Abstract
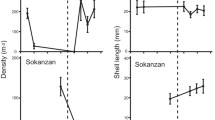
The artificial movement of individuals between populations (translocation) can be an effective way to increase genetic diversity within populations, but few studies have undertaken long term genetic monitoring to determine if variation introduced by translocation is maintained over many generations or whether it can be used to adapt to local conditions. Here, we report on the changes in morphological and molecular variation over a 12-year period in a population of an intertidal littorine snail (Bembicium vittatum) that was created by mixing individuals from three geographically disjunct populations. These source populations differ genetically in shell shape and in allele frequency at several allozyme loci. We found that the translocated population had higher allozyme diversity than any of the source populations and that this pattern was maintained over multiple generations. Variation in shell shape also increased, but this declined over time as shells became taller. Some allozyme loci also showed significant changes in frequency over time. These changes were not consistently towards the genetic makeup of a single source population, and in the case of shell shape, were towards a phenotype that was most suited to the local environment. Our results suggest that genetic variation introduced into a population by translocation can be rapidly incorporated and used to adapt to local conditions without domination by a single source population’s genome. However, more studies are needed before generalisations on the benefits of mixing individuals from disjunct populations can be made.




Similar content being viewed by others
References
Allendorf FW, Luikart G (2006) Conservation and the genetics of populations. Blackwell Publishing, Malden, MA
Black R, Turner SJ, Johnson MS (1994) The early life history of Bembicium vittatum Philippi, 1846 (Gastropoda: Littorinidae). Veliger 37:393–399
Boulding EG (1990) Are the opposing selection pressures on exposed and protected shores sufficient to maintain genetic differentiation between gastropod populations with high intermigration rates? Hydrobiologia 193:41–52
Ellestrand NC, Elam DR (1993) Population genetic consequences of small population size: implications for plant conservation. Ann Rev Ecol Syst 24:217–242
Fenster CB, Galloway LF (2000) Population differentiation in an annual legume: genetic architecture. Evolution 54:1157–1172
Frankham R (2005) Genetics and extinction. Biol Conserv 126:131–140
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to conservation genetics. Cambridge University Press, Cambridge, UK
Gibbs PE (1993) Phenotypic changes in the progeny of Nucella lapillus (Gastropoda) transplanted from an exposed shore to sheltered inlets. J Mollus Stud 59:187–194
Goudet J (2001) FSTAT, a program to estimate and test gene diversities and fixation inices (version 2.9.3). p. Available from <http://www.unil.ch/izea/softwares/fstat.html>
Goudet J, Raymond M, Demeeus T, Rousset F (1996) Testing differentiation in diploid populations. Genetics 144:1933–1940
Gravuer K, von Wettberg E, Schmitt J (2005) Population differentiation and genetic variation inform translocation decisions for Liatris scariosa var. novae-angliae a rare New England grassland perennial. Biol Conserv 124:155–167
Griffith B, Scott JM, Carpenter JW, Reed C (1989) Translocation as a species conservation tool: status and strategy. Science 245:477–480
Huey RB, Gilchrist GW, Carlson ML, Berrigan D, Serra L (2000) Rapid evolution of a geographic cline in size in an introduced fly. Science 287:308–309
Johannesson B, Johannesson K (1996) Population differences in behaviour and morphology in Littorina saxatilis: phenotypic plasticity or genetic differentiation. J Zool 240:475–493
Johannesson K, Johannesson B (1989) Differences in allele frequencies of Aat between high- and mid-rocky shore populations of Littorina saxatilis (Olivi) suggest selection in this enzyme locus. Genet Res 54:7–11
Johannesson K, Johannesson B, Lundgren U (1995) Strong natural selection causes microscale allozyme variation in a marine snail. Proc Natl Acad Sci USA 92:2602–2606
Johannesson K, Tatarenkov A (1997) Allozyme variation in a snail (Littorina saxatilis)—deconfounding the effects of microhabitat and gene flow. Evolution 51:402–409
Johnson MS (1977) Association of allozymes and temperature in the crested blenny Anoplarchus purpurescens. Mar Biol 41:147–152
Johnson MS, Black R (1991) Genetic subdivision of the intertidal snail Bembicium vittatum (Gastropda: Littorinidae) varies with habitat in the Houtman Abrolhos Islands. Heredity 67:205–213
Johnson MS, Black R (1996) Geographic cohesiveness versus associations with habitat: genetic subdivision of Bembicium vittatum Philippi (Gastropoda: Littorinidae) in the Houtman-Abrolhos Islands. Biol J Linn Soc 58:57–74
Johnson MS, Black R (1998) Effects of habitat on growth and shape of contrasting phenotypes of Bembicium vittatum Philippi in the Houtman Abrolhos Islands, Western Australia. Hydrobiologia 378:95–103
Johnson MS, Black R (2000) Associations with habitat versus geographic cohesiveness: size and shape of Bembicium vittatum Philippi (Gastropoda: Littorinidae) in the Houtman Abrolhos Islands. Biol J Linn Soc 71:563–580
Kemp P, Bertness MD (1984) Snail shape and growth rates: evidence for plastic shell allometry in Littorina littorea. Proc Natl Acad Sci USA 81:811–813
Krauss SL, Dixon B, Dixon KW (2002) Rapid genetic decline in a translocated population of the endangered plant Grevillea scapigera. Conserv Biol 16:986–994
Lively CM, Craddock C, Vrijenhoek RC (1990) The Red Queen hypothesis supported by parasitism in sexual and clonal fish. Nature 344:864–866
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer Associates, Sunderland, MA
Madsen T, Shine R, Olsson M, Wittzells H (1999) Conservation biology: restoration of an inbred adder population. Nature 402:34–35
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Newkirk GF, Doyle RW (1975) Genetic analysis of shell-shape variation in Littorina saxatilis on an environmental cline. Mar Biol 30:227–237
Oakeshott JG, Chambers GK, Gibson JB, Eanes WF, Willcocks DA (1983) Geographic variation in G6pd and Pgd allele frequencies in Drosophila melanogaster. Heredity 50:67–72
Parsons KE (1996) The genetic effects of larval dispersal depend on spatial scale and habitat characteristics. Mar Biol 126:403–414
Parsons KE (1997) Contrasting patterns of heritable geographic variation in shell morphology and growth potential in the marine gastropod Bembicium vittatum: evidence from field experiments. Evolution 51:784–796
Piccino P, Viard F, Sarradin P, Le Bris N, Le Guen D, Jollivet D (2004) Thermal selection of Pgm allozymes in newly founded populations of the thermotolerant vent polychaete Alvinella pompejana. Proc R Soc B 271:2351–2359
Rolán-Alvarez E, Johannesson K, Erlandsson J (1997) The maintenance of a cline in the marine snail Littorina saxatilis: the role of home site advantage and hybrid fitness. Evolution 51:1838–1847
Schmidt PS, Rand DM (1999) Intertidal microhabitat and selection at Mpi: interlocus contrasts in the northern acorn barnacle, Semibalanus balanoides. Evolution 53:135–156
Sezgin E, Duvernell DD, Matzkin LM, Duan Y, Zhu C, Verrelli BC, Eanes WF (2004) Single-locus latitudinal clines and their relationship to temperate adaptation in metabolic genes and derived alleles in Drosophila melanogaster. Genetics 168:923–931
Stockwell C, Weeks S (1999) Translocations and rapid evolutionary responses in recently established populations of western mosquitofish (Gambusia affinis). Anim Conserv 2:103–110
Storfer A (1999) Gene flow and endangered species translocations: a topic revisited. Biol Conserv 87:173–180
Trussell GC (2000) Predator-induced plasticity and morphological trade-offs in latitudinally separated populations of Littorina obtusata. Evol Ecol 2:803–822
Trussell GC (2002) Evidence of countergradient variation in the growth of an intertidal snail in response to water velocity. Mar Ecol Prog Ser 243:123–131
Trussell GC, Johnson AS, Rudolph SG, Gilfillian ES (1993) Resistance to dislodgement: habitat and size-specific differences in morphology and tenacity in an intertidal snail. Mar Ecol Prog Ser 100:135–144
Van Zant JL, Wooten MC (2002) Translocation of Choctawhatchee beach mice (Peromyscus polionotus allophrys): hard lessons learned. Biol Conserv 112:405–413
Verrelli BC, Eanes WF (2000) Extensive amino acid polymorphism at the Pgm locus is consistent with adaptive protein evolution in Drosophila melanogaster. Genetics 156:1737–1752
Verrelli BC, Eanes WF (2001) The functional impact of Pgm amino acid polymorphism on glycogen content in Drosophila melanogaster. Genetics 159:201–210
Vilà C, Sundqvist A-K, Flagstad Ø, Seddon J, Björnerfeldt S, Kojola I, Casulli A, Sand H, Wabakken P, Ellegren H (2003) Rescue of a severely bottlenecked wolf (Canis lupus) population by a single immigrant. Proc R Soc B 270:91–97
Vrijenhoek RC (1996) Conservation genetics of North American desert fishes. In: Aj C, Hamrick JL (eds) Conservation genetics: case histories from nature. Chapman and Hall, New York, pp. 367–397
Westemeier RL, Brawn JD, Simpson SA, Esker TL, Jansen RW, Walk JW, Kershner EL, Bouzat JL, Paige KN (1998) Tracking the long-term decline and recovery of an isolated population. Science 282:1695–1698
Acknowledgements
This work was supported by a grant from the Australian Research Council and a University of Western Australia postdoctoral fellowship awarded to W.J.K. We thank Dr Karen Parsons for providing the pre-2005 samples and details of the establishment of the Fremantle population. We also thank members of the population genetics group and two anonymous reviewers for their helpful comments on earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Binks, R.M., Kennington, W.J. & Johnson, M.S. Rapid evolutionary responses in a translocated population of intertidal snail (Bembicium vittatum) utilise variation from different source populations. Conserv Genet 8, 1421–1429 (2007). https://doi.org/10.1007/s10592-007-9293-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-007-9293-3