Abstract
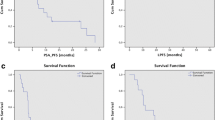
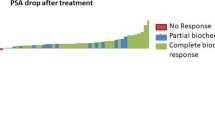
To assess the outcomes of a cohort of bone oligometastatic prostate cancer patients treated with PSMA-PET guided stereotactic body radiotherapy (SBRT). From April 2017 to January 2021, 40 patients with oligorecurrent prostate cancer detected by PSMA-PET were treated with SBRT for bone oligometastases. Concurrent androgen deprivation therapy was an exclusion criterion. A total of 56 prostate cancer bone oligometastases were included in the present analysis. In 28 patients (70%), oligometastatic disease presented as a single lesion, two lesions in 22.5%, three lesions in 5%, four lesions in 2.5%. 30.3% were spine-metastases, while 69.7% were non-spine metastases. SBRT was delivered for a median dose of 30 Gy (24–40 Gy) in 3–5 fractions, with a median EQD2 = 85 Gy2 (64.3—138.9Gy2). With a median follow-up of 22 months (range 2–48 months), local control (LC) 1- and 2-years rates were 96.3% and 93.9%, while distant progression-free survival (DPFS) rates were 45.3% and 27%. At multivariate analysis, the lower PSA nadir value after SBRT remained significantly related to better DPFS rates (p = 0.03). In 7 patients, a second SBRT course was proposed with concurrent ADT, while 11 patients, due to polymetastatic spread, received ADT alone, resulting in 1- and 2-years ADT-free survival rates of 67.5% and 61.8%. At multivariate analysis, a lower number of treated oligometastases maintained a correlation with higher ADT-free survival rates (p = 0.04). In our experience, PSMA-PET guided SBRT resulted in excellent results in terms of clinical outcomes, representing a helpful tool with the aim to delay the start of ADT.

Similar content being viewed by others
References
Testa U, Castelli G, Pelosi E (2019) Cellular and molecular mechanisms underlying prostate cancer development: therapeutic implications. Medicines 6(3):82. https://doi.org/10.3390/medicines6030082
Haffner MC, Zwart W, Roudier MP, True LD, Nelson WG, Epstein JI, De Marzo AM, Nelson PS, Yegnasubramanian S (2021) Genomic and phenotypic heterogeneity in prostate cancer. Nat Rev Urol 18(2):79–92. https://doi.org/10.1038/s41585-020-00400-w
Chalkidou A, Macmillan T, Grzeda MT, Peacock J, Summers J, Eddy S, Coker B, Patrick H, Powell H, Berry L, Webster G, Ostler P, Dickinson PD, Hatton MQ, Henry A, Keevil S, Hawkins MA, Slevin N, van As N (2021) Stereotactic ablative body radiotherapy in patients with oligometastatic cancers: a prospective, registry-based, single-arm, observational, evaluation study. Lancet Oncol 22(1):98–106. https://doi.org/10.1016/S1470-2045(20)30537-4
Guckenberger M, Lievens Y, Bouma AB, Collette L, Dekker A, deSouza NM, Dingemans AC, Fournier B, Hurkmans C, Lecouvet FE, Meattini I, Méndez Romero A, Ricardi U, Russell NS, Schanne DH, Scorsetti M, Tombal B, Verellen D, Verfaillie C, Ost P (2020) Characterisation and classification of oligometastatic disease: a European society for radiotherapy and oncology and European organisation for research and treatment of cancer consensus recommendation. Lancet Oncol 21(1):e18–e28. https://doi.org/10.1016/S1470-2045(19)30718-1
Ost P, Reynders D, Decaestecker K, Fonteyne V, Lumen N, De Bruycker A, Lambert B, Delrue L, Bultijnck R, Claeys T, Goetghebeur E, Villeirs G, De Man K, Ameye F, Billiet I, Joniau S, Vanhaverbeke F, De Meerleer G (2018) Surveillance or metastasis-directed therapy for oligometastatic prostate cancer recurrence: a prospective, randomized, multicenter phase II trial. J Clin Oncol 36(5):446–453. https://doi.org/10.1200/JCO.2017.75.4853
Phillips R, Shi WY, Deek M, Radwan N, Lim SJ, Antonarakis ES, Rowe SP, Ross AE, Gorin MA, Deville C, Greco SC, Wang H, Denmeade SR, Paller CJ, Dipasquale S, DeWeese TL, Song DY, Wang H, Carducci MA, Pienta KJ, Pomper MG, Dicker AP, Eisenberger MA, Alizadeh AA, Diehn M, Tran PT (2020) Outcomes of observation vs stereotactic ablative radiation for oligometastatic prostate cancer: the ORIOLE phase 2 randomized clinical trial. JAMA Oncol 6(5):650–659. https://doi.org/10.1001/jamaoncol.2020.0147
Halabi S, Kelly WK, Ma H, Zhou H, Solomon NC, Fizazi K, Tangen CM, Rosenthal M, Petrylak DP, Hussain M, Vogelzang NJ, Thompson IM, Chi KN, de Bono J, Armstrong AJ, Eisenberger MA, Fandi A, Li S, Araujo JC, Logothetis CJ, Quinn DI, Morris MJ, Higano CS, Tannock IF, Small EJ (2016) Meta-analysis evaluating the impact of site of metastasis on overall survival in men with castration-resistant prostate cancer. J Clin Oncol 34(14):1652–1659. https://doi.org/10.1200/JCO.2015.65.7270
Ost P, Decaestecker K, Lambert B, Fonteyne V, Delrue L, Lumen N, Ameye F, De Meerleer G (2014) Prognostic factors influencing prostate cancer-specific survival in non-castrate patients with metastatic prostate cancer. Prostate 74(3):297–305. https://doi.org/10.1002/pros.22750
Cancer Genome Atlas Research Network (2015) The molecular taxonomy of primary prostate cancer. Cell 163(4):1011–1025. https://doi.org/10.1016/j.cell.2015.10.025
Ku SY, Gleave ME, Beltran H (2019) Towards precision oncology in advanced prostate cancer. Nat Rev Urol 16(11):645–654. https://doi.org/10.1038/s41585-019-0237-8
Evans R, Loeb A, Kaye KS, Cher ML, Martin ET (2017) Infection-related hospital admissions after prostate biopsy in United States men. Open Forum Infect Dis. 4(1):ofw265
Mullane SA, Van Allen EM (2016) Precision medicine for advanced prostate cancer. Curr Opin Urol 26(3):231–239. https://doi.org/10.1097/MOU.0000000000000278.PMID:26909474;PMCID:PMC4955574
Mazzoni A, Oddo CM, Valle G, Camboni D, Strauss I, Barbaro M, Barabino G, Puddu R, Carboni C, Bisoni L, Carpaneto J, Vecchio F, Petrini FM, Romeni S, Czimmermann T, Massari L, di Iorio R, Miraglia F, Granata G, Pani D, Stieglitz T, Raffo L, Rossini PM, Micera S (2020) Morphological neural computation restores discrimination of naturalistic textures in trans-radial amputees. Sci Rep 10(1):527. https://doi.org/10.1038/s41598-020-57454-4.Erratum.In:SciRep.2021Aug11;11(1):16662.PMID:31949245;PMCID:PMC6965126
Cox BW, Spratt DE, Lovelock M, Bilsky MH, Lis E, Ryu S, Sheehan J, Gerszten PC, Chang E, Gibbs I, Soltys S, Sahgal A, Deasy J, Flickinger J, Quader M, Mindea S, Yamada Y (2012) International spine radiosurgery consortium consensus guidelines for target volume definition in spinal stereotactic radiosurgery. Int J Radiat Oncol Biol Phys 83(5):e597-605. https://doi.org/10.1016/j.ijrobp.2012.03.009
Sahgal A, Chang JH, Ma L, Marks LB, Milano MT, Medin P, Niemierko A, Soltys SG, Tomé WA, Wong CS, Yorke E, Grimm J, Jackson A (2019) Spinal cord dose tolerance to stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys S0360–3016(19):33862–33863. https://doi.org/10.1016/j.ijrobp.2019.09.038
Benedict SH, Yenice KM, Followill D, Galvin JM, Hinson W, Kavanagh B, Keall P, Lovelock M, Meeks S, Papiez L, Purdie T, Sadagopan R, Schell MC, Salter B, Schlesinger DJ, Shiu AS, Solberg T, Song DY, Stieber V, Timmerman R, Tomé WA, Verellen D, Wang L, Yin FF (2010) Stereotactic body radiation therapy: the report of AAPM Task Group 101. Med Phys 37(8):4078–4101. https://doi.org/10.1118/1.3438081
Moraes FY, Chen X, Yan M et al (2020) Evolving role of stereotactic body radiation therapy in the management of spine metastases: defining dose and dose constraints. Neurosurg Clin N Am 31(2):167–189. https://doi.org/10.1016/j.nec.2019.12.001
Wang N, Docherty FE, Brown HK, Reeves KJ, Fowles AC, Ottewell PD, Dear TN, Holen I, Croucher PI, Eaton CL (2014) Prostate cancer cells preferentially home to osteoblast-rich areas in the early stages of bone metastasis: evidence from in vivo models. J Bone Miner Res 29(12):2688–2696. https://doi.org/10.1002/jbmr.2300
Zhang X (2019) Interactions between cancer cells and bone microenvironment promote bone metastasis in prostate cancer. Cancer Commun (Lond) 39(1):76. https://doi.org/10.1186/s40880-019-0425-1
Onal C, Ozyigit G, Akgun Z, Atalar B, Igdem S, Oymak E, Agaoglu F, Selek U, Guler OC, Hurmuz P, Mustafayev TZ, Akyol F (2021) Oligometastatic bone disease in castration-sensitive prostate cancer patients treated with stereotactic body radiotherapy using 68Ga-PSMA PET/CT: TROD 09–004 study. Clin Nucl Med 46(6):465–470. https://doi.org/10.1097/RLU.0000000000003558
Rogowski P, Trapp C, von Bestenbostel R, Schmidt-Hegemann NS, Shi R, Ilhan H, Kretschmer A, Stief C, Ganswindt U, Belka C, Li M (2021) Outcomes of metastasis-directed therapy of bone oligometastatic prostate cancer. Radiat Oncol 16(1):125. https://doi.org/10.1186/s13014-021-01849-8
Mazzola R, Francolini G, Triggiani L, Napoli G, Cuccia F, Nicosia L, Livi L, Magrini SM, Salgarello M, Alongi F (2021) Metastasis-directed therapy (SBRT) guided by PET-CT 18F-choline versus PET-CT 68Ga-PSMA in castration-sensitive oligorecurrent prostate cancer: a comparative analysis of effectiveness. Clin Genitourin Cancer 19(3):230–236. https://doi.org/10.1016/j.clgc.2020.08.002
Alongi F, Fersino S, GiajLevra N, Mazzola R, Ricchetti F, Fiorentino A, Ruggieri R, Malfatti V, Cavalleri S, Salgarello M (2015) Impact of 18F-Choline PET/CT in the decision-making strategy of treatment volumes in definitive prostate cancer volumetric modulated radiation therapy. Clin Nucl Med 40(11):e496-500. https://doi.org/10.1097/RLU.0000000000000841
Funding
No fundings received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mazzola, R., Cuccia, F., Pastorello, E. et al. PSMA-guided metastases directed therapy for bone castration sensitive oligometastatic prostate cancer: a multi-institutional study. Clin Exp Metastasis 39, 443–448 (2022). https://doi.org/10.1007/s10585-022-10157-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-022-10157-8