invited technical review. Mol Ecol Resour 11:5–18. https://doi.org/10.1111/j.1755-0998.2010.02927.x
Meirmans PG, Van Tienderen PH (2004) genotype and genodive: two programs for the analysis of genetic diversity of asexual organisms. Mol Ecol Notes 4:792–794. https://doi.org/10.1111/j.1471-8286.2004.00770.x
Miller BT, Miller JL (2005) Prevalence of physical abnormalities in eastern Hellbender (Cryptobranchus alleganiensis alleganiensis) populations of middle Tennessee. Southeast Nat 4:513–520
NCWRC (2015) North Carolina State Wildlife Action Plan. Available from: http://www.ncwildlife.org/Portals/0/Conserving/documents/2015WildlifeActionPlan/NC-WAP-2015-All-Documents.pdf. Accessed 16 June 2017
Nickerson MA, Mays CE (1973a) The hellbenders: North American “giant salamanders. Milwaukee Public Mus Special Publ Biol Geol 1:1–106
Nickerson MA, Mays CE (1973b) A study of the Ozark Hellbender Cryptobranchus alleganiensis bishopi. Ecology 54:1164.. https://doi.org/10.2307/1935586
Nickerson MA, Krysko KL, Owen RD (2002) Ecological status of the hellbender (Cryptobranchus alleganiensis) and the mudpuppy (Necturus maculosus) salamanders in the Great Smoky Mountains National Park. J N C Acad Sci 118:27–34
Peterson CL, Wilkinson RF Jr, Topping MS, Metter DE (1983) Age and growth of the Ozark hellbender (Cryptobranchus alleganiensis bishopi). Copeia 1:225–231
Peterson CL, Metter DE, Miller BT et al (1988) Demography of the hellbender Cryptobranchus alleganiensis in the Ozarks. Am Midl Nat 119:291–303
Petranka JW (1998) Salamanders of the United States and Canada. Smithsonian Institution Press, Washington
Phillips CA (1994) Geographic distribution of mitochondrial DNA variants and the historical biogeography of the spotted salamander, Ambystoma maculatum. Evolution 48:597–607. https://doi.org/10.2307/2410472
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Pugh MW, Groves JD, Williams LA, Gangloff MM (2013) A previously undocumented locality of eastern hellbenders (Cryptobranchus alleganiensis alleganiensis) in the Elk River, Carter County, TN. Southeast Nat 12:137–142. https://doi.org/10.1656/058.012.0111
Pugh MW, Hutchins M, Madritch M et al (2015) Land-use and local physical and chemical habitat parameters predict site occupancy by hellbender salamanders. Hydrobiologia. https://doi.org/10.1007/s10750-015-2570-0
Quinn SA, Gibbs JP, Hall MH, Petokas PJ (2013) Multiscale factors influencing distribution of the eastern hellbender salamander (Cryptobranchus alleganiensis alleganiensis) in the northern segment of its range. J Herpetol 47:78–84
Rissler L, Apodaca J (2007) Adding more ecology into species delimitation: ecological niche models and phylogeography help define cryptic species in the black salamander (Aneides flavipunctatus). Syst Biol 56:924–942. https://doi.org/10.1080/10635150701703063
Ronquist F, Teslenko M, van der Mark P et al (2012) MrBayes 3.2: efficient bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Rousset F (1997) Genetic differentiation and estimation of gene flow from F-statistics under isolation by distance. Genetics 145:1219–1228
Routman E, Wu R, Templeton AR (1994) Parsimony, molecular evolution, and biogeography: the case of the North American giant salamander. Evolution 48:1799–1809. https://doi.org/10.2307/2410509
Sabatino SJ, Routman EJ (2009) Phylogeography and conservation genetics of the hellbender salamander (Cryptobranchus alleganiensis). Conserv Genet 10:1235–1246. https://doi.org/10.1007/s10592-008-9655-5
Shaffer ML, Stein BA (2000) Safeguarding our precious heritage. Precious heritage: The status of biodiversity in the United States. Oxford University Press, New York, pp 301–321
Smith J, Haigh J (1974) The hitch-hiking effect of a favourable gene. Genet Res 23(1):23–35. https://doi.org/10.1017/S0016672300014634
Templeton AR, Shaw K, Routman E, Davis SK (1990) The genetic consequences of habitat fragmentation. Ann Mo Bot Gard 77:13–27. https://doi.org/10.2307/2399621
Tonione M, Johnson JR, Routman EJ (2011) Microsatellite analysis supports mitochondrial phylogeography of the hellbender (Cryptobranchus alleganiensis). Genetica 139:209–219. https://doi.org/10.1007/s10709-010-9538-9
Trauth SE, Wilhide JD, Daniel P (1992) Status of the Ozark hellbender Cryptobranchus bishopi (Urodela: Cryptobranchidae), in the Spring River, Fulton County, Arkansas. Proc Ark Acad Sci 46:83–86
TWRA (2015) Tennessee State Wildlife Action Plan. Available from http://www.tnswap.com/swap.cfm. Accessed 16 June 2017
Unger SD, Fike JA, Sutton T et al (2010) Isolation and development of 12 polymorphic tetranucleotide microsatellite markers for the eastern hellbender (Cryptobranchus alleganiensis alleganiensis). Conserv Genet Resour 2:89–91. https://doi.org/10.1007/s12686-009-9170-0
Unger SD, Rhodes OE, Sutton TM, Williams RN (2013) Population genetics of the eastern hellbender (Cryptobranchus alleganiensis alleganiensis) across multiple spatial scales. PLoS ONE 8:e74180. https://doi.org/10.1371/journal.pone.0074180
United States Fish and Wildlife Service (2011a) Endangered and threatened wildlife and plants; endangered status for the ozark hellbender salamander. 50 CFR Part 23. Fed Regist 76:61956‒61978
United States Fish and Wildlife Service (2011b) Inclusion of the hellbender, including the eastern hellbender and the ozark hellbender, in appendix III of the convention on international trade in endangered species of wild fauna and flora (CITES). 50 CFR Part 23. Fed Regist 76:61978–61985
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) micro-checker: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538. https://doi.org/10.1111/j.1471-8286.2004.00684.x
Weeks AR, Sgro CM, Young AG et al (2011) Assessing the benefits and risks of translocations in changing environments: a genetic perspective: translocations in changing environments. Evol Appl 4:709–725. https://doi.org/10.1111/j.1752-4571.2011.00192.x
Wheeler BA, Prosen E, Mathis A, Wilkinson RF (2003) Population declines of a long-lived salamander: a 20+-year study of hellbenders, Cryptobranchus alleganiensis. Biol Conserv 109:151–156
Williams RD, Gates JE, Hocutt CH, Taylor GJ (1981) The hellbender: a nongame species in need of management. Wildl Soc Bull 9:94–100
Acknowledgements
We thank the many individuals who assisted in conducting fieldwork, including Lee University undergraduate students, D. Hedrick (Chattanooga Zoo), and J. Herrig (USDA National Forest Service). We also thank the following funding sources: Tennessee Wildlife Resource Agency, USDA Forest Service, Appalachian College Association, Lee University, Indiana Department of Natural Resources, University of Tennessee at Chattanooga, Purdue University, Cryptobranchid Interest Group Ron Goellner Fund.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Freake, M., O’Neill, E., Unger, S. et al. Conservation genetics of eastern hellbenders Cryptobranchus alleganiensis alleganiensis in the Tennessee Valley. Conserv Genet 19, 571–585 (2018). https://doi.org/10.1007/s10592-017-1033-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-017-1033-8