Abstract
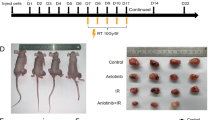
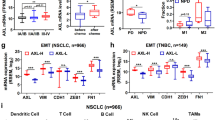
Metastatic recurrence after cancer treatments with radiation, cancer drugs, or even immunotherapeutic agents (cytokine, antibody, lymphocyte etc.) is often intractable and fatal for cancer patients. Therefore, molecular understanding of metastatic recurrence is necessary. Recently, these recurrent and metastatic tumor cells with resistance to cancer drugs have been reported to possess stem-like attributes and epithelial-mesenchymal transition (EMT) phenotype. Previously, we also found that antigen-specific cytotoxic T lymphocyte (CTL)-mediated immunotherapy conferred tumor cells with immune-resistant and stem-like phenotypes by hyper-activating NANOG/TCL1/AKT signaling axis. In this study, we report that these immunoedited cells have high metastatic capability and phenotypes. These cells exhibit enhanced migration, infiltration, and invasiveness in vitro as well as formation of metastatic lung nodules in vivo. Moreover, they display EMT-like features characterized by increased expression of BMI1 and TWIST1. Importantly, these pleiotropic phenotypes of metastasis through the expression of the EMT-associated molecules were critically dependent on the NANOG/TCL1A/AKT signaling axis, which was also conserved across multiple types of human cancer. Thus, we provide proof of the principle that inhibition of the NANOG axis is an effective strategy to control metastasis of immunoedited cancer, particularly, after CTL-based immunotherapy.





Similar content being viewed by others
Abbreviations
- EMT:
-
Epithelial-mesenchymal transition
- CSC:
-
Cancer stem cell
- CTL:
-
Cytotoxic T lymphocyte
- TIC:
-
Tumor initiating cell
References
Brabletz T (2012) To differentiate or not–routes towards metastasis. Nat Rev Cancer 12(6):425–436. doi:10.1038/nrc3265
Knutson KL, Lu H, Stone B, Reiman JM, Behrens MD, Prosperi CM, Gad EA, Smorlesi A, Disis ML (2006) Immunoediting of cancers may lead to epithelial to mesenchymal transition. Journal of immunology 177(3):1526–1533
Enderling H, Hlatky L, Hahnfeldt P (2012) Immunoediting: evidence of the multifaceted role of the immune system in self-metastatic tumor growth. Theor Biol Med Model 9:31. doi:10.1186/1742-4682-9-31
Kudo-Saito C, Shirako H, Takeuchi T, Kawakami Y (2009) Cancer metastasis is accelerated through immunosuppression during Snail-induced EMT of cancer cells. Cancer Cell 15(3):195–206. doi:10.1016/j.ccr.2009.01.023
Tsai JH, Donaher JL, Murphy DA, Chau S, Yang J (2012) Spatiotemporal regulation of epithelial-mesenchymal transition is essential for squamous cell carcinoma metastasis. Cancer Cell 22(6):725–736. doi:10.1016/j.ccr.2012.09.022
Thiery JP, Acloque H, Huang RY, Nieto MA (2009) Epithelial-mesenchymal transitions in development and disease. Cell 139(5):871–890. doi:10.1016/j.cell.2009.11.007
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, Campbell LL, Polyak K, Brisken C, Yang J, Weinberg RA (2008) The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133(4):704–715. doi:10.1016/j.cell.2008.03.027
Liu M, Casimiro MC, Wang C, Shirley LA, Jiao X, Katiyar S, Ju X, Li Z, Yu Z, Zhou J, Johnson M, Fortina P, Hyslop T, Windle JJ, Pestell RG (2009) p21CIP1 attenuates Ras- and c-Myc-dependent breast tumor epithelial mesenchymal transition and cancer stem cell-like gene expression in vivo. Proc Natl Acad Sci USA 106(45):19035–19039. doi:10.1073/pnas.0910009106
Brabletz T, Jung A, Spaderna S, Hlubek F, Kirchner T (2005) Opinion: migrating cancer stem cells: an integrated concept of malignant tumour progression. Nat Rev Cancer 5(9):744–749. doi:10.1038/nrc1694
Polyak K, Weinberg RA (2009) Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer 9(4):265–273. doi:10.1038/nrc2620
Zhang Y, Wang Z, Yu J, Shi J, Wang C, Fu W, Chen Z, Yang J (2012) Cancer stem-like cells contribute to cisplatin resistance and progression in bladder cancer. Cancer Lett 322(1):70–77. doi:10.1016/j.canlet.2012.02.010
Dave B, Mittal V, Tan NM, Chang JC (2012) Epithelial-mesenchymal transition, cancer stem cells and treatment resistance. Breast Cancer Res BCR 14(1):202. doi:10.1186/bcr2938
Facchino S, Abdouh M, Chatoo W, Bernier G (2010) BMI1 confers radioresistance to normal and cancerous neural stem cells through recruitment of the DNA damage response machinery. J Neurosci 30(30):10096–10111. doi:10.1523/jneurosci.1634-10.2010
Watanabe M, Ohnishi Y, Inoue H, Wato M, Tanaka A, Kakudo K, Nozaki M (2014) NANOG expression correlates with differentiation, metastasis and resistance to preoperative adjuvant therapy in oral squamous cell carcinoma. Oncol Lett 7(1):35–40. doi:10.3892/ol.2013.1690
Ocana OH, Nieto MA (2010) Epithelial plasticity, stemness and pluripotency. Cell Res 20(10):1086–1088. doi:10.1038/cr.2010.127
Wang ML, Chiou SH, Wu CW (2013) Targeting cancer stem cells: emerging role of Nanog transcription factor. OncoTarg Ther 6:1207–1220. doi:10.2147/ott.s38114
Lin T, Ding YQ, Li JM (2012) Overexpression of Nanog protein is associated with poor prognosis in gastric adenocarcinoma. Med Oncol 29(2):878–885. doi:10.1007/s12032-011-9860-9
Xu F, Dai C, Zhang R, Zhao Y, Peng S, Jia C (2012) Nanog: a potential biomarker for liver metastasis of colorectal cancer. Dig Dis Sci 57(9):2340–2346. doi:10.1007/s10620-012-2182-8
Chiou SH, Wang ML, Chou YT, Chen CJ, Hong CF, Hsieh WJ, Chang HT, Chen YS, Lin TW, Hsu HS, Wu CW (2010) Coexpression of Oct4 and Nanog enhances malignancy in lung adenocarcinoma by inducing cancer stem cell-like properties and epithelial-mesenchymal transdifferentiation. Cancer Res 70(24):10433–10444. doi:10.1158/0008-5472.can-10-2638
Siu MK, Wong ES, Kong DS, Chan HY, Jiang L, Wong OG, Lam EW, Chan KK, Ngan HY, Le XF, Cheung AN (2013) Stem cell transcription factor NANOG controls cell migration and invasion via dysregulation of E-cadherin and FoxJ1 and contributes to adverse clinical outcome in ovarian cancers. Oncogene 32(30):3500–3509. doi:10.1038/onc.2012.363
Hallermalm K, Seki K, De Geer A, Motyka B, Bleackley RC, Jager MJ, Froelich CJ, Kiessling R, Levitsky V, Levitskaya J (2008) Modulation of the tumor cell phenotype by IFN-gamma results in resistance of uveal melanoma cells to granule-mediated lysis by cytotoxic lymphocytes. J Immunol 180(6):3766–3774
Schreiber RD, Old LJ, Smyth MJ (2011) Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science (New York, NY) 331(6024):1565–1570. doi:10.1126/science.1203486
Noh KH, Kim BW, Song KH, Cho H, Lee YH, Kim JH, Chung JY, Kim JH, Hewitt SM, Seong SY, Mao CP, Wu TC, Kim TW (2012) Nanog signaling in cancer promotes stem-like phenotype and immune evasion. J Clin Investig 122(11):4077–4093. doi:10.1172/jci64057
Noh KH, Lee YH, Jeon JH, Kang TH, Mao CP, Wu TC, Kim TW (2012) Cancer vaccination drives Nanog-dependent evolution of tumor cells toward an immune-resistant and stem-like phenotype. Cancer Res 72(7):1717–1727. doi:10.1158/0008-5472.can-11-3758
Noh KH, Kim SH, Kim JH, Song KH, Lee YH, Kang TH, Han HD, Sood AK, Ng J, Kim K, Sonn CH, Kumar V, Yee C, Lee KM, Kim TW (2014) API5 confers tumoral immune escape through FGF2-dependent cell survival pathway. Cancer Res 74(13):3556–3566. doi:10.1158/0008-5472.can-13-3225
Cheng WF, Hung CF, Lin KY, Ling M, Juang J, He L, Lin CT, Wu TC (2003) CD8+ T cells, NK cells and IFN-gamma are important for control of tumor with downregulated MHC class I expression by DNA vaccination. Gene Ther 10(16):1311–1320. doi:10.1038/sj.gt.3301982
Puisieux A, Brabletz T, Caramel J (2014) Oncogenic roles of EMT-inducing transcription factors. Nat Cell Biol 16(6):488–494. doi:10.1038/ncb2976
Yang MH, Hsu DS, Wang HW, Wang HJ, Lan HY, Yang WH, Huang CH, Kao SY, Tzeng CH, Tai SK, Chang SY, Lee OK, Wu KJ (2010) Bmi1 is essential in Twist1-induced epithelial-mesenchymal transition. Nat Cell Biol 12(10):982–992. doi:10.1038/ncb2099
Tania M, Khan MA, Fu J (2014) Epithelial to mesenchymal transition inducing transcription factors and metastatic cancer. Tumour Biol 35(8):7335–7342. doi:10.1007/s13277-014-2163-y
Noguchi M, Ropars V, Roumestand C, Suizu F (2007) Proto-oncogene TCL1: more than just a coactivator for Akt. FASEB J 21(10):2273–2284. doi:10.1096/fj.06-7684com
Way TD, Huang JT, Chou CH, Huang CH, Yang MH, Ho CT (2014) Emodin represses TWIST1-induced epithelial-mesenchymal transitions in head and neck squamous cell carcinoma cells by inhibiting the beta-catenin and Akt pathways. Eur J Cancer 50(2):366–378. doi:10.1016/j.ejca.2013.09.025
Du R, Xia L, Ning X, Liu L, Sun W, Huang C, Wang H, Sun S (2014) Hypoxia-induced Bmi1 promotes renal tubular epithelial cell-mesenchymal transition and renal fibrosis via PI3 K/Akt signal. Mol Biol Cell 25(17):2650–2659. doi:10.1091/mbc.E14-01-0044
Hasmim M, Noman MZ, Lauriol J, Benlalam H, Mallavialle A, Rosselli F, Mami-Chouaib F, Alcaide-Loridan C, Chouaib S (2011) Hypoxia-dependent inhibition of tumor cell susceptibility to CTL-mediated lysis involves NANOG induction in target cells. J Immunol 187(8):4031–4039. doi:10.4049/jimmunol.1101011
Lu X, Mazur SJ, Lin T, Appella E, Xu Y (2014) The pluripotency factor nanog promotes breast cancer tumorigenesis and metastasis. Oncogene 33(20):2655–2664. doi:10.1038/onc.2013.209
Meng HM, Zheng P, Wang XY, Liu C, Sui HM, Wu SJ, Zhou J, Ding YQ, Li J (2010) Over-expression of Nanog predicts tumor progression and poor prognosis in colorectal cancer. Cancer Biol Ther 9(4):295–302. doi:10.4161/cbt.9.4.10666
Tiwari N, Gheldof A, Tatari M, Christofori G (2012) EMT as the ultimate survival mechanism of cancer cells. Semin Cancer Biol 22(3):194–207. doi:10.1016/j.semcancer.2012.02.013
Chang B, Li S, He Q, Liu Z, Zhao L, Zhao T (1840) Wang A (2014) Deregulation of Bmi-1 is associated with enhanced migration, invasion and poor prognosis in salivary adenoid cystic carcinoma. Biochim Biophys Acta 12:3285–3291. doi:10.1016/j.bbagen.2014.08.005
Xie X, Piao L, Cavey GS, Old M, Teknos TN, Mapp AK, Pan Q (2014) Phosphorylation of Nanog is essential to regulate Bmi1 and promote tumorigenesis. Oncogene 33(16):2040–2052. doi:10.1038/onc.2013.173
Kim D, Kim S, Koh H, Yoon SO, Chung AS, Cho KS, Chung J (2001) Akt/PKB promotes cancer cell invasion via increased motility and metalloproteinase production. FASEB J 15(11):1953–1962. doi:10.1096/fj.01-0198.com
Nacerddine K, Beaudry JB, Ginjala V, Westerman B, Mattiroli F, Song JY, van der Poel H, Ponz OB, Pritchard C, Cornelissen-Steijger P, Zevenhoven J, Tanger E, Sixma TK, Ganesan S, van Lohuizen M (2012) Akt-mediated phosphorylation of Bmi1 modulates its oncogenic potential, E3 ligase activity, and DNA damage repair activity in mouse prostate cancer. J Clin Invest 122(5):1920–1932. doi:10.1172/jci57477
Acknowledgments
This work was supported by NRF grants funded by the MEST, Republic of Korea (2012R1A6A3A010105372 2012R1A2A2A01007527 and 2013M3A9D3045881, and 2014R1A2A1A10054205), and Korea University Grant.
Conflict of interest
No potential conflicts of interest were disclosed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hyo-Jung Lee and Kyung Hee Noh have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, HJ., Noh, K.H., Lee, YH. et al. NANOG signaling promotes metastatic capability of immunoedited tumor cells. Clin Exp Metastasis 32, 429–439 (2015). https://doi.org/10.1007/s10585-015-9717-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-015-9717-2