Abstract
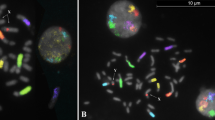
Causes of chromosomal differences such as mosaicism between embryos developed in vivo and in vitro may be resolved using animal models to compare embryos generated in vivo with those generated by different production systems. The aims of this study were: (1) to test a ZOO-FISH approach (using bovine painting probes) to detect abnormal chromosome make-up in the sheep embryo model, and (2) to examine the extent of chromosome deviation in sheep embryos derived in vivo and in vitro. Cytogenetic analysis was performed on day 6 in-vivo and in-vitro derived sheep embryos using commercially available bovine chromosome painting probes for sex chromosomes X–Y and autosomes 1–29. A total of 8631 interphase and metaphase nuclei were analyzed from 49 in-vitro-derived and 51 in-vivo-derived embryos. The extent of deviation from normal ovine chromosome make-up was higher (p < 0.05) in in-vitro-produced embryos relative to in-vivo-derived embryos (65.3% vs. 19.6% respectively) mainly due to diploid–polyploid mosaicism. Polyploid cells ranged from 3n to 8n with tetraploids most predominant among non-diploid cells. The proportions of polyploid cells per mixoploid embryo in in-vitro-produced embryos ranged from 1.4% to 30.3%, in contrast to less than 10% among the in-vivo-derived embryos. It was concluded that in-vitro-derived embryos are vulnerable to ploidy change compared to their in-vivo counterparts. The application of ZOO-FISH to domestic animal embryos is an effective approach to study the chromosome complement of species for which DNA probes are unavailable.
Similar content being viewed by others
References
Alexander B, Coppola G, Di Berardino D et al. (2006) The effect of 6-dimethylaminopurine (6-DMAP) and cycloheximide (CHX) on the development and chromosomal complement of sheep parthenogenetic and nuclear transfer embryos. Mol Reprod Dev 73: 20–30.
Ansari HA, Bosma AA, Broad TE et al. (1999) Standard C-Q and R-banded ideograms of the domestic sheep (Ovis aries): homology with cattle (Bos taurus). Report of the Committee for the Standardization of the Sheep Karyotype. Cytogenet Cell Genet 85: 317–324.
Bean CJ, Hassold TJ, Judis L, Hunt PA (2002) Fertilization in vitro increases non disjunction during early cleavage divisions in a mouse model system. Hum Reprod 17: 2362–2367.
Bielanska M, Tan SL, Ao A (2002) Chromosomal mosaicism throughout human preimplantation development in vitro: incidence, type and relevance to embryo outcome. Hum Reprod 17: 413–419.
Bielanska M, Jin S, Bernier M, Tan SL, Ao A (2005) Diploid–aneuploid mosaicism in human embryos cultured to the blastocyst stage. Fertil Steril 84: 336–342.
Bond DJ, Chandley AC (1983) The origins and causes of aneuploidy in experimental organisms. In Aneuploidy. Oxford: Oxford University Press, pp. 27–54.
Boue A, Boue J, Gropp A (1985) Cytogenetics of pregnancy wastage. Adv Hum Genet 14: 1–57.
Crozet N (1993) Fertilization in vivo and in vitro. In Thibault C, Lavasseur MC, Hunter RHF, eds., Reproduction in Mammals and Man. Paris: Ellipses, pp. 328–347.
Delhanty JD, Harper JC, Ao A, Handyside AH, Winston RM (1997) Multicolour FISH detects frequent chromosomal mosaicism and chaotic division in normal preimplantation embryos from fertile patients. Hum Genet 99: 755–760.
Derhaag JG, Coonen E, Bras M et al. (2003) Chromosomally abnormal cells are not selected for the extra-embryonic compartment of the human preimplantation embryo at the blastocyst stage. Hum Reprod 18: 2565–2574.
Di Berardino D, Vozdova M, Kubickova S et al. (2004) Sexing river buffalo (Bubalus bubalis L.), sheep (Ovis aries L.), goat (Capra hircus L.), and cattle spermatozoa by double color FISH using bovine (Bos taurus L.) X- and Y-painting probes. Mol Reprod Dev 67: 108–115.
Evsikov S, Verlinsky Y (1998) Mosaicism in the inner cell mass of human blastocysts. Hum Reprod 11: 3151–3155.
Ford JH, Schultz CJ, Correll AT (1988) Chromosome elimination in micronuclei: a common cause of hypoploidy. Am J Hum Genet 43: 733–740.
Gallagher DS, Womack JE (1992) Chromosome conservation in the Bovidae. J Hered 83: 287–298.
Gearhart JD, Davisson MT, Oster-Granite ML (1986) Autosomal aneuploidy in mice: generation and developmental consequences. Brain Res Bull 16: 789–801.
Hare WC, Singh EL, Betteridge KJ et al. (1980) Chromosomal analysis of 159 bovine embryos collected 12 to 18 days after estrus. Can J Genet Cytol 22: 615–626.
Hayes H, Petit E, Dutrillaux B (1991) Comparison of RGB-banded karyotypes of cattle, sheep and goats. Cytogenet Cell Genet 57: 51–55.
Hayes H, Petit E, Bouinol C, Popescu P (1993) Localization of the alpha S2-casein gene (CASAS2) to the homologous cattle, sheep and goat chromosome 4 by in situ hybridization. Cytogenet Cell Genet 64: 281–285.
Iannuzzi L, Di Meo GP (1995) Chromosomal evolution in bovids: a comparison of cattle, sheep and goat G- and R-banded chromosomes and cytogenetic divergences among cattle, goat and river buffalo sex chromosomes. Chromosome Res 3: 291–299.
Iannuzzi L, Di Meo GP, Perucatti A, Schibler L, Cribiu EP (2000) Comparative FISH mapping of bovine X chromosomes reveals homologies and divergences between the subfamilies Bovinae and Caprinae. Cytogenet Cell Genet 89: 171–176.
ISCNDB (1989) International System for Chromosome Nomenclature of Domestic Bovids. Cytogenet Cell Genet 53: 65–79.
ISCNDB (2000) International System for Chromosome Nomenclature of Domestic Bovids. Cytogenet Cell Genet 92: 283–299.
Iwasaki S, Hamano S, Kuwayama M et al. (1992) Developmental changes in the incidence of chromosome anomalies of bovine embryos fertilized in vitro. J Exp Zool 261: 79–85.
Jacobs PA, Hassold TJ, Henry A, Pettay D, Takaesu N (1987) Trisomy 13 ascertained in a survey of spontaneous abortions. J Med Genet 24: 721–7244.
James RM, Klerkx AH, Keighren M, Flockhart JH, West JD (1995) Restricted distribution of tetraploid cells in mouse tetraploid⇔diploid chimaeras. Dev Biol 167: 213–226.
King WA (1990) Chromosome abnormalities and pregnancy failure in domestic animals. Adv Vet Sci Comp Med 34: 229–250.
Lightfoot DA, Kouznetsova A, Mahdy E, Wilbertz J, Hoog C (2006) The fate of mosaic aneuploid embryos during mouse development. Dev Biol 289: 384–394.
Long CR, Pinto-Correia C, Duby RT et al. (1993) Chromatin and microtubule morphology during the first cell cycle in bovine zygotes. Mol Reprod Dev 36: 23–32.
Long SE, Williams CV (1982) A comparison of the chromosome complement of inner cell mass and trophoblast cells in day-10 pig embryos. J Reprod Fertil 66: 645–648.
MacAuley A, Cross JC, Werb Z (1998) Reprogramming the cell cycle for endoreduplication in rodent trophoblast cells. Mol Biol Cell 9: 795–807.
McGaughey RW, Polge C (1971) Cytogenetic analysis of pig oocytes matured in vitro. J Exp Zool 76: 383–395.
Morris LHA (1998) Effect of male in an in vitro embryo production system. DVSc thesis, University of Guelph, Guelph, Ontario, Canada.
Murray JD, Moran C, Boland MP et al. (1986) Polyploid cells in blastocycts and early fetuses from Australian Merino sheep. J Reprod Fertil 78: 439–446.
O’Brian JK, Catt SL, Ireland KA, Maxwell WMC, Evans G (1997) In vitro and in vivo developmental capacity of oocytes from prepubertal and adult sheep. Theriogenology 47: 1433–1443.
Peura T, Kleemann D, Rudiger S, Nattrass G, McLaughlan C, Walker S (2003) Effect of nutrition of oocyte donor on the outcomes of somatic cell nuclear transfer in the sheep. Biol Reprod 68: 45–50.
Plachot M (1989) Chromosome analysis of spontaneous abortions after IVF: a European survey. Hum Reprod 4: 425–429.
Ptak G, Loi P, Dattena M, Tischner M, Cappai P (1999) Offspring from one-month old lambs: studies on the developmental capability of prepubertal oocytes. Biol Reprod 61: 1568–1574.
Rambags BP, Krijtenburg PJ, Drie HF et al. (2005) Numerical chromosomal abnormalities in equine embryos produced in vivo and in vitro. Mol Reprod Dev 72: 77–87.
Ruangvutilert P, Delhanty JD, Serhal P, Simopoulou M, Rodeck CH, Harper JC (2000) FISH analysis on day 5 post-insemination of human arrested and blastocyst stage embryo. Prenat Diagn 20: 552–560.
Rubes J, Vozdova M, Oracova E, Perreault SD (2005) Individual variation in the frequency of sperm aneuploidy in humans. Cytogenet Genome Res 111: 229–236.
Sandalinas M, Sadowy S, Alikani M, Calderon G, Cohen J, Munne S (2001) Developmental ability of chromosomally abnormal human embryos to develop to the blastocyst stage. Hum Reprod 16: 1954–1958.
Schatten G, Simerly C, Schatten H (1985) Microtubule configurations during fertilization, mitosis, and early development in the mouse and the requirement for egg microtubule-mediated motility during mammalian fertilization. Proc Natl Acad Sci USA 82: 4152–4156.
Schatten G, Simerly C, Schatten H (1986) Microtubules in mouse oocytes, zygotes, and embryos during fertilization and early development: unusual configurations and arrest of mammalian fertilization with microtubule inhibitors. Ann N Y Acad Sci 466: 945-948.
Shi Q, King RW (2005) Chromosome nondisjunction yields tetraploid rather than aneuploid cells in human cell lines. Nature 437: 1038–1042.
Simerly C, Wu GJ, Zoran S et al. (1995) The paternal inheritance of the centrosome, the cell’s microtubule-organizing center, in humans, and the implications for infertility. Nat Med 1: 47–52.
Veiga A, Gil Y, Boada M et al. (1999) Confirmation of diagnosis in preimplantation genetic diagnosis (PGD) through blastocyst culture: preliminary experience. Prenat Diagn 19: 1242–1247.
Viuff D, Rickords L, Offenberg H et al. (1999) A high proportion of bovine blastocysts produced in vitro are mixoploid. Biol Reprod 60: 1273–12788.
Viuff D, Greve T, Avery B , Hyttel P, Brockhoff PB, Thomsen PD (2000) Chromosome aberrations in in-vitro-produced bovine embryos at days 2–5 post-insemination. Biol Reprod 63: 1143–1148.
Viuff D, Hendriksen PJ, Vos PL et al. (2001) Chromosomal abnormalities and developmental kinetics in in-vivo-developed cattle embryos at days 2 to 5 after ovulation. Biol Reprod 65: 204–208.
Viuff D, Palsgaard A, Rickords L et al. (2002) Bovine embryos contain a higher proportion of polyploid cells in the trophectoderm than in the embryonic disc. Mol Reprod Dev 62: 483–488.
Walker S, Hill J, Kleeman D, Nancarrow C (1996) Development of ovine embryos in synthetic oviductal fluid containing amino acids at oviductal fluid concentrations. Biol Reprod 55: 703–708.
Wilcox AJ, Weinberg CR, O’Connor JF et al. (1988) Incidence of early loss of pregnancy. N Engl J Med 319: 189–194.
Zudova D, Rezacova O, Kubickova S, Rubes J (2003) Aneuploidy detection in porcine embryos using fluorescence in situ hybridization. Cytogenet Genome Res 102: 179–183.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coppola, G., Alexander, B., Di Berardino, D. et al. Use of cross-species in-situ hybridization (ZOO-FISH) to assess chromosome abnormalities in day-6 in-vivo- or in-vitro-produced sheep embryos. Chromosome Res 15, 399–408 (2007). https://doi.org/10.1007/s10577-007-1125-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-007-1125-2