Abstract
Faeces have proved to be a suitable non-invasive DNA source for microsatellite analysis in wildlife research. For the success of such studies it is essential to obtain the highest possible PCR amplification success rate. These rates are still relatively low in most carnivorous species, especially in the otter (Lutra lutra). We therefore optimised the entire microsatellite genotyping process by combining our findings with results from previous studies to gain a high rate of reliable genotypes. We investigated the influence of otter faecal quality in relation to the quantity of slimy secretions and three levels of storage periods at −20°C on amplification success. Further, we tested the cost-effective and time-saving Chelex extraction method against the profitable QIAamp® DNA Stool Kit (Qiagen), and compared three PCR methods - a standard single-step PCR protocol, a single-locus two-step PCR procedure and a multiplex two-step PCR procedure - regarding success rate and genotyping errors. The highest amplification success rate (median: 94%; mean: 78%) was achieved using faecal samples consisting only of jelly extracted with the QIAamp® DNA Stool Mini Kit (Qiagen) immediately after collection and amplified following the time and cost efficient multiplex two-step PCR protocol. The two-step procedure, also referred to as pre-amplification approach, turned out to be the main improvement as it increases amplification success about 11% and reduces genotyping errors about 53%, most notably allelic dropouts.




Similar content being viewed by others
References
Amos B, Pemberton J (1992) DNA fingerprinting in non-human populations. Curr Opin Genet Dev 2:857–860
Banks SC, Piggott MP, Hansen BD, Robinson NA, Taylor AC (2002) Wombat coprogenetics enumerating a common wombat population by microsatellite analysis of faecal DNA. Aust J Zool 50:193–204
Broquet T, Petit E (2004) Quantifying genotyping errors in noninvasive population genetics. Mol Ecol 13:3601–3608
Brownstein MJ, Carpten JD, Smith JR (1996) Modulation of non-templated nucleotide addition by Taq DNA polymerase: primer modifications that facilitate genotyping. Biotechniques 20:1004–1006
Bruford MW, Wayne RK (1993) Microsatellites and their application to population genetic studies. Curr Opin Genet Dev 3:939–943
Carss DN, Parkinson SG (1996) Errors associated with otter Lutra lutra faecal analysis. 1. Assessing general diet from spraints. J Zool 238:301–317
Conroy JWH, French DD (1991) Seasonal patterns in the sprainting behaviour of otters (Lutra lutra L.) in Shetland. In: Reuther C, Röchert R (eds) Proceedings of 5th International Otter Colloquium, Hankensbüttel, 1989
Coxon K, Chanin P, Dallas J, Sykes T (1999) The use of DNA fingerprinting to study the population dynamics of otters (Lutra lutra) in southern Britain a feasibility study. Environment Agency R&D Project W1-025
Creel S, Spong G, Sands JL, Rotella J, Zeigle J, Joe L, Murphy KM, Smith D (2003) Population size estimation in Yellowstone wolves with error-prone noninvasive microsatellite genotypes. Mol Ecol 12:2003–2009
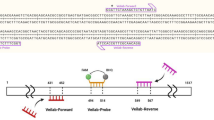
Dallas JF, Piertney SB (1998) Microsatellite primers for the Eurasian otter. Mol Ecol 7:1247–1263
Dallas JF, Bacon PJ, Carss DN, Conroy JWH, Green R, Jefferies DJ, Kruuk H, Marshall F, Piertney SB, Racey PA (1999) Genetic diversity in the Eurasian otterLutra lutra in Scotland Evidence from microsatellite polymorphism. Biol J Linn Soc 68:73–86
Dallas JF, Marshall F, Piertney SB, Bacon PJ, Racey PA (2002) Spatially restricted gene flow and reduced microsatellite polymorphism in the Eurasion otter Lutra lutra in Britain. Conserv Genet 3:15–29
Dallas JF, Coxon KE, Sykes T, Chanin PRF, Marshall F, Carss DN, Bacon PJ, Piertney SB, Racey PA (2003) Similar estimates of population genetic composition and sex ratio derived from carcasses and faeces of Eurasian otter Lutra lutra. Mol Ecol 12:275–282
Deuter R, Pietsch S, Hertel S, Müller O (1995) A method for preparation of fecal DNA suitable for PCR. Nucleic Acids Res 23:3800–3801
Ernest HB, Penedo MCT, May BP, Syvanen M, Boyce WM (2000) Molecular tracking of mountain lions in the Yosemite Valley region in California genetic analysis using microsatellites and faecal DNA. Mol Ecol 9:433–441
Farrell LE, Roman J, Sunquist ME (2000) Dietary separation of sympatric carnivores identified by molecular analysis of scats. Mol Ecol 9:1583–1590
Flagstad O, Roed K, Stacy JE, Jakobsen KS (1999) Reliable noninvasive genotyping based on excremental PCR of nuclear DNA purified with a magnetic bead protocol. Mol Ecol 8:879–883
Frantz AC, Pope LC, Carpenter PJ, Roper TJ, Wilson GJ, Delahay RJ, Burke T (2003) Reliable microsatellite genotyping of the Eurasian badger (Meles meles) using faecal DNA. Mol Ecol 12:1649–1661
Frantz AC, Schaul M, Pope LC, Fack F, Schley L, Muller CP, Roper TJ (2004) Estimating population size by genotyping remotely plucked hair the Eurasian badger. J Appl Ecol 41:985–995
Frantzen MAJ, Silk JB, Fergusen JWH, Wayne RK, Kohn MH (1998) Empirical evaluation of preservation methods for faecal DNA. Mol Ecol 7:1423–1428
Gerloff U, Schlötterer C, Rassmann K, Rambold I, Hohmann G, Fruth B, Tautz D (1995) Amplification of hypervariable simple sequence repeats (microsatellites) from excremental DNA of wild living bonobos (Pan paniscus). Mol Ecol 4:515–518
Goossens B, Chikhi L, Utami SS, de Ruiter J, Bruford MW (2000) A multi-samples multi-extracts approach for microsatellite analysis of faecal samples in an arboreal ape. Conserv Genet 1:157–162
Gorman ML, Jenkins D, Harper RJ (1978) The anal scent sacs of the otter (Lutra lutra). J Zool 186:463–474
Hájková P, Zemanová B, Bryja J, Hájek B, Roche K, Tkadlec E, Zima J (2006) Factors affecting success of PCR amplification of microsatellite loci from otter faeces. Mol Ecol Notes 6:559–562
Hedmark E, Ellegren H (2006) A test of the multiplex pre-amplification approach in microsatellite genotyping of wolverine faecal DNA. Conserv Genet 7:289–293
Idaghdour Y, Broderick D, Korrida A (2003) Faeces as a source of DNA for molecular studies in a threatened population of great bustards. Conser Genet 4:789–792
Jansman HAH, Chanin PRF, Dallas JF (2001) Monitoring otter populations by DNA typing of spraints. IUCN Otter Spec Group Bull 18:12–19
Kohn M, Knauer F, Stoffella A, Schröder W, Pääbo S (1995) Conservation genetics of the European brown bear—a study using excremental PCR of nuclear and mitochondrial sequences. Mol Ecol 4:95–103
Kohn MH, Wayne RK (1997) Facts from feces revisited. Trends Ecol Evol 12:223–227
Kohn MH, York EC, Kamradt DA, Haught G, Sauvajot RM, Wayne RK (1999) Estimating population size by genotyping faeces. Proc R Soc Lond B 266:657–663
Liebich HG (1999) Funktionelle Histologie der Haussäugetiere: Lehrbuch und Farbatlas für Studium und Praxis, 3rd edn. Schattauer, Stuttgart
Macdonald SM, Mason CF (1987) Seasonal marking in an otter population. Acta Theriol 32:449–462
Morin PA, Chambers KE, Boesch C, Vigilant L (2001) Quantitative polymerase chain reaction analysis of DNA from noninvasive samples for accurate microsatellite genotyping of wild chimpanzees (Pan troglodytes versus). Mol Ecol 10:1835–1844
Murphy MA, Waits LP, Kendall KC (2000) Quantitative evaluation of fecal drying methods for brown bear DNA analysis. Wildlife Soc B 28:951–957
Murphy MA, Waits LP, Kendall KC, Wasser SK, Higbee JA, Bogden R (2002) An evaluation of long-term preservation methods for brown bear (Ursus arctos) faecal DNA samples. Conserv Genet 3:435–440
Nsubuga AM, Robbins MM, Roeder AD, Morin PA, Boesch C, Vigilant L (2004) Factors affecting the amount of genomic DNA extracted from ape faeces and the identification of an improved sample storage method. Mol Ecol 13:2089–2094
Palomares F, Godoy A, Piriz A, O’Brien J, Johnson WE (2002) Faecal genetic analysis to determine the presence and distribution of elusive carnivores design and feasibility for the Iberian lynx. Mol Ecol 11:2171–2182
Paxinos E, Mcintosh C, Ralls K, Fleischer R (1997) A noninvasive method for distinguishing among canid species amplification and enzyme restriction of DNA from dung. Mol Ecol 6:483–486
Piggott MP, Taylor AC (2003) Extensive evaluation of faecal preservation and DNA extraction methods in Australian native and introduced species. Aust J Zool 51:341–355
Piggott MP, Bellemain E, Taberlet P, Taylor AC (2004) A multiplex pre-amplification method that significantly improves microsatellite amplification and error rates for faecal DNA in limiting conditions. Conserv Genet 5:417–420
Queller DC, Strassmann JE, Hughes CR (1993) Microsatellites and kinship. Trends Ecol Evol 8:285–288
Reed JZ, Tollit DJ, Thompson PM, Amos W (1997) Molecular scatology the use of molecular genetic analysis to assign species sex and individual identity to seal faeces. Mol Ecol 6:225–234
Roeder AD, Archer FI, Poinar HN, Morin PA (2004) A novel method for collection and preservation of faeces for genetic studies. Mol Ecol Notes 4:761–764
Sidransky D (1992) Identification of rare mutations in the stool of patients with curable colorectable tumors. Science 256:102–105
Taberlet P, Griffin S, Goossens B, Questiau S, Manceau V, Escaravage N, Waits LP, Bouvet J (1996) Reliable genotyping of samples with very low DNA quantities using PCR. Nucleic Acids Res 24:3189–3194
Taberlet P, Luikart G (1999) Non-invasive genetic sampling and individual identification. Biol J Linn Soc 68:41–55
Tikel D, Blair D, Marsh HD (1996) Marine mammal faeces as a source of DNA. Mol Ecol 5:456–457
Tschirch W (1995) Neue biomedizinische Methoden für die Feldforschung - dargestellt am Beispiel des Fischotters (Lutra lutra). In: Stubbe M, Stubbe A, Heidecke D (eds) Methoden der feldökologischen Säugetierforschung (Methods in mammalian field ecology), vol 1. Martin-Luther-Universität, Halle/Saale, pp 203–210
Vigilant L (1999) An evaluation of techniques for the extraction and amplification of DNA from naturally shed hairs. Biol Chem 380:1329–1331
Walsh PS, Metzger DA, Higuchi R (1991) Chelex® 100 as a medium for simple extraction of DNA for PCR-based typing form forensic material. Biotechniques 10:506–513
Wasser SK, Houston CS, Koehler GM, Cadd GG, Fain SR (1997) Techniques for application of faecal DNA methods to field studies of ursids. Mol Ecol 6:1091–1097
Welsch U (2006) Lehrbuch Histologie, 2nd edn. Urban & Fischer, München
Acknowledgements
We would like to thank R. Klenke for helpful discussions and pertinent knowledge about the otter biology. A. Vallentin and U. Hempel are thanked for collecting faecal samples. Thanks to E. Küster and the Department of Bioanalytical Ecotoxicology and also to W. Durka and the Department of Community Ecology for giving access to the laboratories. We are grateful to the members of the Institute of Applied Ecology, University of Canberra for proof-reading the manuscript and to the anonymous reviewers who have greatly contributed to improve a first version of this paper. We kindly acknowledge private landowners for their permission to access the ponds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lampa, S., Gruber, B., Henle, K. et al. An optimisation approach to increase DNA amplification success of otter faeces. Conserv Genet 9, 201–210 (2008). https://doi.org/10.1007/s10592-007-9328-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-007-9328-9