Abstract
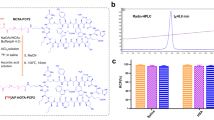
Metallic nanoparticles, because of their size, chemical and physical properties, are particularly attractive as therapeutic probes in treating cancer. Central to any clinical advances in nanoparticulate based therapy will be to produce hybrid nanoparticles that can be targeted to vascular, extracellular or cell surface receptors. Development of hybrid nanoparticles that specifically target cancer vasculature has received considerable attention. Most cancers have leaky vasculature and the defective vascular architecture, created due to the rapid vascularization necessary to serve fast growing cancers, in combination with poor lymphatic drainage allows increased permeation and retention effects. The leaky vasculature, because of higher porosity and permeability, serve as natural high affinity targets to metallic nanoparticles. Another attractive approach toward the application of nanotechnology to nanomedicine is the utility of nanoparticles that display inherent therapeutic properties. For example radioactive gold nanoparticles present attractive prospects in therapy of cancer. The radioactive properties of Au-198 (βmax=0.96 MeV; t1/2=2.7 d) and Au-199 (βmax=0.46 MeV; t1/2=3.14 d) make them ideal candidates for use in radiotherapeutic applications. In addition, they both have imageable gamma emissions for dosimetry and pharmacokinetic studies and Au-199 can be made carrier-free by indirect methods. Gold nanoparticles are of interest for treatment of disease as they can deliver agents directly into cells and cellular components with a higher concentration of radioactivity, e.g. higher dose of radioactivity, to cancerous tumor cells.
Similar content being viewed by others
References
Balogh L., Nigavekar S. S., Cook A. T., Minc L., and Khan M. K.: Abstracts of Papers of the American Chemical Society 225 (2003) U270.
Bielinska A., Eichman J. D., Lee I., Baker J. R., and Balogh L.: Journal of Nanoparticle Research 4 (2002) 395.
Nigavekar S. S., Balogh L. P., Cook A. C., Minc L. and Khan M. K.: PharmaChem (2003) 94–99.
J. F. Hainfeld, D. N. Slatkin, and H. M. Smilowitz: Physics in Medicine and Biology 49 (2004) N309-N315.
Hainfeld J. F. and Powell R. D.: Journal of Histochemistry & Cytochemistry 48 (2000) 471.
Hainfeld J. F. and Robinson J. M.: Journal of Histochemistry & Cytochemistry 48 (2000) 459.
Hainfeld J.F., Powell R. D., and Hacker G.W.: Nanobiotechnology (Eds. C. M. Niemeyer and Mirkin, C.A.), Wiley-Vch, Germany, 2004 p. 353–386.
Dilmanian F. A., Morris G. M., Zhong N., Bacarian T., Hainfeld J. F., Kalef-Ezra J., Brewington L. J., Tammam J., and Rosen E. M.: Radiation Research 159 (2003) 632.
Billinghurst M.: In: Radiolabeled and Magnetic Particulates in Medicine & Biology; Vol. 3, edited by R. Arshady (2001), p. 149–76.
Alfonso A., Hassan A., Gardner B., Stein S., Patti J., Solomon N., McCarthy J., and Steigman J.: Cancer Res 38 (1978) 2740.
Ashamalla R. S. H., Zaki B, Ikoro N.C., Ross P.: Brachytherapy 1 (2002) 161.
Emery J. F., Leddicotte G. W., Radiochemistry of Gold ((National Academy of Sciences—National Research Council) NAS-NS-3036, 1961).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Katti, K.V., Kannan, R., Katti, K. et al. Hybrid gold nanoparticles in molecular imaging and radiotherapy. Czech J Phys 56, D23–D34 (2006). https://doi.org/10.1007/s10582-006-1033-2
Issue Date:
DOI: https://doi.org/10.1007/s10582-006-1033-2