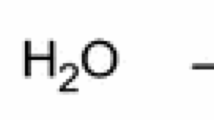Abstract
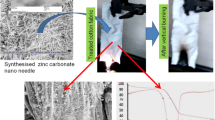
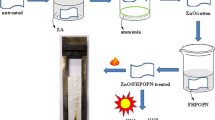
Zinc carbonate hydroxide [ZnCO3·Zn(OH)2] synthesized by means of the multiple-bath method was deposited onto a cotton fabric, and its uniformity was ensured by means of squeeze rolls. Prolonged burning was observed on treated specimens: 200.5 sec for 20.20% of ZnCO3·Zn(OH)2 added to 100 g of a dry fabric increased to 337.5 sec for a 45.39% addition. The ashes of the treated specimens were subjected to X-ray diffraction analysis, and the result was compared with data for zinc and zinc-oxide powders. The existence of zinc oxide was detected in the ashes, but no traces of the metallic zinc were discovered. Therefore, it can plausibly be assumed that a reduction-oxidation reaction occurs during the smoldering process.
Similar content being viewed by others
REFERENCES
C. Martin, “In the line of fire,” Chem. Brit., 34, 20 (1998).
C. E. Housecroft and A. G. Sharpe, Inorganic Chemistry, Pearson Education, London (2001).
S. M. Mostashari, “Effect of deposited zinc borate on the flame-retardancy imparted to cotton fabric, ” Int. J. Chem., 13, 115 (2003).
J. E. Ramsbottom, Fire-Proofing of Fabric, His Majesty’s Stationary Office, London (1947).
F. M. Farhan, S. M. Mostashari, and G. Ghazi Moghadam, “Effect of selected inorganic chlorides on the flame-retardancy imparted to cotton fabric,” Int. J. Chem., 1, 117–121 (1990).
F. M. Farhan, S. M. Mostashari, and G. Ghazi Moghadam, “Effect of deposited magnesium hydroxide or magnesium carbonate on the flame-retardancy imparted to cotton fabric,” Int. J. Chem., 2, No.4, 163–166 (1991).
J. G. Frick, J. David Reid, Jr., and H. B. Moore, “Fluorine and iodine compounds as flame retardants for cotton,” Text. Res. J., 26, 525 (1956).
Y. L. Hsieh and I. R. Haradin, “Effect of selected inorganic salts on cotton flammability,” Text. Res. J., 54, 171–179 (1984).
S. M. Mostashari, “The production of flame-retarded acetate rayon,” M. Phil. Thesis, Univ. of Leeds, Leeds (1978).
K. N. Vpadhyaya, A Text Book of Inorganic Chemistry, Vikas, New Delhi (1998).
U. S. Department of Commerce Standard for Flammability of Children’s Sleepwear (DOC FF 3-71), Federal Register, Vol. 36, No.146, July 19 (1971).
R. E. Kirke and D. F. Othmer, Encyclopedia of Chemical Technology, Vol. 16, Wiley-Inter Science, New York (1981), p. 621.
S. M. Mostashari, “Some basic aspects on terminology concerning flammability,” Asian J. Chem., 16, 555–557 (2004).
D. Price, A. R. Horrocks, and M. Tunc, “Textile flammability,” Chem. Brit., 23, 235–240 (1987).
C. Z. Carroll Porezynsky, “Flammability of composite fabrics,” Ph. D. Thesis, Univ. of Leeds, Leeds (1972).
M. Kesner and W. de Vos, “Teaching about flame-retardants,” J. Chem. Educ., 78, No.1, 42 (2001).
Author information
Authors and Affiliations
Additional information
__________
Translated from Fizika Goreniya i Vzryva, Vol. 41, No. 4, pp. 73–77, July–August, 2005.
Rights and permissions
About this article
Cite this article
Mostashari, S.M., Zanjanchi, M.A. & Baghi, O. Burning of a Cotton Fabric Impregnated by Synthetic Zinc Carbonate Hydroxide as a Flame Retardant. Combust Explos Shock Waves 41, 426–429 (2005). https://doi.org/10.1007/s10573-005-0052-9
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s10573-005-0052-9