Abstract
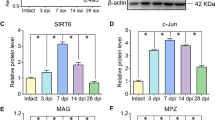
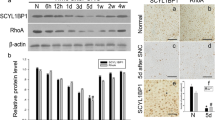
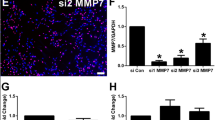
PPARγ coactivator-1 alpha (PGC-1α) is an essential transcription factor co-activator that regulates gene transcription and neural regeneration. Schwann cells, which are unique glial cells in peripheral nerves that dedifferentiate after peripheral nerve injury (PNI) and are released from degenerative nerves. Wallerian degeneration is a series of stereotypical events that occurs in response to nerve fibers after PNI. The role of PGC-1α in Schwann cell dedifferentiation and Wallerian degeneration is not yet clear. As Wallerian degeneration plays a crucial role in PNI, we conducted a study to determine whether PGC-1α has an effect on peripheral nerve degeneration after injury. We examined the expression of PGC-1α after sciatic nerve crush or transection using Western blotting and found that PGC-1α expression increased after PNI. Then we utilized ex vivo and in vitro models to investigate the effects of PGC-1α inhibition and activation on Schwann cell dedifferentiation and nerve degeneration. Our findings indicate that PGC-1α negatively regulates Schwann cell dedifferentiation and nerve degeneration. Through the use of RNA-seq, siRNA/plasmid transfection and reversal experiments, we identified that PGC-1α targets inhibit the expression of paraoxonase 1 (PON1) during Schwann cell dedifferentiation in degenerated nerves. In summary, PGC-1α plays a crucial role in preventing Schwann cell dedifferentiation and its activation can reduce peripheral nerve degeneration by targeting PON1.
Graphical Abstract
PGC-1α inhibits Schwann cell dedifferentiation and peripheral nerve degeneration. PGC-1α negatively regulates Schwann cell dedifferentiation and peripheral nerve degeneration after injury by targeting PON1.









Similar content being viewed by others
Data Availability
The data that support the findings are available from the corresponding author upon reasonable request.
References
Catenaccio A, Llavero Hurtado M, Diaz P, Lamont DJ, Wishart TM, Court FA (2017) Molecular analysis of axonal-intrinsic and glial-associated co-regulation of axon degeneration. Cell Death Dis 8(11):e3166. https://doi.org/10.1038/cddis.2017.489
Chandrasekaran K, Anjaneyulu M, Choi J, Kumar P, Salimian M, Ho CY, Russell JW (2019) Role of mitochondria in diabetic peripheral neuropathy: influencing the NAD(+)-dependent SIRT1-PGC-1α-TFAM pathway. Int Rev Neurobiol 145:177–209. https://doi.org/10.1016/bs.irn.2019.04.002
Chen P, Piao X, Bonaldo P (2015) Role of macrophages in Wallerian degeneration and axonal regeneration after peripheral nerve injury. Acta Neuropathol 130(5):605–618. https://doi.org/10.1007/s00401-015-1482-4
Cowell RM, Blake KR, Inoue T, Russell JW (2008) Regulation of PGC-1alpha and PGC-1alpha-responsive genes with forskolin-induced Schwann cell differentiation. Neurosci Lett 439(3):269–274. https://doi.org/10.1016/j.neulet.2008.04.104
Han B, Jiang W, Cui P, Zheng K, Dang C, Wang J, Li H, Chen L, Zhang R, Wang QM, Ju Z, Hao J (2021) Microglial PGC-1α protects against ischemic brain injury by suppressing neuroinflammation. Genome Med 13(1):47. https://doi.org/10.1186/s13073-021-00863-5
Jang SY, Yoon BA, Shin YK, Yun SH, Jo YR, Choi YY, Ahn M, Shin T, Park JI, Kim JK, Park HT (2017) Schwann cell dedifferentiation-associated demyelination leads to exocytotic myelin clearance in inflammatory segmental demyelination. Glia 65(11):1848–1862. https://doi.org/10.1002/glia.23200
Jessen KR, Arthur-Farraj P (2019) Repair Schwann cell update: adaptive reprogramming, EMT and stemness in regenerating nerves. Glia 67(3):421–437. https://doi.org/10.1002/glia.23532
Kotur-Stevuljevic J, Spasic S, Jelic-Ivanovic Z, Spasojevic-Kalimanovska V, Stefanovic A, Vujovic A, Memon L, Kalimanovska-Ostric D (2008) PON1 status is influenced by oxidative stress and inflammation in coronary heart disease patients. Clin Biochem 41(13):1067–1073. https://doi.org/10.1016/j.clinbiochem.2008.06.009
Monje PV (2018) Scalable differentiation and dedifferentiation assays using neuron-free Schwann cell cultures. Methods Mol Biol 1739:213–232. https://doi.org/10.1007/978-1-4939-7649-2_14
Namgung U (2014) The role of Schwann cell-axon interaction in peripheral nerve regeneration. Cells Tissues Organs 200(1):6–12. https://doi.org/10.1159/000370324
Nocera G, Jacob C (2020) Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol Life Sci 77(20):3977–3989. https://doi.org/10.1007/s00018-020-03516-9
Rius-Pérez S, Torres-Cuevas I, Millán I, Ortega ÁL, Pérez S (2020) PGC-1α, inflammation and oxidative stress: an integrative view in metabolism. Oxid Med Cell Longev 2020:1452696. https://doi.org/10.1155/2020/1452696
Stierli S, Imperatore V, Lloyd AC (2019) Schwann cell plasticity-roles in tissue homeostasis, regeneration and disease. Glia 67(11):2203–2215. https://doi.org/10.1002/glia.23643
Thomas GA (1948) Quantitative histology of Wallerian degeneration: II Nuclear population in two nerves of different fibre spectrum. J Anat 82(3):135–145
Wong KM, Babetto E, Beirowski B (2017) Axon degeneration: make the Schwann cell great again. Neural Regen Res 12(4):518–524. https://doi.org/10.4103/1673-5374.205000
Yang DP, Zhang DP, Mak KS, Bonder DE, Pomeroy SL, Kim HA (2008) Schwann cell proliferation during Wallerian degeneration is not necessary for regeneration and remyelination of the peripheral nerves: axon-dependent removal of newly generated Schwann cells by apoptosis. Mol Cell Neurosci 38(1):80–88. https://doi.org/10.1016/j.mcn.2008.01.017
Yang C, Sui G, Li D, Wang L, Zhang S, Lei P, Chen Z, Wang F (2021) Exogenous IGF-1 alleviates depression-like behavior and hippocampal mitochondrial dysfunction in high-fat diet mice. Physiol Behav 229:113236. https://doi.org/10.1016/j.physbeh.2020.113236
You Y, Hou Y, Zhai X, Li Z, Li L, Zhao Y, Zhao J (2016) Protective effects of PGC-1α via the mitochondrial pathway in rat brains after intracerebral hemorrhage. Brain Res 1646:34–43. https://doi.org/10.1016/j.brainres.2016.04.076
Zhang L, Dong W, Ma Y, Bai L, Zhang X, Sun C, Li J, Zhang L (2022) Pon1 deficiency promotes Trem2 pathway-mediated microglial phagocytosis and inhibits pro-inflammatory cytokines release in vitro and in vivo. Mol Neurobiol 59(7):4612–4629. https://doi.org/10.1007/s12035-022-02827-1
Zou Y, Zhang J, Liu J, Xu J, Fu L, Ma X, Xu Y, Xu S, Wang X, Guo J (2022) SIRT6 negatively regulates schwann cells dedifferentiation via targeting c-jun during wallerian degeneration after peripheral nerve injury. Mol Neurobiol 59(1):429–444. https://doi.org/10.1007/s12035-021-02607-3
Funding
This work was supported by the National Key R&D Program of China (No. 2021YFA0804903) and China Postdoctoral Science Foundation (No. 2022M721351).
Author information
Authors and Affiliations
Contributions
YZ, YG and SL conceived and designed the experiments. SW and FW provided experimental technical support and assisted in completing the study. YZ wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Jinan University (Date: 2022-06-16/No: 20220616-05).
Informed Consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zou, Y., Wu, S., Wen, F. et al. PGC-1α Inhibits Schwann Cell Dedifferentiation and Delays Peripheral Nerve Degeneration by Targeting PON1. Cell Mol Neurobiol 43, 3767–3781 (2023). https://doi.org/10.1007/s10571-023-01395-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-023-01395-9