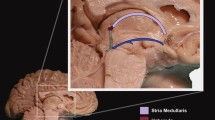
Abstract
Is the cerebrum involved in its own activation to states of attention or arousal? “Telencephalon” is a term borrowed from embryology to identify not only the cerebral hemispheres of the forebrain, but also the basal forebrain. We review a generally undercited literature that describes nucleus basalis of Meynert, located within the substantia innominata of the ventrobasal forebrain, as a telencephalic extension of the ascending reticular activating formation. Although that formation’s precise anatomical definition and localization have proven elusive over more than 70 years, a careful reading of sources reveals that there are histological features common to certain brainstem neurons and those of the nucleus basalis, and that a largely common dendritic architecture may be a morphological aspect that helps to define non-telencephalic structures of the ascending reticular activating formation (e.g., in brainstem) as well as those parts of the formation that are telencephalic and themselves responsible for cortical activation. We draw attention to a pattern of dendritic arborization described as “isodendritic,” a uniform (isos-) branching in which distal dendrite branches are significantly longer than proximal ones. Isodendritic neurons also differ from other morphological types based on their heterogeneous, rather than specific afferentation. References reviewed here are consistent in their descriptions of histology, particularly in studies of locales rich in cholinergic neurons. We discuss the therapeutic implications of a basal forebrain site that may activate cortex. Interventions that specifically target nucleus basalis and, especially, the survival of its constituent neurons may benefit afflictions in which higher cortical function is compromised due to disturbed arousal or attentiveness, including not only coma and related syndromes, but also conditions colloquially described as states of cognitive “fog” or of “long-haul” mental compromise.



Similar content being viewed by others
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
References
Alves PN, Foulon C, Karolis V, Bzdok D, Margulies DS, Volle E, Thiebaut de Schotten M (2019) An improved neuroanatomical model of the default-mode network reconciles previous neuroimaging and neuropathological findings. Commun Biol 2:370. https://doi.org/10.1038/s42003-019-0611-3
Arendt T, Brückner MK, Bigl V, Marcova L (1995) Dendritic reorganization in the basal forebrain under degenerative conditions and its defects in Alzheimer’s disease. 3. The basal forebrain compared with other subcortical areas. J Comp Neurol 351:223–246. https://doi.org/10.1002/cne.903510204
Arnold SE, Louneva N, Cao K, Wang L-S, Han L-Y, Wolk DA, Negash S, Leurgans SE, Schneider JA, Buchman AS, Wilson RS, Bennett DA (2013) Cellular, synaptic and biochemical features of resilient cognition in Alzheimer’s disease. Neurobiol Aging 34:157–168. https://doi.org/10.1016/j.neurobiolaging.2012.03.004
Ballinger EC, Ananth M, Talmage DA, Role LW (2016) Basal forebrain cholinergic circuits and signaling in cognition and cognitive decline. Neuron 91:1199–1218. https://doi.org/10.1016/j.neuron.2016.09.006
Benton JS, Bowen DM, Allen SJ, Haan EA, Davison AN, Neary D, Murphy DRP, Snowden JS (1982) Alzheimer’s disease as a disorder of isodendritic core. Lancet 319:456. https://doi.org/10.1016/s0140-6736(82)91667-1
Bishop GH (1958) The place of cortex in a reticular system. In: Jasper HH, Proctor LD, Knighton RS, Noshay WC, Costello RT (eds) Reticular Formation of the Brain. Little, Brown and Company, Boston, pp 413–421
Bloem B, Schoppink L, Rotaro DC, Faiz A, Hendriks P, Mansvelder HD, van de Berg WDJ, Wouterlood FG (2014) Topographic mapping between basal forebrain cholinergic neurons and the medial prefrontal cortex in mice. J Neurosci 34:16234–16246. https://doi.org/10.1523/JNEUROSCI.3011-14.2014
Bremer F (1937) L’activité cérébrale au cours du sommeil et de la narcose. Contribution à l’étude du mécanisme du sommeil. Bull Acad Roy Med Belgique 2:68–86
Bucci D, Busceti CL, Calierno MT, Di Pietro P, Madonna M, Biagioni F, Ryskalin L, Limanaqi F, Nicoletti F, Fornai F (2017) Systematic morphometry of catecholamine nuclei in the brainstem. Front Neuroanat 11:98. https://doi.org/10.3389/fnana.2017.00098
Cadet JL (1984) Disorders of the isodendritic core of the brainstem. Schizophr Bull 10:1–3. https://doi.org/10.1093/schbul/10.1.1
Cadet JL (1988) A unifying theory of movement and madness: involvement of free radicals in disorders of the isodendritic core of the brainstem. Med Hypotheses 27:59–63. https://doi.org/10.1016/0306-9877(88)90085-0
Duque A, Tepper JM, Detari L, Ascoli GA, Zaborsky L (2007) Morphological characterization of electrophysiologically and immunohistochemically identified basal forebrain cholinergic and neuropeptide Y-containing neurons. Brain Struct Funct 212:55–73. https://doi.org/10.1007/s00429-007-0143-3
Edlow BL, Takahashi E, Wu O, Benner T, Dai G, Bu L, Grant PE, Greer DM, Greenberg SM, Kinney HC, Folkerth RD (2012) Neuroanatomic connectivity of the human ascending arousal system critical to consciousness and its disorders. J Neuropathol Exp Neuro 71:531–546. https://doi.org/10.1097/NEN.0b013e3182588293
Ehrenberg AJ, Nguy AK, Theofilas P, Dunlop S, Suemoto CK, Di Lorenzo Alho AT, Leite RP, Diehl Rodriguez R, Mejia MB, Rüb U, Farfel JM, de Lucena Ferretti-Rebustini RE, Nascimento CF, Nitrini R, Pasquallucci CA, Jacob-Filho W, Miller B, Seeley WW, Heinsen H, Grinberg LT (2017) Quantifying the accretion of hyperphosphorylated tau in the locus coeruleus and dorsal raphe nucleus: the pathological building blocks of early Alzheimer’s disease. Neuropathol Appl Neurobiol 43:393–408. https://doi.org/10.1111/nan.12387
Engelhardt E (2013) Meynert and the basal nucleus. Dement Neuropsychol 7:435–438. https://doi.org/10.1590/S1980-57642013DN7400013
Ertürk A, Becker K, Jährling N, Mauch CP, Hojer CD, Egen JG, Hellal F, Bradke F, Sheng M, Dodt H-U (2012) Three-dimensional imaging of solvent-cleared organs using 3DISCO. Nat Protoc 7:1983–1995. https://doi.org/10.1038/nprot.2012.119
Fuller P, Sherman D, Pedersen NP, Saper CB, Lu J (2011) Reassessment of the structural basis of the ascending arousal system. J Comp Neurol 519:933–956. https://doi.org/10.1002/cne.22559
Gage FH (2000) Mammalian neural stem cells. Science 287:1433–1438. https://doi.org/10.1126/science.287.5457.1433
Gebhart GF (1982) Opiate and opioid peptide effects on brain stem neurons: relevance to nociception and antinociception mechanisms. Pain 12:93–140. https://doi.org/10.1016/0304-3959(82)90189-0
Geser F (2021) The dendroarchitectonics of E. Ramon-Moliner [abstract]. Eur J Neurol 28suppl1:920.
Gielow MR, Zaborsky L (2017) The input-output relationship of the cholinergic basal forebrain. Cell Rep 18:1817–1830. https://doi.org/10.1016/j.celrep.2017.01.060
Golgi C (1906) The neuron doctrine—theory and facts. www.nobelprize.org/prizes/medicine/1906/golgi/lecture/>. Accessed 30 April 2022.
Greenfield JG, Bosanquet FD (1953) The brain-stem lesions in Parkinsonism. J Neurol Neurosurg Psychiat 16:213–226. https://doi.org/10.1136/jnnp.16.4.213
Halliday G, McCann H, Shepherd C (2012) Evaluation of the Braak hypothesis: how far can it explain the pathogenesis of Parkinson’s disease? Exp Rev Neurother 12:673–686. https://doi.org/10.1586/ern.12.47
Hardenacke K, Shubina E, Bührle CP, Zapf A, Lenartz D, Klosterkötter J, Visser-Vandewalle V, Kuhn J (2013) Deep brain stimulation as a tool for improving cognitive functioning in Alzheimer’s dementia: a systematic review. Front Psychiatry 4:159. https://doi.org/10.3389/fpsyt.2013.00159
Hobson JA (1980) Toward a cellular neurophysiology of the reticular formation: conceptual and methodological milestones. In: Hobson JA, Brazier MAB (eds) The Reticular Formation Revisited. Raven Press, New York, pp 7–29
Holden TJ (1989) An integrated neuropathophysiological model of schizophrenia. Med Hypotheses 28:109–113. https://doi.org/10.1016/0306-9877(89)90023-6
Jacobs HIL, Müller-Ehrenberg L, Priovoulos N, Roebroeck A (2018) Curvilinear locus coeruleus functional connectivity trajectories over the adult lifespan: a 7T MRI study. Neurobiol Aging 69:167–176. https://doi.org/10.1016/j.neurobiolaging.2018.05.021
Jasper H, Proctor LD, Knighton RS, Noshay WC, Costello RT (1958) Reticular Formation of the Brain. Little Brown, Boston
Jiang Y, Alam JJ, Gomperts SN, Maruff P, Lemstra AW, Germann UA, Stavrides PH, Darji S, Malampati S, Peddy J, Bleiwas C, Pawlik M, Pensalfini A, Yang D-S, Subbanna S, Basavarajappa BS, Smiley JF, Gardner A, Blackburn K, Chu H-M, Prins ND, Teunissen CE, Harrson JE, Scheltens P, Nixon RA (2022) Preclinical and randomized clinical evaluation of the p38α kinase inhibitor neflamapimod for basal forebrain cholinergic degeneration. Nature Comm 13:5308. https://doi.org/10.1038/s41467-022-32944-3
Khurana V, Tardiff DF, Chung CY, Lindquist S (2015) Toward stem cell-based phenotypic screens for neurodegenerative diseases. Nat Rev Neurol 11:339–350. https://doi.org/10.1038/nrneurol.2015.79
Kim J-H, Jung A-H, Jeong D, Choi H, Kim K, Shin S, Kim SJ, Lee S-H (2016) Selectivity of neuromodulatory projections from the basal forebrain and locus ceruleus to primary sensory cortices. J Neurosci 36:5314–5327. https://doi.org/10.1523/JNEUROSCI.4333-15.2016
Kline S, Felten DL (1985) Ventral tegmental area of the rabbit brain: a developmental Golgi study. Brain Res Bull 14:485–492. https://doi.org/10.1016/0361-9230(85)90027-9
Koelliker A (1896) Ausstrahlungen des rothen Kernes. Basales Ganglion (Ganglion der Ansa peduncularis, Meynert). In: Koelliker A Handbuch der Gewebelehre des Menschen. Zweiter Band, Wilhelm Engelmann, Leipzig, pp. 454–458.
Lagomarsino VN, Pearse RV, Liu L, Hsieh Y-C, Fernandez MA, Vinton EA, Pauli D, Felsky D, Tasaki S, Gaiteri C, Vardarajan B, Lee H, Muratore CR, Benoit CR, Chou V, Fancher SB, He A, Merchant JP, Duong DM, Martinez H, Zhou M, Bah F, Vicent MA, Stricker JMS, Xu J, Dammer EB, Levey AI, Chibnik LB, Menon V, Seyfried NT, De Jager PL, Noggie S, Selkoe DJ, Bennett DA, Young-Pearse TL (2021) Stem cell derived neurons reflect features of protein networks, neuropathology and cognitive outcome of their aged human donors. Neuron 109:3402-3420.e9. https://doi.org/10.1016/j.neuron.2021.08.003
Lees AJ (2009) The Parkinson chimera. Neurology 72(7):S2-11. https://doi.org/10.1212/WNL.0b013e318198daec
Leontovich TA, Zhukova GP (1963) The specificity of the neuronal structure and topography of the reticular formation in the brain and spinal cord of carnivora. J Comp Neurol 121:347–381. https://doi.org/10.1002/cne.901210305
Lewy FH (1923) Spezielle pathologie der paralysis agitans. In: Lewy FH Die Lehre vom Tonus under der Bewegung Zugleich Systematische Untersuchungen zur Klinik, Physiologie, Pathologie und Pathogenese der Paralysis Agitans, Julius Springer, Berlin, pp. 171–316.
Li X, Yu B, Sun Q, Zhang Y, Ren M, Zhang X, Li A, Yuan J, Madisen L, Luo Q, Zeng H, Gong H, Qiu Z (2018) Generation of a whole-brain atlas for the cholinergic system and mesoscopic projectome analysis of basal forebrain cholinergic neurons. PNAS 115:415–420. https://doi.org/10.1073/pnas.1703601115
Li J, Curley WH, Guerin B, Dougherty DD, Dalca AV, Fischl B, Horn A, Edlow BL (2021) Mapping the subcortical connectivity of the human default mode network. Neuroimage 245:118758. https://doi.org/10.1016/j.neuroimage.2021.118758
Lindsley DB, Bowden JW, Magoun HW (1949) Effect upon the EEG of acute injury to the brain stem activating system. Electroenceph Clin Neurophysiol 1:475–486
Liu RP, Hamilton BL (1976) Intranuclear bodies in neurons of the periaqueductal gray matter in the cat. Am J Anat 147:139–145. https://doi.org/10.1002/aja.1001470115
Liu AKL, Chang RCC, Pearce RKB, Gentleman SM (2015) Nucleus basalis of Meynert revisited: anatomy, history and differential involvement in Alzheimer’s and Parkinson’s disease. Acta Neuropathol 129:527–540. https://doi.org/10.1007/s00401-015-1392-5
Magoun HW (1964) Reticulo-cortical influences for wakefulness, orienting and attention. In: Charles C (ed) Magoun HW, The Waking Brain, 2nd. Thomas, Springfield, pp 74–97
Mannen H (1960) “Noyau fermé” et “noyau ouvert” Contribution à l’etude cytoarchitectonique du tronc cerebral envisage du point de vue du mode d’arborisation dendritique. Archiv Italiennes De Biologie 98:333–350
Mesulam MM, Geula C (1988) Nucleus basalis (Ch4) and cortical cholinergic innervation in the human brain: observations based on the distribution of acetylcholinesterase and choline acetyltransferase. J Comp Neurol 275:216–240. https://doi.org/10.1002/cne.902750205
Mesulam MM, van Hoesen GW (1976) Acetylcholinesterase-rich projections from the basal forebrain of the rhesus monkey to neocortex. Brain Res 109:152–157. https://doi.org/10.1016/0006-8993(76)90385-1
Mesulam MM, Geula C, Bothwell MA, Hersh LB (1989) Human reticular formation: cholinergic neurons of the pedunculopontine and laterodorsal tegmental nuclei and some cytochemical comparisons to forebrain cholinergic neurons. J Comp Neurol 281:611–633. https://doi.org/10.1002/cne.902830414
Meynert T (1872) Vom Gehirne der Säugetiere. In: Stricker S (ed) Meynert T. Handbuch der Lehre von den Geweben des Menschen under der Thiere. Zweiter Band. Wilhelm Engelmann, Leipzig, pp 694–808
Meynert T (1884) Formen und Zusammenhang des Gehirnes. Meynert T Psychiatrie. Klinik der Erkankungen des Vorderhirns. Erste Hälfte. Wilhelm Braumüller, Wien, pp 1–125
Moruzzi G, Magoun HW (1949) Brain stem reticular formation and activation of the EEG. Electroenceph Clin Neurophysiol 1:455–473
Newman DB, Liu RP (1987) Nuclear origins of brainstem reticulocortical systems in the rat. Am J Anat 178:279–299. https://doi.org/10.1002/aja.1001780309
Nieuwenhuys R (1985) Survey of chemically defined cell groups and pathways. Nieuwenhuys R Chemoarchitecture of the Brain, Springer-Verlag, Berlin, Heidelberg, Acetylcholine. In, pp 7–11
Parvisi J, Damasio A (2001) Consciousness and the brainstem. Cognition 79:135–159. https://doi.org/10.1016/s0010-0277(00)00127
Pearce JMS (2003) The nucleus of Theodor Meynert (1833–1892). J Neurol Neurosurg Psychiatry 74:1358. https://doi.org/10.1136/jnnp.74.9.1358
Ramón-Moliner E (1962) An attempt at classifying nerve cells on the basis of their dendritic patterns. J Comp Neurol 119:211–227. https://doi.org/10.1002/cne.901190207
Ramón-Moliner E, Nauta WJH (1966) The isodendritic core of the brain stem. J Comp Neurol 126:311–335. https://doi.org/10.1002/cne.901260301
Rossor MN (1981) Parkinson’s disease and Alzheimer’s disease as disorders of the isodendritic core. Br Med J 283:1588–1590. https://doi.org/10.1136/bmj.283.6306.1588
Rotolo T, Smallwood PM, Williams J, Nathans J (2008) Genetically-directed, cell type-specific sparse labeling for the analysis of neuronal morphology. PLoS ONE 3:e4099. https://doi.org/10.1371/journal.pone.0004009
Scheibel ME, Scheibel AB (1958) Structural substrates for integrative patterns in the brain stem reticular core. In: Jasper HH, Proctor LD, Knighton RS, Noshay WC, Costello RT (eds) Reticular Formation of the Brain. Little, Brown and Company, Boston, Toronto, pp 31–68
Seidel K, Mahlke J, Siswanto S, Krüger R, Heinsen H, Auburger G, Bouzrou M, Grinberg LT, Wicht H, Korf H-W, den Dunnen W (2015) The brainstem pathologies of Parkinson’s disease and dementia with Lewy bodies. Brain Path 25:121–135. https://doi.org/10.1111/bpa.12168
Swiech L, Heidenreich M, Banerjee A, Habib N, Li Y, Rombetta J, Sur M, Zhang F (2015) In vivo interrogation of gene function in the mammalian brain using CRISPR-Cas9. Nat Biotechnol 33:102–106. https://doi.org/10.1038/nbt.3055
The Human Brain Project, https://brains.anatomy.msu.edu/brains/human/coronal/1680_fiber.html. Accessed 01 May 2022.
Theofilas P, Dunlop S, Heinsen H, Grinberg LT (2015) Turning on the light within: subcortical nuclei of the isodendritic core and their role in Alzheimer’s disease pathogenesis. J Alzheimers Dis 46:17–34. https://doi.org/10.3233/JAD-142682
Vilalta A, Brown GC (2017) Neurophagy, the phagocytosis of live neurons and synapses by glia, contributes to brain development and disease. FEBS J 285:3541–3683. https://doi.org/10.1111/febs.14323
Wu H, Williams J, Nathans J (2014) Complete morphologies of basal forebrain cholinergic neurons in the mouse. Elife 3:e02444. https://doi.org/10.7554/eLife.02444
Yeo SS, Change PH, Jang SH (2013) The ascending reticular activating system from pontine reticular formation to the thalamus in the human brain. Front Hum Neurosci 7:416. https://doi.org/10.3389/fnhum.2013.00416
Zaborszky L, Duque A, Gielow M, Gombkoto P, Nadasdy Z, Somogyi J (2015) Organization of the basal forebrain cholinergic projection system: specific or diffuse? In: Paxinos G (ed) The Rat Nervous System. Elsevier, Amsterdam, pp 491–507
Zahm DS (2006) The evolving theory of basal forebrain functional-anatomical ‘macrosystems.’ Neurosci Biobehav Rev 30:148–172. https://doi.org/10.1016/jneubiorev.2005.06.003
Acknowledgements
Umberto DeGirolami, M.D. (Mass General Brigham Department of Pathology and Harvard Medical School) helped with the translation of Mannen (1960); he corroborated, corrected, and then elaborated on the first author’s translation. The authors thank Drs. Vikram Khurana and Tracy Young-Pearse, both from Mass General Brigham Department of Neurology and Harvard Medical School, for discussions relevant to the uses of induced pluripotent stem cells.
Funding
E.K.M. reports no funding sources. S.B. receives support from the Judy Pauline Staples Aull Fund at Brigham and Women’s Hospital, Boston, MA USA and research support from Alexion Pharmaceuticals and National Institute of Health under award number OT2HL161847-01. M.T. is supported by the National Cancer Institute of the National Institutes of Health under award number F32CA257210. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception. The literature review was performed by EKM, who also wrote the first draft of the manuscript. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miyawaki, E.K., Bhattacharyya, S. & Torre, M. Revisiting a Telencephalic Extent of the Ascending Reticular Activating System. Cell Mol Neurobiol 43, 2591–2602 (2023). https://doi.org/10.1007/s10571-023-01339-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-023-01339-3