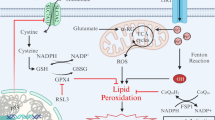
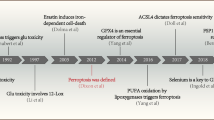
Abstract
The current report briefly summarizes the existing hypotheses and relevant evidence of oxytosis/ferroptosis-mediated cell death and outlines future perspectives of neurodegeneration research. Furthermore, it highlights the potential application of specific markers (e.g., activators, inhibitors, redox modulators, antioxidants, iron chelators) in the study of regulatory mechanisms of oxytosis/ferroptosis. It appears that these markers may be a suitable option for experimental investigations targeting key pathways of oxytosis/ferroptosis, such as the inhibition of the cystine/glutamate antiporter/glutathione/glutathione peroxidase 4 axis, glutamate oxidative toxicity, glutathione depletion, iron dyshomeostasis, iron-mediated lipid peroxidation, and others. From a clinical perspective, an innovative research approach to investigate the oxytosis/ferroptosis pathways in cells of the central nervous system and their relationship to neurodegenerative diseases is desirable. It is necessary to expand the existing knowledge about the molecular mechanisms of neurodegenerative diseases and to provide innovative diagnostic procedures to prevent their progression, as well as to develop effective neuroprotective treatment. The importance of preclinical studies focused predominantly on oxytosis/ferroptosis inhibitors (iron chelators or lipoxygenase inhibitors and lipophilic antioxidants) that could chelate iron or inhibit lipid peroxidation is also discussed. Specifically, this targeted inhibition of neuronal death could represent a potential therapeutic strategy for some neurodegenerative diseases.
Graphical Abstract

Similar content being viewed by others
Data Availability
Data sharing does not applicable to this article as no datasets were generated or analyzed during the current study.
Abbreviations
- CNS:
-
Central nervous system
- Fer-1:
-
Ferrostatin-1
- FINs:
-
Ferroptosis-inducing compound
- FSP1:
-
Ferroptosis suppressor protein
- GSH:
-
Glutathione
- GPX4:
-
Glutathione peroxidase 4
- Lip-1:
-
Liproxstatin
- LPO:
-
Lipid peroxidation
- LOX:
-
Lipoxygenase
- PEBP1:
-
Phosphatidylethanolamine-binding protein 1
- PUFAs:
-
Polyunsaturated fatty acids
- ROS:
-
Reactive oxygen species
- Xc¯:
-
Cystine/glutamate antiporter
References
Abrams RP, Carrol WL, Woerpel KA (2016) Five-membered ring peroxide selectively initiates ferroptosis in cancers. ACS Chem Biol 11:1305–1312
Angelova PR, Choi ML, Berezhnov AV, Horrocks MH, Hughes CD, De S et al (2020) Alpha synuclein aggregation drives ferroptosis: an interplay of iron, calcium and lipid peroxidation. Cell Death Differ 27:2781–2796
Ashraf A, Jeandriens J, Parkes HG, So PW (2020) Iron dyshomeostasis, lipid peroxidation and perturbed expression of cystine/glutamate antiporter in Alzheimer’s disease: evidence of ferroptosis. Redox Biol 32:101494
Ayton S, Portbury S, Kalinowski P, Agarwal P, Diouf I, Schneider JA et al (2021) Regional brain iron associated with deterioration in Alzheimer’s disease: a large cohort study and theoretical significance. Alzheimer’s Dement 17:1244–1256
Bersuker K, Hendricks JM, Li Z, Magtanang L, Ford B, Tang PH et al (2019) The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575:688–692
Chen L, Hambright WS, Na R, Ran Q (2015) Ablation of the ferroptosis inhibitors glutathione peroxidase 4 in neurons results in rapid motor neuron degeneration and paralysis. J Biol Chem 290:28097–28106
Chen X, Kang R, Kroemer G, Tang D (2021) Organelle-specific regulation of ferroptosis. Cell Death Differ 28:2843–2856
Czapski GA, Czubowicz K, Strosznajder RP (2012) Evaluation of the antioxidative properties of lipoxygenase inhibitors. Pharmacol Rep 64:1179–1188
Dixon SJ, Patel DN, Welsch M, Skouta R, Lee ED, Hayano M et al (2014) Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. Elife 3:1–25
Doll S, Freitas FP, Shah R, Aldrovandi M, Costa de Silva M, Ingold I et al (2019) FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575:693–698
Feng W, Xiao Y, Zhao C, Zhang Z, Liu W, Ma J et al (2022) New deferric amine compounds efficiently chelate excess iron to treat iron overload disorders and to prevent ferroptosis. Adv Sci 9:e2202679
Friedman Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ et al (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16:1180–1191
Gaschler MM, Andia AA, Liu H, Csuka JM, Hurlocker B, Vaiana CA et al (2018) FINO2 initiates ferroptosis through GPX4 inactivation and iron oxidation. Nat Chem Biol 14:507–515
Huang L, McClatchy DB, Maher P, Liang Z, Diedrich JK, Soriano-Castell D et al (2020) Intracellular amyloid toxicity induces oxytosis/ferroptosis regulated cell death. Cell Death Dis 11:828
Karuppagounder SS, Alin L, Chen Y, Brand D, Bourassa MW, Dietrich K et al (2018) N-acetylcysteine targets 5 lipoxygenase-derived, toxic lipids and can synergize with prostaglandin E2 to inhibit ferroptosis and improve outcomes following hemorrhagic stroke in mice. Ann Neurol 84:854–872
Landshamer S, Hoehn M, Barth N, Duvezin-Caubet S, Schwake G, Tobaben S et al (2008) Bid-induced release of AIF from mitochondria causes immediate neuronal cell death. Cell Death Differ 15:1553–1563
Lewerenz J, Hewett SJ, Huang Y, Lambros M, Gout PW, Kalivas PW et al (2013) The cystine/glutamate antiporter system xc− in health and disease: from molecular mechanisms to novel therapeutic opportunities. Antioxid Redox Signal 18:522–555
Lewerenz J, Ates G, Methner A, Conrad M, Maher P (2018) Oxytosis/ferroptosis-(re-) emerging roles for oxidative stress-dependent non-apoptotic cell death in diseases of the central nervous system. Front Neurosci 12:214
Maher P, Currais A, Schubert D (2020) Using the oxytosis/ferroptosis pathway to understand and treat age-associated neurodegenerative diseases. Cell Chem Biol 27:1456–1471
Marchetti L, Klein M, Schlett K, Pfizenmaier K, Eisel ULM (2004) Tumor necrosis factor (TNF)-mediated neuroprotection against glutamate-induced excitotoxicity is enhanced by N-methyl-D-aspartate receptor activation: essential role of TNF receptor 2-mediated phosphatidylinositol 3-kinase-dependent NF-kappaB pathway. J Biol Chem 279:32869–32881
Mariani E, Polidori MC, Cherubini A, Mecocci P (2005) Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. J Chromatogr B Analyt Technol Biomed Life Sci 827:65–75
Martin WRW, Wieler M, Gee M (2008) Midbrain iron content in early Parkinson disease. A potential biomarker of disease status. Neurology 70:1411–1417
Neitemeier S, Jelinek A, Laino V, Hoffman L, Eisenbach I, Eying R et al (2017) BID links ferroptosis to mitochondrial cell death pathways. Redox Biol 12:558–570
Obrocki P, Khatun A, Ness D, Senkevich K, Hanrieder J, Capraro F et al (2020) Perspectives in fluid biomarkers in neurodegeneration from the 2019 biomarkers in neurodegenerative diseases course - a joint PhD student course at University College London and University of Gothenburg. Alzheimer Res Ther 12:20
Pedrera L, Espiritu RA, Ros U, Weber J, Schmitt A, Stroh J et al (2021) Ferroptotic pores induce Ca2+ fluxes and ESCRT-III activation to modulate cell death kinetics. Cell Death Differ 28:1644–1657
Qin D, Wang J, Le A, Wang TJ, Chen X, Wang J (2021) Traumatic brain injury: ultrastructural features in neuronal ferroptosis, glial cell activation, and polarization, and blood-brain barrier breakdown. Cells 10:1009
Ratan RR (2020) The chemical biology of ferroptosis in the central nervous system. Cell Chem Biol 27:479–498
Rockfield S, Chhabra R, Robertson M, Rehman N, Bisht R, Nanjundan M (2018) Links between iron and lipids: Implications in some major human diseases. Pharmaceuticals 11:113
Shimada K, Skouta R, Kaplan A, Yang WS, Hayano M, Dixon SJ et al (2016) Global survey of cell death mechanisms reveals metabolic regulation of ferroptosis. Nature Chem Biol 12:497–503
Skouta R, Dixon SJ, Wang J, Dunn DE, Orman M, Shimada K et al (2014) Ferrostatins inhibit oxidative lipid damage and cell death in diverse disease models. J Am Chem Soc 136:4551–4556
Soriano-Castell D, Currais A, Maher P (2021) Defining a pharmacological inhibitor fingerprint for oxytosis/ferroptosis. Free Radic Biol Med 171:219–213
Sun Y, Zheng Y, Wang C, Liu Y (2018) Glutathione depletion induces ferroptosis, autophagy, and premature cell senescence in retinal pigment epithelial cells. Cell Death Dis 9:753
Sun Y, Berleth N, Wu W, Schlutermann D, Deitersen J, Stuhldreier F et al (2021) FIN56-induced ferroptosis is supported by autophagy-mediated GPX4 degradation and functions synergistically with mTOR inhibition to kill bladder cancer cells. Cell Death Dis 12:1028
Tan S, Schubert D, Maher P et al (2001) Oxytosis: a novel form of programmed cell death. Curr Top Med Chem 1:497–506
Ulatowski LM, Manor D (2015) Vitamin E and neurodegeneration. Neurobiol Dis 84:78–83
Ward RJ, Zucca FA, Duyn JH, Crichton RR, Zecca L (2014) The role of iron in brain ageing and neurodegenerative disorders. Lancet Neurol 13:1045–1060
Wenzel SE, Tyutina YY, Zhao J, Croix CMS, Dar HH, Mao G et al (2017) PEBP1 wardens ferroptosis by enabling lipoxygenase generation of lipid death signals. Cell 171:628–641
Xie Y, Song X, Sun X, Huang J, Zhong M, Lotze MT et al (2016) Identification of baicalein as a ferroptosis inhibitor by natural product library screening. Biochem Biophys Res Commun 473:775–780
Yang WS, SriRamaratnam R, Welsch ME, Skouta R, Viswanathan VS, Cheah JH et al (2014) Regulation of ferroptotic cancer cell death by GPX4. Cell 156:317–331
Yang K, Zeng L, Yuan X, Wang S, Ge A, Xu H, Zeng J (2022) The mechanism of ferroptosis regulating oxidative stress in ischemic stroke and the regulation mechanism of natural pharmacological active compounds. Biomed Pharmacother 154:113611
Zeng X, An H, Yu F, Wang K, Zheng L, Zhou W et al (2021) Benefits of iron chelators in the treatment of Parkinson’s disease. Neurochem Res 46:1239–1251
Zhang Y, Swanda RV, Nie C, Koppula P, Cheng W, Zhang J et al (2021a) mTORC1 couples cyst(e)ine availability with GPX4 protein synthesis and ferroptosis regulation. Nat Commun 12:1589
Zhang Y, Lu X, Tai B, Li W, Li T (2021b) Ferroptosis and its multifaceted roles in cerebral stroke. Front Cell Neurosci 15:615372
Zhao J, Dar HH, Deng Y, St. Croix CM, Li Z, Minami Y et al (2020) PEBP1 acts as a rheostat between prosurvival autophagy and ferroptotic death in asthmatic epithelial cells. Proc Natl Acad Sci USA 117:14376–14385
Zilka O, Shah R, Li B, Friedmann Angeli JP, Griesser M, Conrad M et al (2017) On the mechanism of cytoprotection by ferrostatin-1 and liproxstatin-1 and the role of lipid peroxidation in ferroptotic cell death. ACS Cent Sci 3:232–243
Zille M, Karuppagounder SS, Chen Y, Gough PJ, Bertin J, Finger J, Milner TA, Jonas EA, Ratan RR (2017) Neuronal death after hemorrhagic stroke in vitro and in vivo shares feature of ferroptosis and necrosis. Stroke 48:1033–1043
Zou Y, Li H, Graham ET, Deik AA, Eaton JK, Wang W et al (2020) Cytochrome P450 oxidoreductase contributes to phospholipid peroxidation in ferroptosis. Nat Chem Biol 16:302–309
Funding
No funding was obtained for this study.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Competing interests
The author has no relevant financial or non-financial interests to disclose.
Ethical Approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Viktorinova, A. Future Perspectives of Oxytosis/Ferroptosis Research in Neurodegeneration Diseases. Cell Mol Neurobiol 43, 2761–2768 (2023). https://doi.org/10.1007/s10571-023-01353-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-023-01353-5